A kind of synthetic method of 2β-azidomethyl-2α-methylpenicillane-3α-carboxylate diphenylmethyl ester
A technology of diphenylmethyl carboxylate and methyl penicillane, applied in the field of medicine, can solve problems such as waste, and achieve the effects of controlling production cost, simple and easy reaction, and mild process conditions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
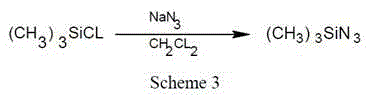
[0017] Embodiment 1: the synthesis of trimethylsilyl azide:
[0018] At room temperature, in a 1000ml four-necked bottle, add 400ml of dichloromethane, 100g of sodium azide, 1.2g of catalyst zinc iodide and 2g of phase transfer catalyst polyethylene glycol, slowly drop 162g of trimethyl chloride For silane, the dropping time is 4 hours, and after the dropping is completed, the reaction is kept at 40°C for 16 hours. After the reaction is completed, filter the solid, collect the mother liquor, rectify the mother liquor, collect the solvent dichloromethane at 40~42°C, and collect the product trimethylsilyl azide (TMSN) at 93~95°C 3 ), to obtain 140g product, yield 80%.
Embodiment 2
[0019] Example 2: 3-Methyl-[2-oxo-4-(2-benzothiazoledithio)-1-azetidinyl]-3-butenediphenylmethyl ester (Compound 5) preparation:
[0020] Mix 240g of debrominated sulfoxide ester (compound 4), 105g of 2-mercaptobenzothiazole, and 1500m1 of toluene, reflux for 3 hours, do TLC to detect no raw material spots, distill to oil, and obtain oily foam Compound 5, yield 84.6 %, the content is 78% (HPLC normalization method), and the next step reaction can be directly carried out without purification.
Embodiment 3
[0021] Example 3: Preparation of 2β-chloromethyl-2α-methyl-6,6-dihydropenicillanic acid benzhydryl ester (compound 6):
[0022] Take the oil (compound 5) after thermal cracking in the previous step and add 2000ml of dichloromethane to dissolve it, cool down, add 100ml of purified water at t≤-5°C, slowly add 600ml of 36% concentrated hydrochloric acid at 0~-5°C. t≤-5℃, add dropwise sodium nitrite solution (43gNaNO 2 +300mlH 2 O), after dropping, keep it under 0~3℃ for 1.5 hours. After the reaction, the solid was removed by filtration, the filter cake was washed with an appropriate amount of dichloromethane, the organic layer was washed with sodium bicarbonate and saturated sodium chloride, evaporated under reduced pressure to obtain an oil, and the next reaction was carried out at a conversion rate of 100%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More