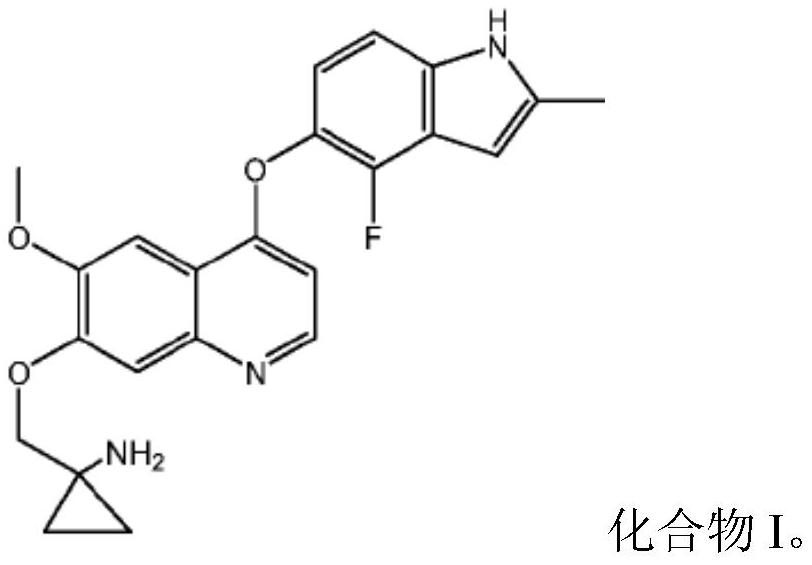
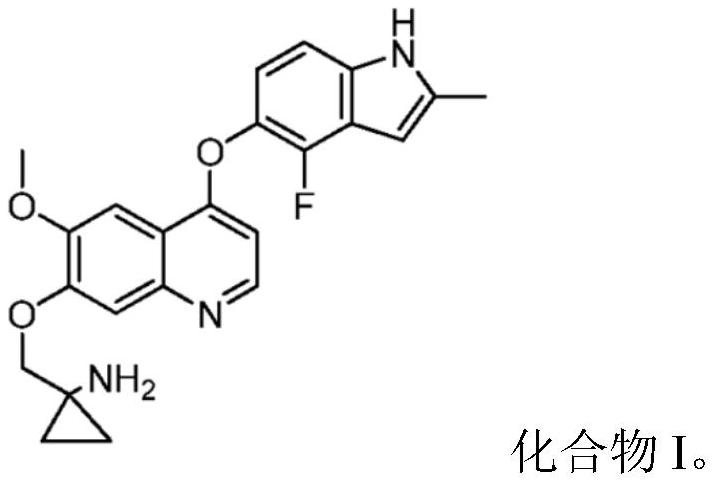
Quinoline compound for combined treatment on gastric carcinoma
A compound, gastric cancer technology, applied in the field of pharmaceutical preparations and medicine, can solve problems such as lack of therapeutic drugs
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0172] Patients with pathologically confirmed advanced gastric adenocarcinoma (including gastroesophageal junction adenocarcinoma, Siewert type II, type III) are given anlotinib, cisplatin and S-1 in combination, and the specific dosage regimen is as follows:
[0173] Anlotinib Hydrochloride Capsules: Before breakfast, take Anlotinib Hydrochloride Capsules on an empty stomach, once a day, 12mg each time (1 capsule). Continuous oral administration for 2 weeks, stop for 1 week, that is, 3 weeks (21 days) is a treatment cycle. If there is a missed dose, it is confirmed that the time from the next dose is less than 12 hours, and no supplementary dose will be given.
[0174] Cisplatin injection: 60~80mg / m 2 Intravenous infusion, administration on d1, 3 weeks (21 days) as a treatment cycle;
[0175] S-1 capsule: 40-60mg po bid, d1-14, 3 weeks (21 days) as a treatment cycle.
[0176] Observation efficacy indicators: objective response rate (ORR=CR+PR), progression-free survival (P...
Embodiment 2
[0179] For patients with unresectable advanced or metastatic gastric and gastroesophageal junction adenocarcinoma (including signet ring cell carcinoma, mucinous adenocarcinoma, hepatoid adenocarcinoma) confirmed by histopathological and / or cytological examination, the combination drug Rotinib and toripalimab, the specific dosage regimen is as follows:
[0180] Anlotinib hydrochloride capsules, orally, 1 time / day, 12mg / time, medication from the 1st day to the 14th day of each cycle, every 3 weeks is a treatment cycle; toripalimab, 3mg / kg, intravenous Infusion for 1h±5min, once every 2 weeks (administration on the 1st and 15th day of each cycle), every 4 weeks is a treatment cycle, until the treatment termination criteria are met. Efficacy evaluation was performed according to RECIST1.1 standard every 8 weeks.
[0181] Observe the curative effect index:
[0182] Objective response rate (ORR=PR+CR), progression-free survival (PFS), overall survival (OS), disease control rate (...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More