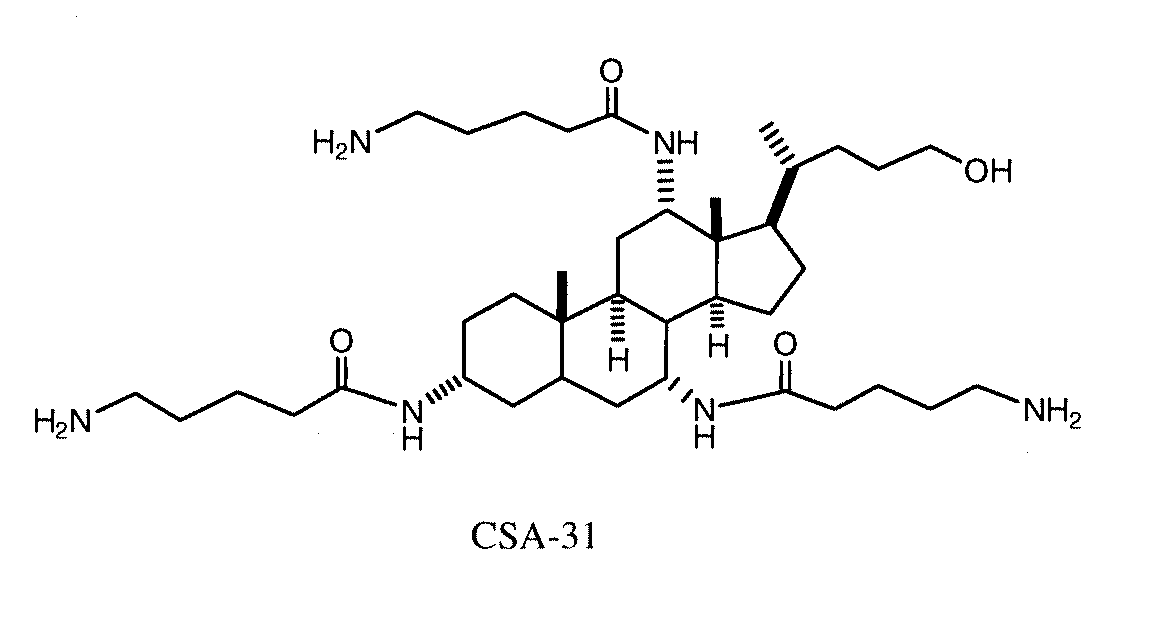
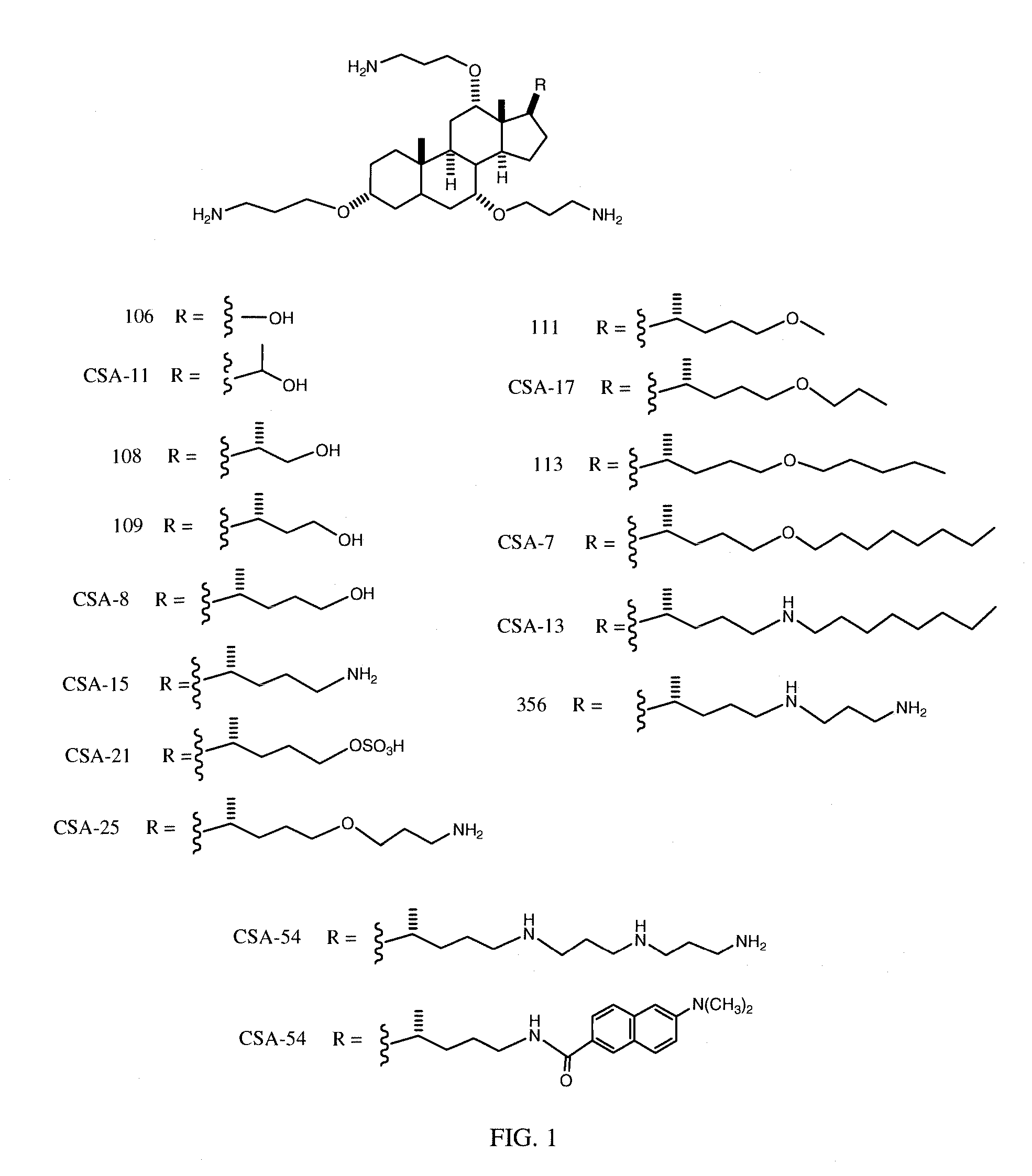
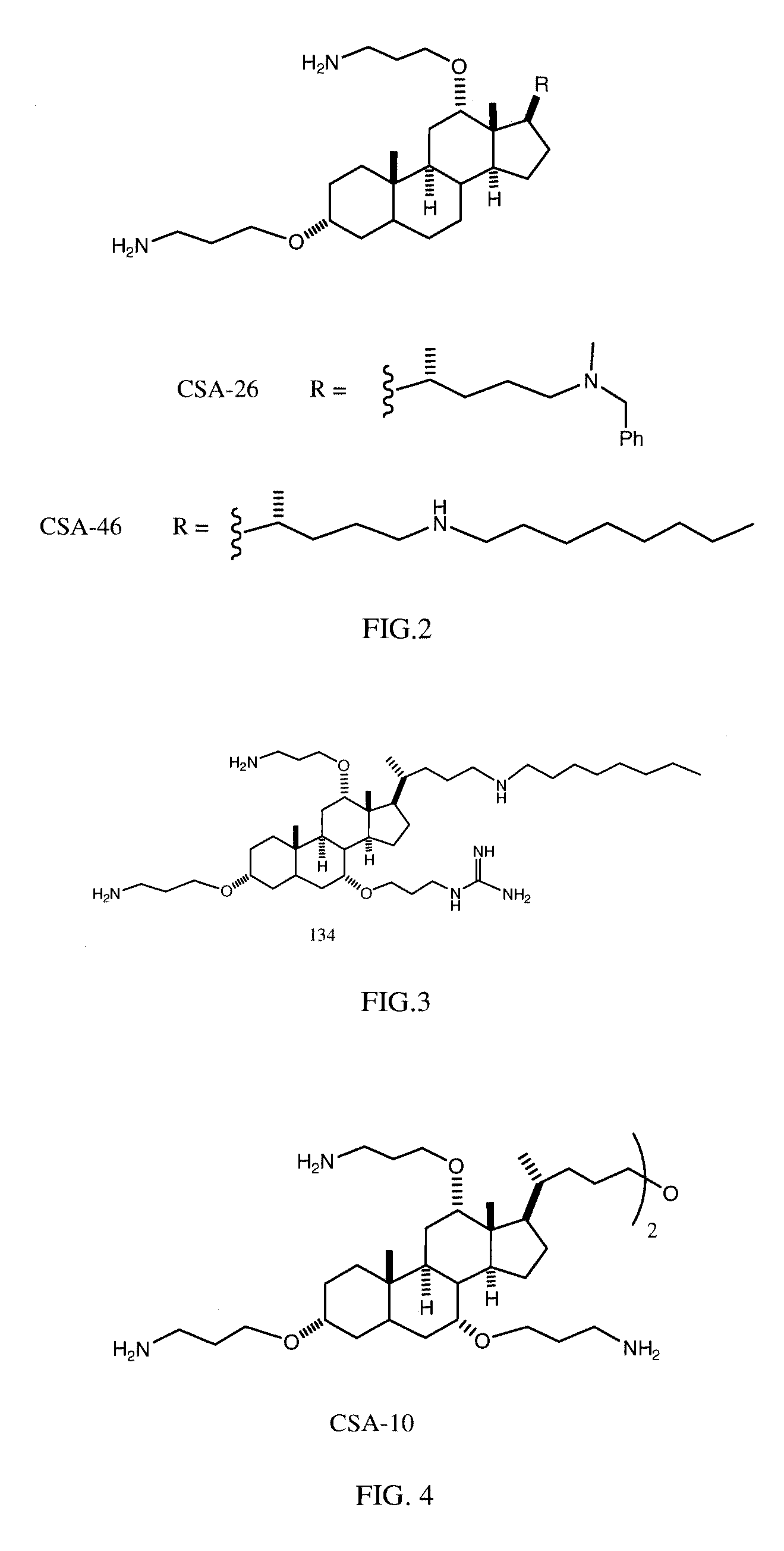
Cationic Steroid Antimicrobial Compositions and Methods of Use
a technology of cationic steroid and composition, applied in the field of cationic steroid antimicrobial composition and composition of use, can solve the problem that the immunotherapeutic effect of herpes vaccine seems less convincing
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 1
[0120] This example includes a description of one or more exemplary synthestic procedures for obtaining Compounds 1-5, 13-20 and 22-27.
[0121] Compound 13: To a 1 L round-bottom flask were added methyl cholate (30.67 g, 72.7 mmol) in dry THF (600 mL) and LiAlH4 (4.13 g, 109 mmol). After reflux for 48 hours, saturated aqueous Na2SO4 (100 mL) was introduced slowly, and the resulted precipitate was filtered out and washed with hot THF and MeOH. Recrystallization from MeOH gave colorless crystals of 13 (28.0 g, 98% yield). m.p. 236.5-238° C.; IR (KBr) 3375, 2934, 1373, 1081 cm−1; 1H NMR (CDCl3 / MeOH-d4, 200 MHz) δ 3.98 (bs, 1H), 3.83 (bs, 1H), 3.60-3.46 (m, 2H), 3.38 (bs, 5H), 2.30-2.10 (m, 2H), 2.05-1.05 (series of multiplets, 22H), 1.03 (bs, 3H), 0.92 (s, 3H), 0.71 (s, 3H); 13C NMR (CDCl3 / MeOH-d4, 50 MHz) δ 73.89, 72.44, 68.99, 63.51, 48.05, 47.12, 42.49, 40.37, 39.99, 36.62, 36.12, 35.58, 35.40, 32.77, 30.69, 30.04, 29.02, 28.43, 27.27, 23.96, 23.08, 18.00, 13.02; HRFAB-MS (thioglycer...
example 2
[0146] This example includes a description of one or more exemplary synthestic procedures for obtaining Compounds 3, 28 and 29.
[0147] Compound 28: A suspension of 19 (0.641 g, 0.614 mmol) and KCN (0.40 g, 6.14 mmol) in anhydrous DMSO (5 mL) was stirred under N2 at 80° C. overnight followed by the addition of H2 O (50 mL). The aqueous mixture was extracted with EtOAc (4×20 mL). The combined extracts were washed with brine once, dried over anhydrous Na2 SO4 and concentrated in vacuo. The residue was dissolved in CH2 Cl2 (3 mL) and MeOH (3 mL) and catalytic amount of p-toluenesulfonic acid (5.84 mg, 0.03 mmol) was added. The solution was stirred at room temperature for 3 hours before the introduction of saturated NaHCO3 solution (10 mL). After the addition of brine (60 mL), the mixture was extracted with EtOAc (4×30 mL). The combined extracts were washed with brine once and dried over anhydrous Na2 SO4 and concentrated. The residue afforded the desired product (0.342 g, 92% yield) as ...
example 3
[0150] This example includes a description of one or more exemplary synthestic procedures for obtaining Compounds 6, 7 and 30-33.
[0151] Compound 30: Cholic acid (3.0 g, 7.3 mmol) was dissolved in CH2Cl2 (50 mL) and methanol (5 mL). Dicyclohexylcarbodiimide (DCC) (1.8 g, 8.8 mmol) was added followed by N-hydroxysuccinimide (about 100 mg) and benzylmethylamine (1.1 g, 8.8 mmol). The mixture was stirred for 2 hours, then filtered. The filtrate was concentrated and chromatographed (SiO2, 3% MeOH in CH2Cl2) to give 3.0 g of a white solid (81% yield). m.p. 184-186° C.; IR (neat) 3325, 2984, 1678 cm−1; 1H NMR (CDCl3, 200 MHz) δ 7.21 (m, 5H), 4.51 (m, 2H), 3.87 (m, 1H), 3.74 (m, 2H), 3.36 (m, 2H), 2.84 (s, 3H), 2.48-0.92 (series of multiplets, 28H), 0.80 (s, 3H), 0.58 (d, J=6.5 Hz, 3H); 13C NMR (CDCl3, 50 MHz) δ 174.30, 173.94, 137.36, 136.63, 128.81, 128.46, 127.85, 127.50, 127.18, 126.28, 72.96, 71.76, 68.35, 53.39, 50.65, 48.77, 46.91, 46.33, 41.44, 39.36, 39.18, 35.76, 35.27, 34.76, 33...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Time | aaaaa | aaaaa |
| Frequency | aaaaa | aaaaa |
| Hydrophobicity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More