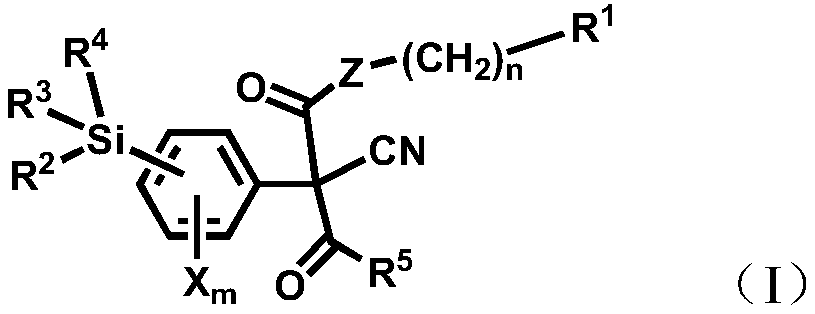
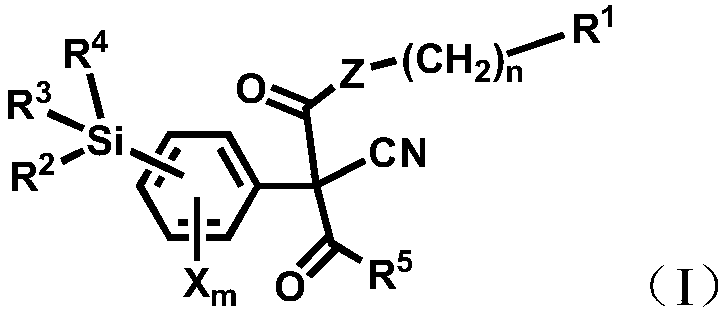
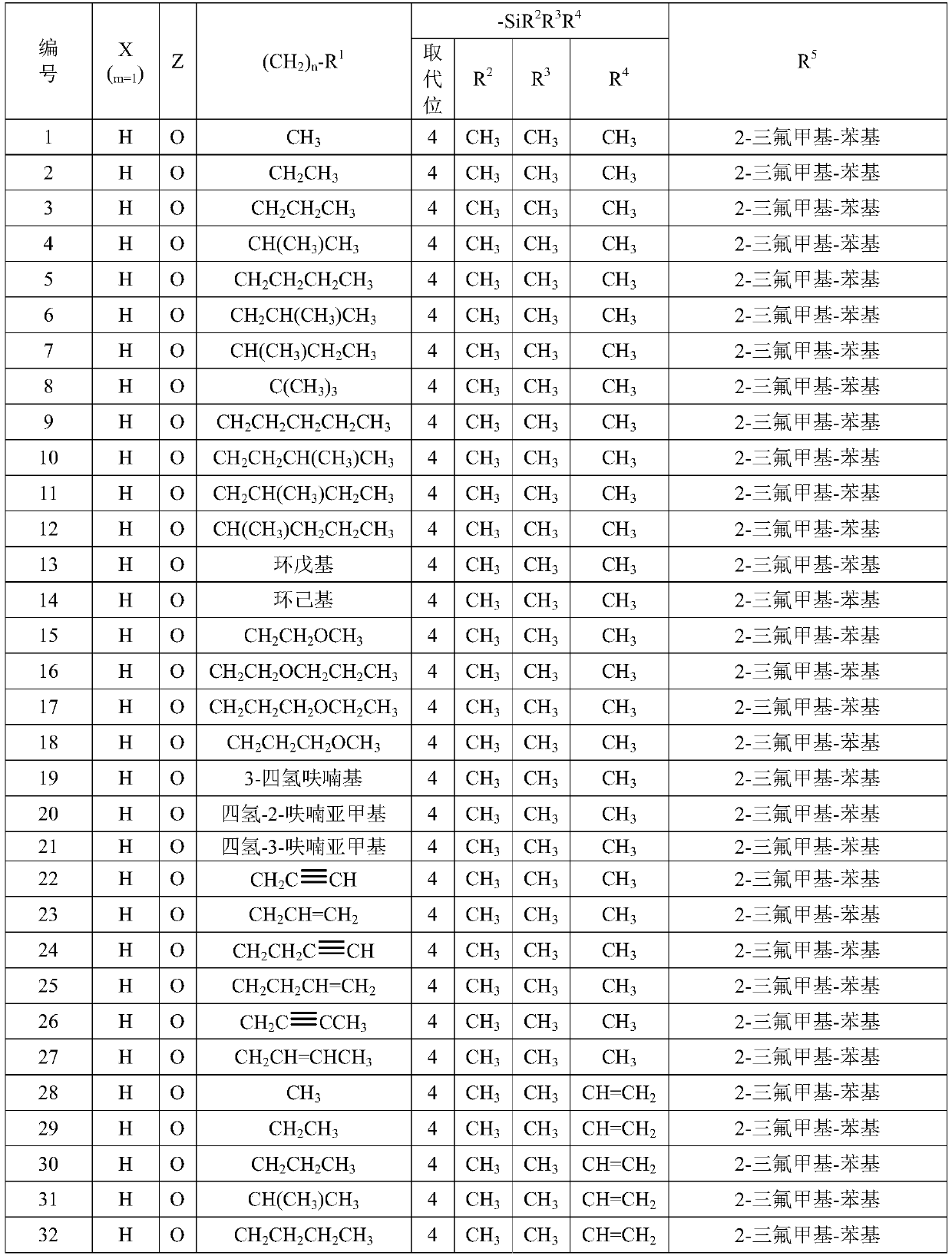
Silicon acyl acetonitrile compounds and preparation method and application thereof
A technology of silicyl acetonitrile and compound, which is applied in the field of insecticides and/or acaricides, and silicyl acetonitrile-containing compounds, and can solve the problems that the research of new acaricides cannot meet the needs and other problems.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0149] The preparation method of the compound of the present invention
[0150] The preparation method of the compound of formula I of the present invention is described in more detail below, but these specific methods do not constitute any limitation to the present invention. The compounds of the present invention can also be conveniently prepared by optionally combining various synthetic methods described in the specification or known in the art, and such combinations can be easily performed by those skilled in the art to which the present invention belongs.
[0151] The present invention also provides a general method for preparing the compound described in the first aspect, namely the compound of formula I.
[0152] In a specific embodiment, the compound of formula I is prepared by the following reaction:
[0153] Reaction 1:
[0154]
[0155] Reaction 2:
[0156]
[0157] Reaction 3:
[0158]
[0159] Reaction 4:
[0160]
[0161] Reaction 5:
[0162] ...
Embodiment 1
[0192] The preparation of embodiment 1 compound 4
[0193] (1). Preparation of p-trimethylsilylbromobenzene:
[0194]
