Extracyclic Z, E-diene-containing cis-tetrahydrobenzofuran compound and synthesis method thereof
A synthesis method and benzofuran technology are applied in the field of cis-tetrahydrobenzofuran structural compounds, can solve problems such as the method not being reported, and achieve the effects of reasonable reaction path, mild reaction conditions and high efficiency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Example Embodiment
[0040] Example 1:
[0041] The synthetic route of compound 3aa is as follows:
[0042]
[0043] The synthesis steps of compound 3aa are as follows:
[0044] The compound 1a (0.6mmol, 3.0eq), Cp*Rh(OAc) 2 -H 2 O (0.02mmol, 0.1eq), compound 2a (0.2mmol, 1.0eq), react at 60℃ until the material 2a disappears completely (about 9h) as monitored by TLC; spin dry the solvent, and separate by column chromatography (wash Removal agent: petroleum ether / ethyl acetate volume ratio 2:1) to obtain 55 mg of yellow solid 3aa, melting point 61-63°C, yield: 85%.
[0045] 1 H NMR(600MHz, CDCl 3 ): 7.32(t,J=7.5Hz,2H),7.24-7.20(m, 3H),7.09(dd,J=11.9,1.7Hz,1H),6.59(d,J=10.2Hz,1H),6.52 (d,J = 11.9Hz, 1H), 6.03 (d, J = 10.2Hz, 1H), 4.52-4.43(m, 2H), 3.68(s, 2H), 3.14-3.12(m, 1H), 2.59( dd, J = 16.4, 7.3 Hz, 1H), 2.50 (dd, J = 16.4, 6.1 Hz, 1H), 1.43 (s, 3H). 13 C NMR(100MHz, CDCl 3 ):197.1,171.8,150.8,149.5,138.9,137.4,129.1,128.8,126.7,118.8,79.6,70.9,46.0,40.2, 39.4,24.4.HRMS(ESI-TOF)m / z:[M+Na] + Calcd fo...
Example Embodiment
[0046] Example 2:
[0047]
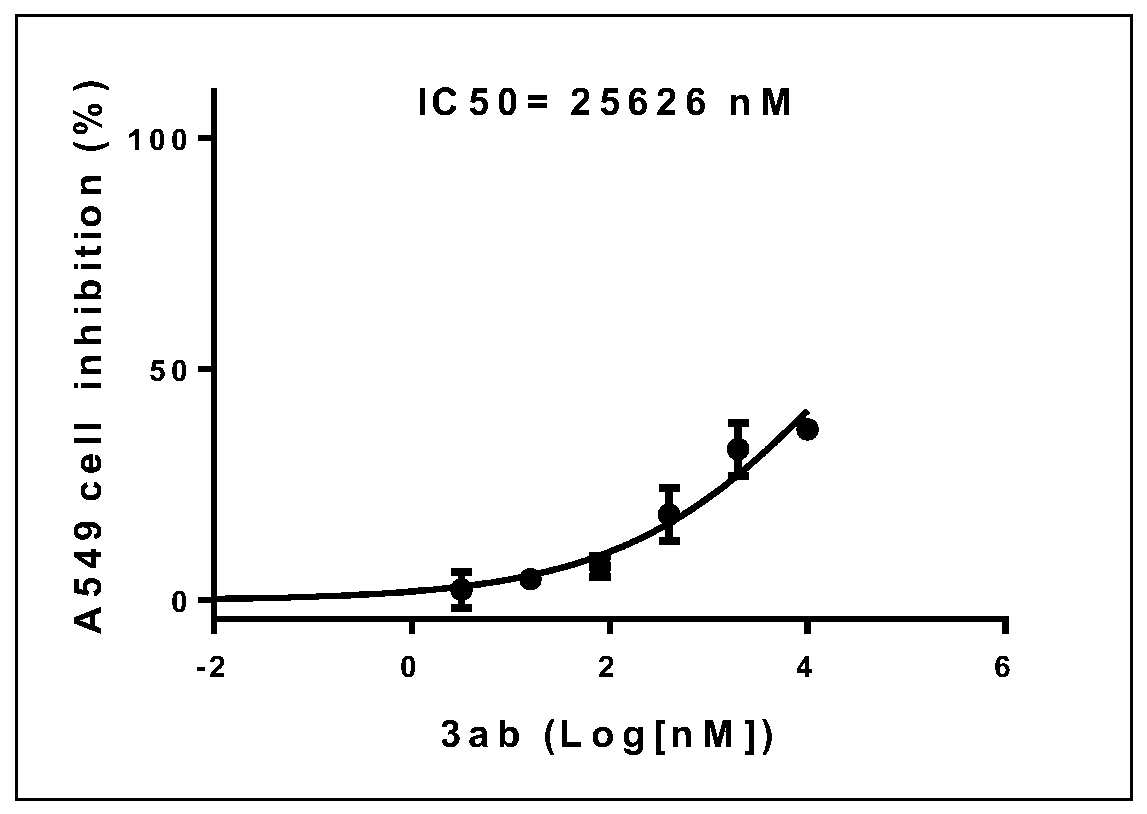
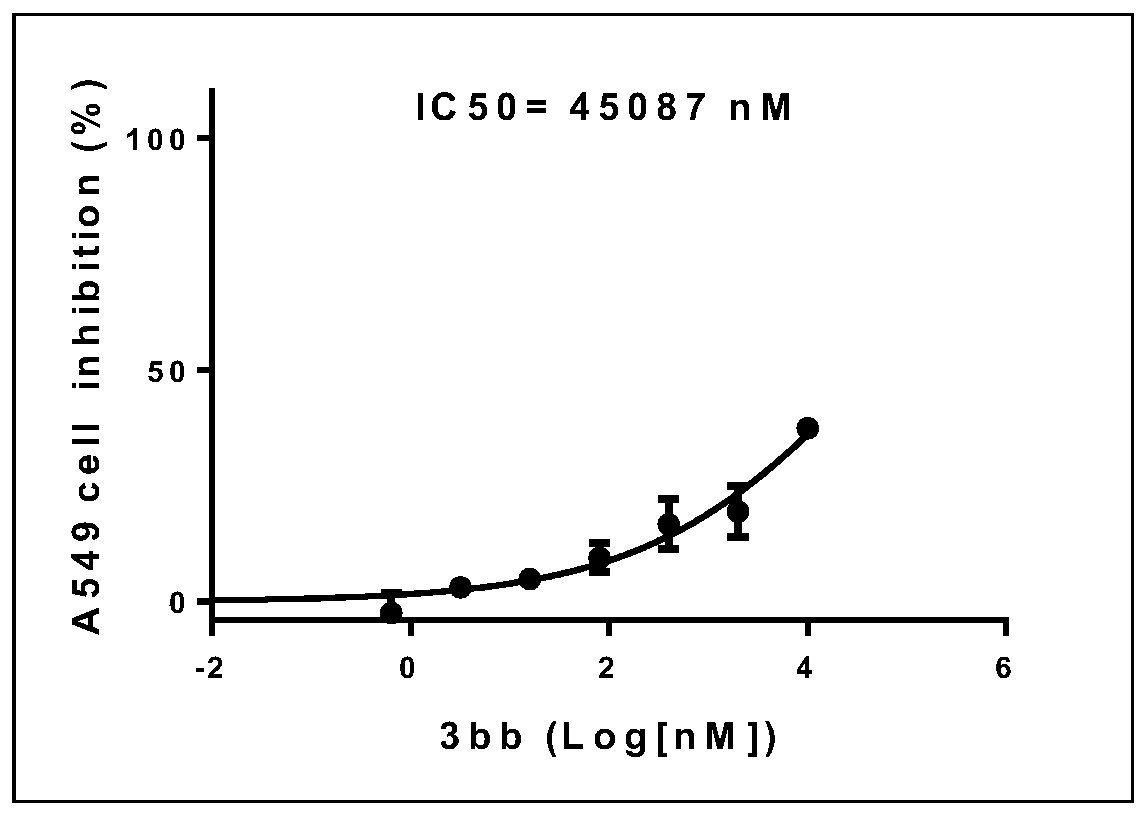
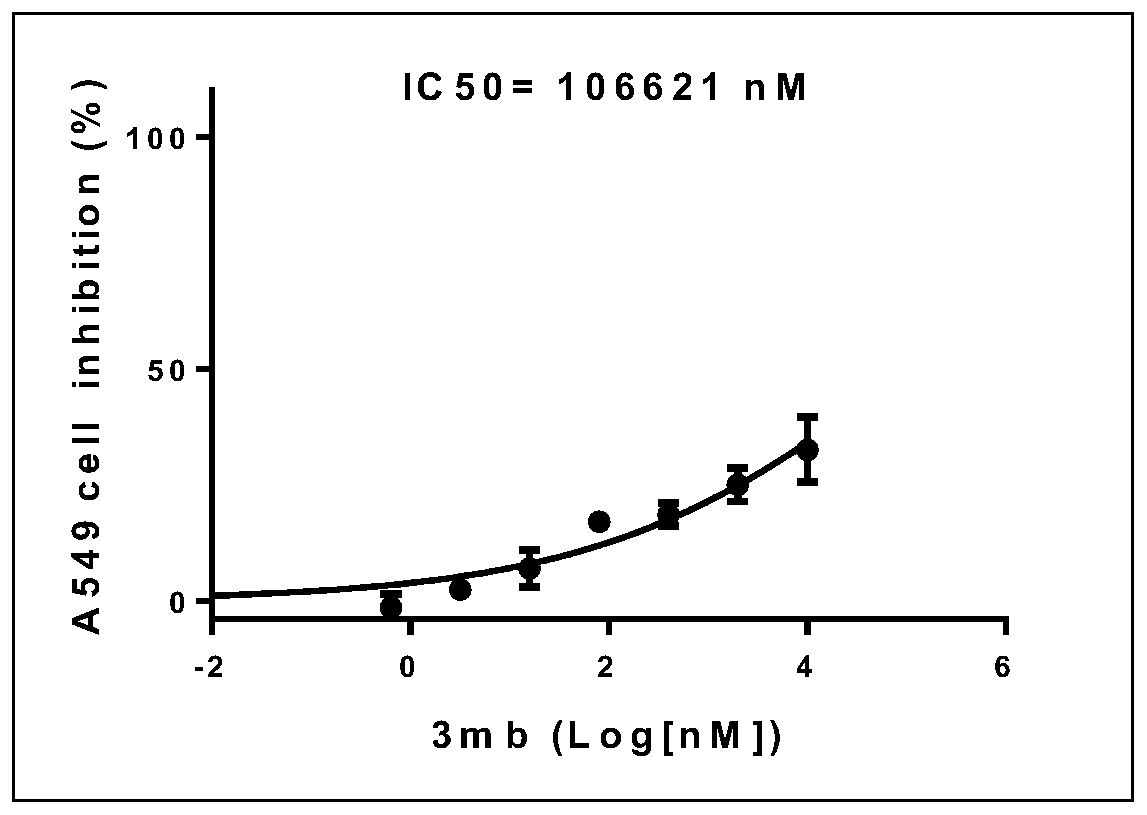
[0048] use replace The reaction temperature was 60°C, and other experimental procedures and purification methods were carried out with reference to Example 1; within 9 hours, 62 mg of yellow solid 3ab was obtained, the melting point was 43-45°C, and the yield: 91%.
[0049] 1 H NMR(600MHz, CDCl 3 ): 7.32(t,J=7.5Hz,2H),7.24-7.20(m, 3H),7.07(dd,J=11.9,1.6Hz,1H),6.54(d,J=11.9Hz,1H),6.37 (d,J = 0.9Hz, 1H), 4.51 (d, J = 15.0Hz, 1H), 4.46 (d, J = 15.1Hz, 1H), 3.68 (s, 2H), 3.12-3.10 (m, 1H) , 1.79(s, 3H), 1.39(s, 3H). 