Novel coronavirus detection primer probe combination and kit and application thereof
A detection kit and coronavirus technology, which is applied in the direction of recombinant DNA technology, microbial measurement/inspection, and resistance to vector-borne diseases, etc., can solve the problems of false negatives, high cost, difficulty in popularization, etc., and achieve high detection sensitivity Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0034] 1. Sample processing and RNA extraction
[0035] 1) Types of samples: upper respiratory tract specimens, such as throat swabs, nasal swabs, nasopharyngeal extracts; lower respiratory tract specimens, such as deep cough sputum, respiratory tract extracts, bronchial lavage fluid, alveolar lavage fluid, lung tissue biopsy specimen.
[0036] 2) RNA extraction: RNA was extracted by using the viral genome DNA / RNA extraction kit produced by Tiangen Biochemical Technology (Beijing) Co., Ltd.;
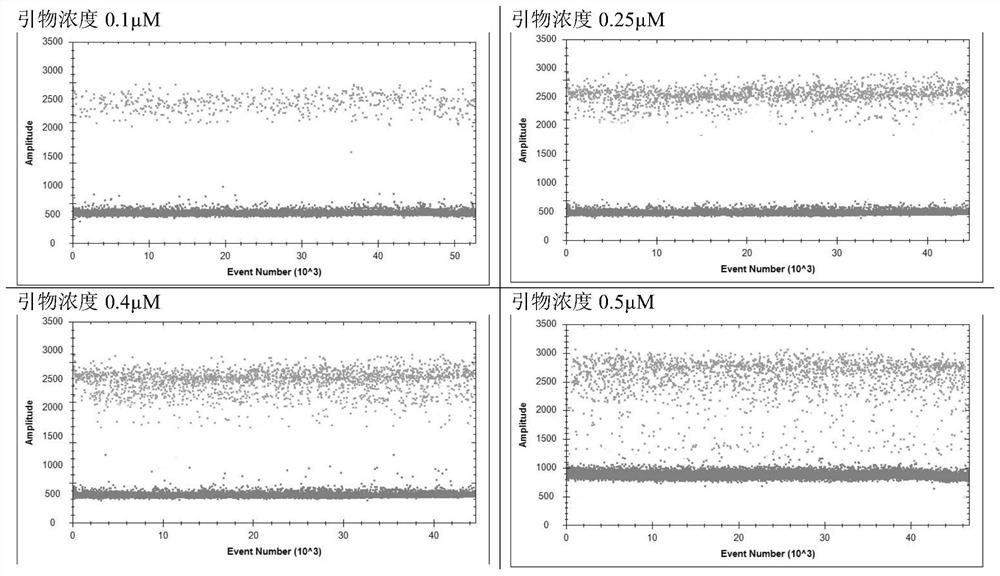
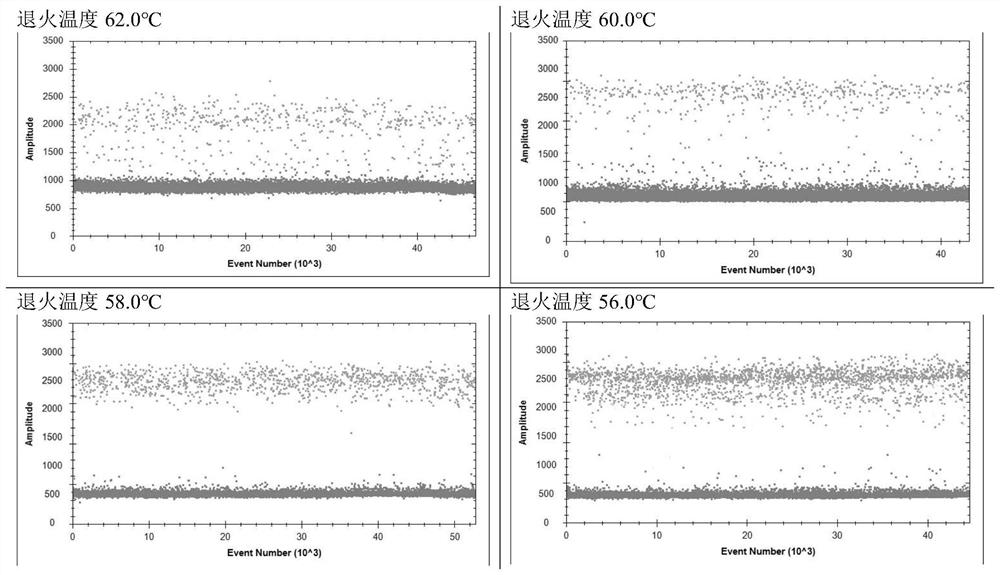
[0037] 2. Specific primers, probe screening and system optimization for the detection of novel coronavirus
[0038] 1) Screening of specific primers and probes: Design primers and probes according to the gene sequence of the novel coronavirus announced by the China National Center for Disease Control and Prevention, and send the designed primers and probes to Beijing Ruibo Xingke Biotechnology Co., Ltd. The company's synthesis, the specific sequence is shown in the table below:
[003...
