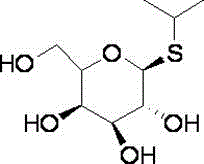
Preparation method of isopropyl-β-d-thiogalactoside
A technology of thiogalactoside and propylthioacetyl, which is applied in the field of preparation of isopropyl-β-D-thiogalactoside, can solve the problems of high cost, complicated operation and low yield, and achieve The operation is simple, the raw materials are easy to obtain, and the effect of improving the safety factor
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0022] a) Add 5.5mol acetic anhydride and 1mol aluminum trichloride at room temperature, add 1mol galactose in 9 batches at 5-10°C, add 1.1mol isopropyl mercaptan after the reaction is completed, and obtain 0.765mol isopropyl mercaptan after the reaction is completed Propylthioacetylgalactose;
[0023] b) Add 0.765 mol of isopropyl thioacetylgalactose to 10 mol of methanol to dissolve, add 0.01 mol of sodium methoxide, add 0.01 mol of acetic acid to neutralize after the reaction is completed, and obtain 0.743 mol of isopropyl-β-D- Thiogalactosides. The yield was 74.3%.
[0024] The post-treatment in step a is to drop 10mol of water at 0-5°C and stir for 2 hours, extract the aqueous layer with 5mol of dichloromethane, separate the organic phase and wash the organic phase with 10mol of water three times, separate the organic phase and concentrate, add 1mol tert-butyl methyl ether and 2mol isohexane mixed solution crystallized, filtered and dried.
[0025] The post-treatment i...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More