Pharmaceutical composition for inhibiting infection of Aeromonas hydrophila
A technology of Aeromonas hydrophila and its composition, which is applied in the direction of antibacterial drugs, active ingredients of hydroxyl compounds, and pharmaceutical formulations, can solve the problems of aggravated bacterial drug resistance, achieve no drug residues, use low doses, and treat good effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0014] Embodiment 1: The minimum inhibitory concentration determination of dihydromyricetin and honokiol alone or in combination to Aeromonas hydrophila
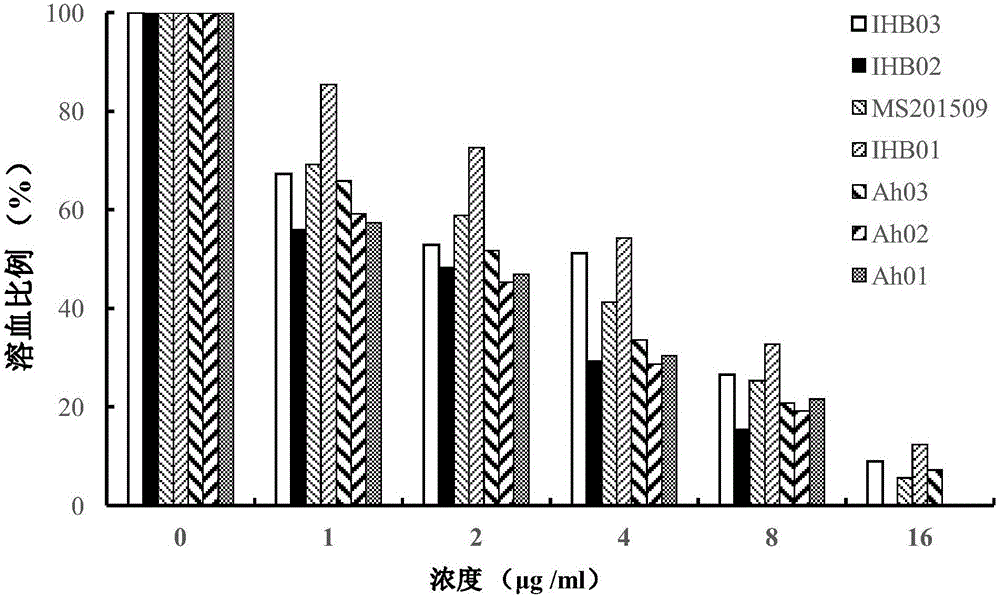
[0015] The minimum inhibitory concentrations (MICs) of dihydromyricetin and honokiol alone or in combination were determined by the microdilution method recommended by CLSI. The 7 strains of Aeromonas hydrophila from different sources involved in the present invention are all preserved by the Yangtze River Fisheries Research Institute, and their names are shown in Table 1. The steps are:
[0016] (1) Dilute the prepared drug in a 96-well cell culture plate, and the concentrations of dihydromyricetin are 128 μg / mL, 64 μg / mL, 32 μg / mL, 16 μg / mL, 8 μg / mL, 4 μg / mL, 2μg / mL, 1μg / mL, 0.5μg / mL, 0.25μg / mL; honokiol concentrations were 512μg / mL, 256μg / mL, 128μg / mL, 64μg / mL, 32μg / mL, 16μg / mL, 8μg / mL, 4μg / mL, 2μg / mL, 1μg / mL, drug volume per well 100μL;
[0017] (2) Aseptically inoculate single colonies of 7 strains of Aeromonas hydr...
Embodiment 2
[0022] Embodiment 2: Checkerboard microdilution test to determine the effect of dihydromyricetin and honokiol combined use
[0023] The inhibitory effect of different concentrations of dihydromyricetin combined with honokiol on Aeromonas hydrophila Ah01 was determined according to the standard method published by CLSI. The steps are:
[0024] (1) Dilute the two different drug combinations used in the test in sterile centrifuge tubes respectively, so that the concentrations of dihydromyricetin are 128 μg / mL, 64 μg / mL, 32 μg / mL, 16 μg / mL, 8 μg / mL, respectively. mL, 4μg / mL, 2μg / mL, 1μg / mL, 0.5μg / mL, 0.25μg / mL; honokiol concentrations were 512μg / mL, 256μg / mL, 128μg / mL, 64μg / mL, 32μg / mL , 16 μg / mL, 8 μg / mL, 4 μg / mL, then respectively add 50 μL of dihydromyricetin and honokiol to the 96-well cell culture plate, so that the concentration of dihydromyricetin is from left to right (1-10 ) decreased sequentially, and the concentration of honokiol decreased sequentially from top to bot...
Embodiment 3
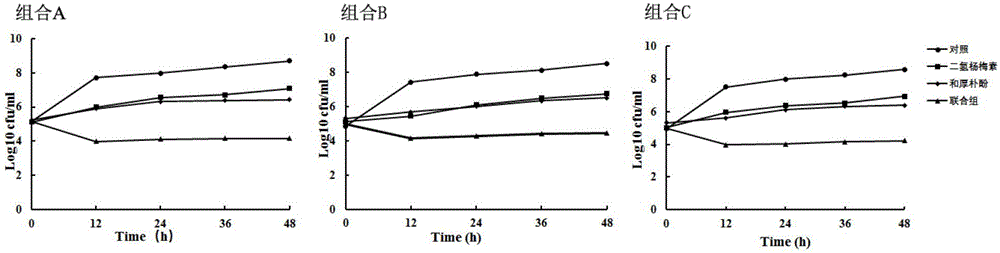
[0027] Example 3: Determining time-killing curves to further verify the synergistic effect of different combinations of dihydromyricetin and honokiol in combination
[0028] In order to verify the synergistic effect obtained in Example 2, according to the test method, the combination of dihydromyricetin and honokiol with a concentration ratio of 1:2, 1:8, and 1:16 after the combined drug with synergistic effect is required. , the MS201509 strain was used as a representative strain for time-killing curve determination. Bacteria were cultured until OD600 was about 0.3, divided into 4 bottles, each bottle was 10mL, and the following combinations of drugs were added: 1 / 2MIC honokiol, 1 / 2MIC dihydromyricetin, 1 / 2MIC honokiol+1 / 2MIC dihydromyricetin and negative control, see Table 2 for details. Shake culture at 30°C, take the bacterial solution at 0, 12, 24, 36 and 48 hours, dilute it, smear it on the plate, and count it with a counter. Judgment criteria: when the logarithm of th...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap