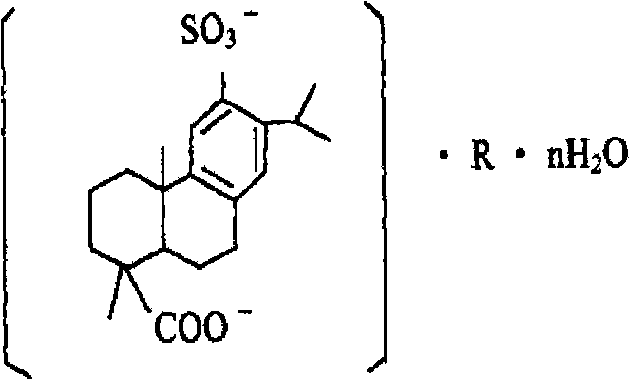
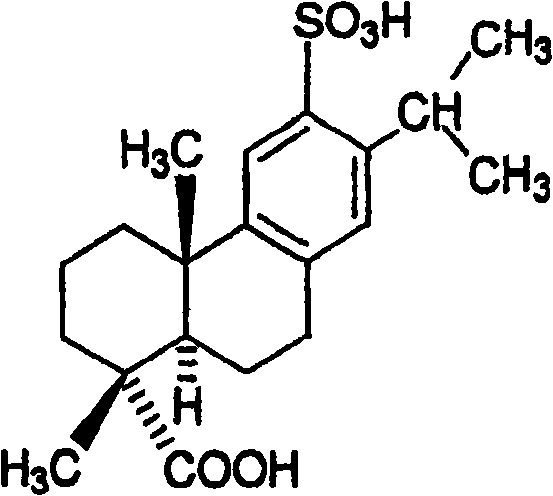
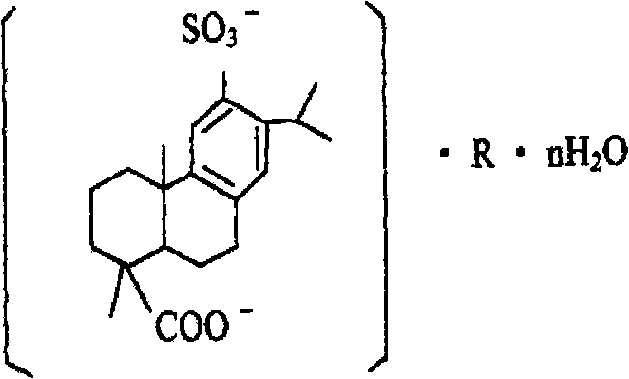
Disodium sulfodehydroabietate (DSDA) and composition and application thereof
A technology of disodium abietic acid and dehydroabietic acid, which is applied in drug combination, digestive system, drug delivery, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0203] Preparation of disodium sulfonated dehydroabietic acid:
[0204] (1) Preparation of abietic acid
[0205] Take 5 kg (about 15.5 mol) of golden yellow 310 resin, that is, rosin calcium grease, crush it, place it in a three-necked flask equipped with a stirrer and a reflux condenser, add 3 L of industrial ethyl acetate (about 30.6 mol) and 600 ml of hydrochloric acid, Adjust the pH value to 3-4, heat and reflux under stirring, react for 1 hour, stop stirring and let stand for a while, so that the insoluble matter settles completely, pour out the supernatant while it is hot, and place the supernatant for crystallization (about 2 days). Filtrate, wash the filter cake with an appropriate amount of ethyl acetate, combine the lotion and filtrate, concentrate and recover about 1L of ethyl acetate, continue to place it for crystallization, crystallize and filter out, then wash with industrial ethyl acetate, dry the crystallization, and obtain crude abietic acid A total of 2225g...
Embodiment 2
[0215] Preparation of disodium sulfonated dehydroabietic acid granules, capsules and solutions:
[0216] (1) The weight proportion of combination and the binder selected in each prescription are listed in Table 8
[0217] The weight proportion of combination and the adhesive selected for use in each prescription of table 8 Unit: gram
[0218] Raw materials
Prescription 1
Prescription 2
Prescription 3
Prescription 4
Prescription 5
Prescription 6
Prescription 7
Sulfonated dehydroabietic acid II
Sodium (calculated as anhydrous)
500
500
500
500
500
500
500
300
400
500
400
300
400
500
70
40
70
70
40
70
90
10% pvp
water
30% B
70% B
95%
10% pvp B
alcohol solution
5%...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More