[0195]Weigh 2.9g of p-dibromobenzene into the Schlenk reaction flask, add 100ml of anhydrous THF to dissolve, stir at -78°C, slowly add 5ml of n-BuLi (2.5M) dropwise, and continue at -78°C Stirring was followed by the dropwise addition of 1.8 ml trimethylchlorosilane. Gradually warm to room temperature, add saturated NH 4 Cl solution, extracted with ethyl acetate (30 mL×3), combined organic phases, washed with saturated sodium chloride solution, and dried over anhydrous sodium sulfate. The solvent was removed under reduced pressure and purified by column chromatography to obtain the target compound as a colorless oil with a yield of 74.7%. 1 H NMR (400MHz, Chloroform-d) δ 7.48 (d, J = 8.2Hz, 2H), 7.37 (d, J = 8.3Hz, 2H), 0.25 (s, 9H) ppm.
[0196] (2). Preparation of p-trimethylsilylphenylacetonitrile:
[0197]
[0198] Weigh 37 mg of allylpalladiu...
Embodiment 2
[0208] The preparation of embodiment 2 compound 6
[0209] (1). Preparation of 2-(4-trimethylsilylbenzene)-2-methoxyisobutyl cyanoacetate:
[0210]
[0211] Weigh 600mg of ethyl 2-(4-trimethylsilylphenyl)-cyanoacetate and dissolve it in 15ml of isobutanol, add two drops of concentrated sulfuric acid as a catalyst dropwise, and react at 100°C for 12 hours. The reaction solution was poured into 30ml of water, extracted with ethyl acetate (20mL×3), the combined organic phases were washed with saturated sodium chloride solution, and dried over anhydrous sodium sulfate. The solvent was removed under reduced pressure and purified by column chromatography to obtain the target compound as a light yellow oil with a yield of 82.2%; 1 H NMR (400MHz, Chloroform-d) δ7.44 (d, J = 8.1Hz, 2H), 7.31 (d, J = 8.0Hz, 2H), 4.02 (ddd, J = 29.5, 10.5, 6.5Hz, 2H) , 2.09–1.94 (m, 1H), 0.95 (d, J=6.7Hz, 6H), 0.30 (s, 9H) ppm.
[0212] (2).(RS)-2-(4-trimethylsilylphenyl)2-cyano-3-oxo-3-(α,α,α,-tri...
PUM
| Property | Measurement | Unit |
|---|---|---|
| diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More