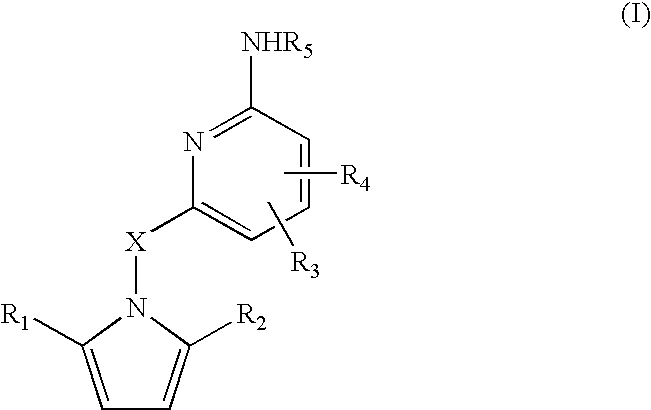
Amino-pyridines as inhibitors of β-secretase
a technology of -secretase and aminopyridine, which is applied in the field of0002amyloid deposits and neurofibrillary tangles, can solve the problems of eventual death and severe impairmen
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Preparation of N-{4-[1-(6-Amino-pyridin-2-ylmethyl)-5-(2-chloro-phenyl)-1H-pyrrol-2-yl]-phenyl}-3-methyl-benzamide
[0162]
Step 1: Preparation of Intermediate 3
[0163]Method A: Zinc chloride (24.5 g, 180 mmol) was dried under vacuum at 200° C. for 15 min. After cooling to room temperature, benzene (90 mL), triethylamine (13.7 g, 135 mmol), then t-butanol (10.0 g, 135 mmol) were added and the mixture stirred at room temperature. After 3 h, compound 1 (21.9 g, 90.0 mmol) and compound 2 (20.9 g, 135 mmol) were added and stirring continued for 5 d. The reaction was then quenched with 5% aqueous H2SO4 (200 mL) and diluted with ethyl acetate (1500 mL) and water (150 mL). The organic layer was separated and washed with brine (500 mL), dried over magnesium sulfate, filtered and concentrated to afford 34.6 g of a light yellow solid. Recrystallization from ethyl acetate afforded intermediate 3 (16.5 g, 58%) as a yellow-brown solid: 1H NMR (300 MHz, CDCl3) δ 8.34 (d, J=9.0 Hz, 2H), 8.18 (d, J=9.0 ...
example 2
Preparation of N-{4-[1-(6-Amino-pyridin-2-ylmethyl)-5-(2-chloro-phenyl)-1H-pyrrol-2-yl]-phenyl}-3-bromo-benzamide
[0168]
[0169]The reaction of 6a with 7 was performed essentially as described in Example 1, step 4. Purification by flash chromatography (silica, 2:3 ethyl acetate / hexanes) afforded the title compound (53.0 mg, 17%) as an off-white solid: Rf 0.44 (1:4 ethyl acetate / hexanes); mp 116-122° C.; 1H NMR (300 MHz, CDCl3) δ 8.00 (s, 1H), 7.80-7.50 (m, 5H), 7.45-7.30 (m, 4H), 7.30-7.20 (m, 4H), 6.41 (d, J=3.5 Hz, 1H), 6.33 (d, J=3.5 Hz, 1H). 6.21 (d, J=8.1 Hz, 1H), 5.76 (d, J=7.1 Hz, 1H), 5.01 (s, 2H), 4.36 (s, 2H); IR (ATR) 1522, 1461, 1316 cm−1; ESI MS m / z 559 [C29H22BrClN4O+H]+; HPLC (Method 1)>99% (AUC), tR=14.31 min. Anal. Calcd for C29H22BrClN4O: C, 62.44; H, 3.97; N, 10.04. Found: C, 62.05; H, 4.04; N, 9.51.
example 3
Preparation of N-{4-[1-(6-Amino-pyridin-2-ylmethyl)-5-(2-chloro-phenyl)-1H-pyrrol-2-yl]-phenyl}-benzamide
[0170]
[0171]The reaction of 6b with 7 was performed essentially as described in Example 1, step 4. Purification by flash chromatography (silica, 1:1 ethyl acetate / hexanes) afforded the title compound (121 mg, 31%) as a pale yellow solid: Rf 0.31 (1:1 ethyl acetate / hexanes); mp 196-198° C.; 1H NMR (300 MHz, CDCl3) δ 7.86 (d, J=6.6 Hz, 2H), 7.80 (s, 1H), 7.62-7.21 (m, 12H), 6.41 (d, J=3.5 Hz, 1H), 6.33 (d, J=3.6 Hz, 1H), 6.21 (d, J=8.2 Hz, 1H), 5.76 (d, J=7.2 Hz, 1H), 5.01 (s, 2H), 4.21 (s, 2H); IR (ATR) 1576, 1517, 1462 cm−1; ESI MS m / z 479 [C29H23ClN4O+H]+; HPLC (Method 1) 98.8% (AUC), tR=13.39 min.
PUM
| Property | Measurement | Unit |
|---|---|---|
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More