Embodiment 2
[0056] 1. Performance verification of the kit
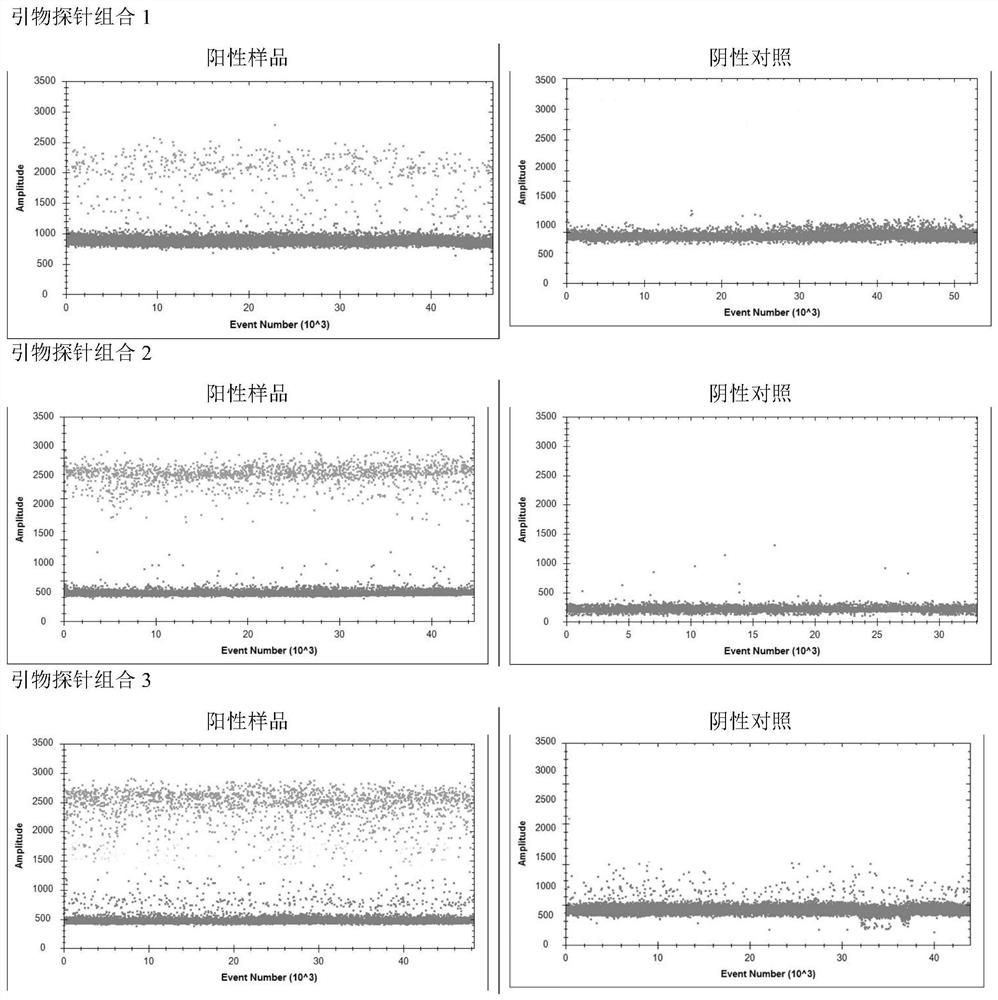
[0057] 1) Specificity test
[0058] The optimized digital PCR reaction system was used to test one case of throat swabs from patients with positive samples of new coronavirus and its interference cases (influenza A / B virus, mycoplasma pneumoniae, chlamydia pneumoniae, adenovirus, and respiratory syncytial virus). Test, and select throat swabs from normal people as a negative control to test the specificity of the established method.
[0059] serial number sample copy number 1 COVID-19 Positive Sample 5751 2 negative control - 3 Influenza A virus - 4 Influenza B virus - 5 Mycoplasma pneumoniae - 6 Chlamydia pneumoniae - 7 Adenovirus - 8 respiratory syncytial virus -
[0060] The results show that the novel coronavirus gene detection kit provided by the present invention can accurately detect the novel coronavirus samples containing the target gene, without ...
Embodiment 3
[0068] 1. Clinical sample testing:
[0069] Blind testing was carried out on 30 clinically collected samples, including 20 throat swab samples pathologically diagnosed as novel coronavirus and 10 normal control throat swab samples. RNA was extracted according to the experimental method, and the PCR amplification system and amplification conditions were established. Test and set up negative and positive controls. At the same time, the primer-probe combination qPCR detection method provided by the present invention was used as reference 1, and the primer-probe combination disclosed in the "Technical Guidelines for Laboratory Detection of Novel Coronavirus Infected Pneumonia" was used for qPCR detection as reference 2 to compare the detection effects of the kits.
[0070] The results are shown in the table below
[0071]
[0072] The results show that the detection kit for detection of novel coronavirus digital PCR provided by the present invention has a detection sensitivity...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Sensitivity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap