Rifaximin uterine injection agent and preparation method and application thereof
A technology of rifaximin and injection, which is applied in the field of rifaximin uterine injection and its preparation and application, can solve the problem of insignificant effect, and achieve the effect of convenient and quick administration, high promotion value and significant curative effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0051] The rifaximin uterine injection provided by the invention is a uniformly distributed preparation, and the specific preparation method comprises the following steps:
[0052] A. Let the vegetable oil (such as neutral soybean oil) cool after filtration and heat sterilization;
[0053] B. Take an appropriate amount of pretreated vegetable oil and add 0.125-1.25 g of glyceryl monostearate;
[0054] C. Heat at 60-80°C for 15-30 minutes, wait for the glyceryl stearate to melt, stir evenly, cool to about 40°C, add 0.125-1.25g of suspending agent (such as Span 80), 0.05-0.5 g of rifaximin (particle size 5-10 μm), then add vegetable oil to a total of 25 g;
[0055] D. Grind evenly to prepare rifaximin uterine injection;
[0056] F. Put the medicine into the injection tube, seal it, sterilize it, inspect it with light, and pack it.
[0057]In the following examples, the detection method of the sedimentation volume ratio of the uterine injection: take 50ml of the test product w...
Embodiment 1
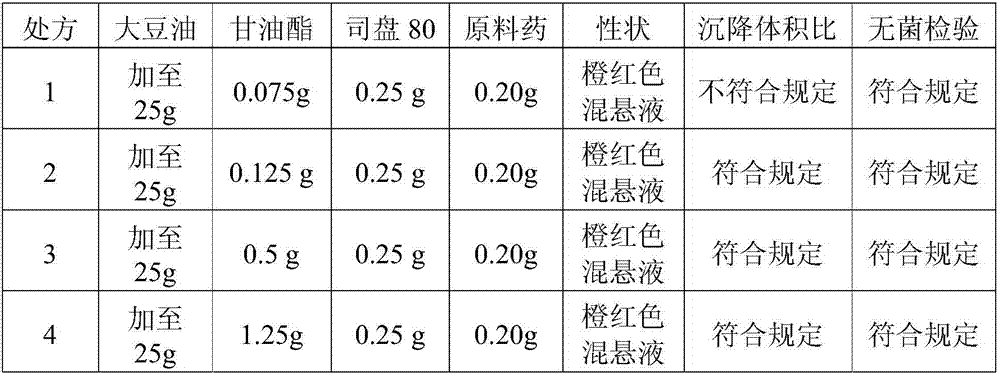
[0060] Embodiment 1, prescription screening of rifaximin uterine injection
[0061] 1. Screening of glyceryl monostearate content in the formula (note: glyceryl monostearate in the table is referred to as glyceride)
[0062] Table 1
[0063]
[0064] The preparation method is as follows:
[0065] 1) Obtain rifaximin aseptic micropowder (particle size: 5-10 μm) by superfinely pulverizing the aseptic rifaximin powder;
[0066] 2) After the soybean oil is filtered and heat sterilized (sterilized at 150° C. for 3 hours), it is placed at room temperature for use;
[0067] 3) Take an appropriate amount of pretreated soybean oil, add glyceryl stearate and suspending agent Span 80; heat and stir (heating at 80°C for 15 minutes), after the glyceryl stearate melts, add rifaxi Bright;
[0068] 5) Grind evenly in a sterile colloid mill to prepare rifaximin uterine injection.
[0069] As can be seen from table 1 result, when the content of glyceryl monostearate is 0.3% (w / w, g / g), ...
Embodiment 2
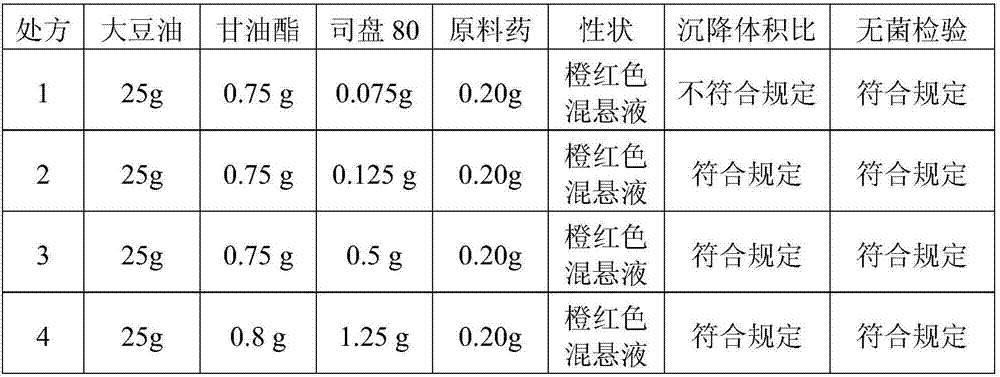
[0075] Embodiment 2, preparation rifaximin uterine injection
[0076]
[0077] The preparation method is as follows:
[0078] 1) Ultrafine pulverizing the rifaximin aseptic powder to obtain rifaximin aseptic micropowder with a particle size of 5-10 μm;
[0079] 2) After the soybean oil is filtered and heat sterilized (sterilized at 150° C. for 3 hours), it is placed at room temperature for use;
[0080] 3) Take an appropriate amount of pretreated soybean oil (40% of the total amount of soybean oil), add glyceryl monostearate; heat and stir (heating at 80°C for 15 minutes), and wait for glyceryl monostearate to melt , after stirring evenly, cool to 40°C, add Span 80, rifaximin, and add soybean oil to the full amount;
[0081] 4) Grind evenly in a sterile colloid mill to prepare rifaximin uterine injection.
[0082] The property of the obtained rifaximin uterine injection is orange-red to dark red oily suspension.
[0083] The sedimentation volume ratio and sterility test...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Particle size | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap