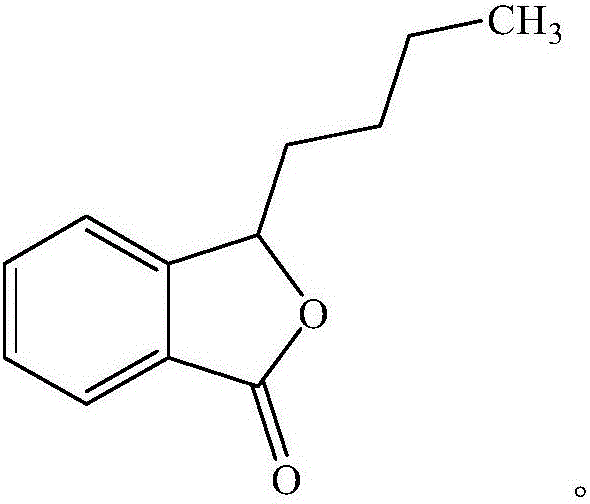
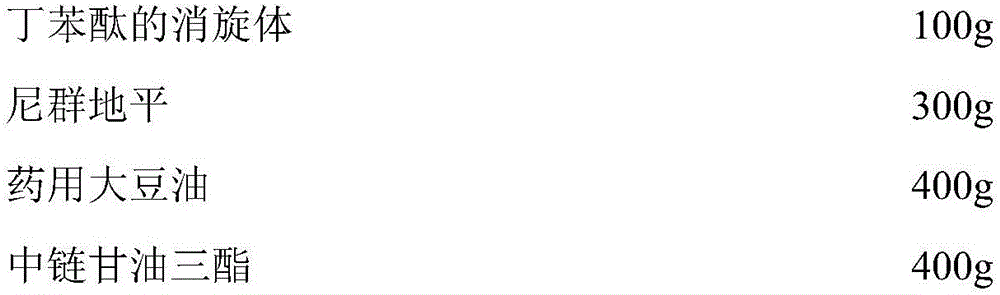Butylphthalide and nitrendipine soft capsule for relieving acute ischemic stroke
A technology for ischemic stroke and butylphthalide nitrendipine, which is applied in the field of butylphthalide nitrendipine soft capsules, can solve the problems of limited treatment methods, high price, and heavy drug burden for patients, and achieve cost reduction and low price , Alleviate the effect of acute ischemic stroke
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0023] Capsule contents (5000 capsules):
[0024]
[0025] Capsule shell:
[0026]
[0027]
[0028] Preparation:
[0029] (1) Weigh glycerin and water in the formula amount, add to the tank and heat up to 75°C, stir evenly, slowly add gelatin and tragacanth gum, stir for 2 hours, remove air bubbles in a vacuum, cool down to 60°C and keep warm to obtain a capsule shell solution, spare;
[0030] (2) Take by weighing the butylphthalide and medicinal soybean oil of formula quantity, stir evenly, obtain material A, set aside;
[0031] (3) Take by weighing the nitrendipine and medium-chain triglycerides of the formula amount, stir evenly, obtain material B, and set aside;
[0032] (4) Material A and material B are mixed and stirred evenly to obtain the mixed material, and then the mixed material is mixed with the capsule shell solution by a soft capsule machine to make a soft capsule; The box and the spray body are heated, and the water pump circulation and cooling are...
Embodiment 2
[0037] Capsule contents (5000 capsules):
[0038]
[0039] Capsule shell:
[0040]
[0041] Preparation:
[0042] Take by weighing the sorbitol and water of recipe amount in the step (1), all the other are with embodiment 1.
[0043] The prepared samples (including 10 parallel samples numbered 1 to 10) were tested for content and disintegration time limit, and the test results are shown in Table 2. As can be seen from Table 2, the content uniformity of embodiment 2 is good.
[0044] Table 2. Content and disintegration time limit detection of embodiment 2
[0045]
Embodiment 3
[0047] Capsule contents (5000 capsules):
[0048]
[0049] Capsule shell:
[0050]
[0051] Preparation:
[0052] In the step (1), take by weighing the mannitol and water of formula quantity, all the other are with embodiment 1.
[0053] The content and disintegration time limit of the prepared samples (including 10 parallel samples numbered 1 to 10) were tested, and the test results are shown in Table 3. As can be seen from Table 3, the content uniformity of embodiment 3 is good.
[0054] Table 3. Content and disintegration time limit detection of embodiment 3
[0055]
[0056]
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap