Combination therapy with glatiramer acetate and mitoxantrone for the treatment of multiple schlerosis
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Clinical Trial of Multiple Sclerosis
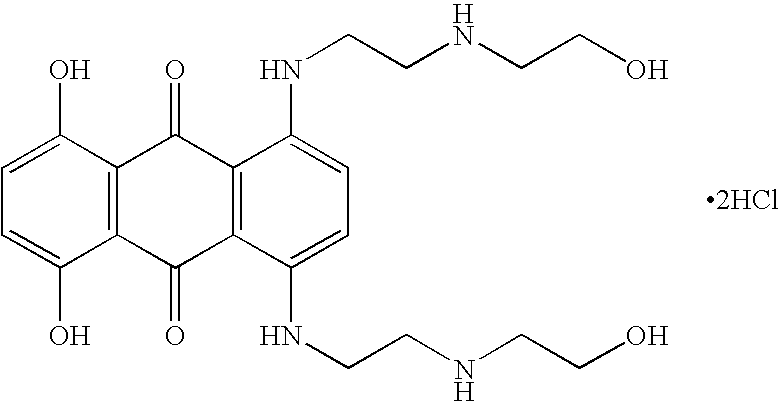
[0067] The primary objective of the trial is to determine whether short-term immunosuppression with mitoxantrone (Novantrone®) followed by chronic treatment with Glatiramer Acetate (Copaxone®) in comparison to treatment with Copaxone® for the same period of time without immunosuppression by mitoxantrone (Novantrone®) is well-tolerated and safe in patients with relapsing forms of MS as determined by clinical, laboratory and magnetic resonance imaging (MRI) parameters. The primary endpoints are tolerability and safety as measured by laboratory assessments and incidence of adverse experience. The secondary objective is to determine whether short-term immunosuppression with Novantrone® as an induction therapy accelerates the onset and enhances the efficacy of Copaxone® treatment in patients with relapsing forms of MS in 15 months of treatment based on MRI and clinical assessment.
[0068] The design of this trial is a multi-centered, randomized, two-ar...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Time | aaaaa | aaaaa |
| Mass | aaaaa | aaaaa |
| Mass | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com