13 CNMR(150MHz, CDCl 3 ):197.6,172.2,151.5,144.6,139.0,137.8,136.6,129.4,129.0,128.7,126.7,118.6,80.1,70.7,46.2,40.2,39.6,24.8,15.8.HRMS(ESI-TOF)m / z: [M+Na] + Calcd for Chemical Formula:C 21 H 22 NaO 4 + 361.1410, Found:361.1414.
Example Embodiment
[0050] Example 3:
[0051]
[0052] use instead Other experimental procedures and purification methods were carried out with reference to the steps in Example 1; within 9 hours, 70 mg of white solids were obtained, the melting point was 79-81°C, and the yield was 87%.
[0053] 1 H NMR(600MHz, CDCl 3 ): 7.33 (t, J = 7.6 Hz, 2H), 7.25-7.19 (m, 3H), 7.09 (dd, J = 11.9, 1.8 Hz, 1H), 7.03 (s, 1H), 6.46 (d, J = 11.9Hz, 1H),4.58-4.41(m,2H),3.74-3.63(m,2H),3.15-3.13(m,J=6.0Hz,1H), 2.77(dd,J=16.3,7.1Hz,1H ), 2.63(dd,J=16.3,5.8Hz,1H),1.46(s, 3H). 13 C NMR(150MHz, CDCl 3 ): 188.9,172.1,150.0,149.3,138.7,137.0,130.3,129.2,128.8,126.8,125.0,119.3,82.2,71.1,46.1,40.1, 39.3,24.2.HRMS(ESI-TOF)m / z:(M +Na] + Calcd for C 20 H 19 NaO 4 + 425.0359, Found: 425.0354.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Melting point | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap