Opioid agonist/opioid antagonist/acetaminophen combinations
a technology of acetaminophen and naloxone, which is applied in the direction of heterocyclic compound active ingredients, drug compositions, biocides, etc., can solve the problems of profound antagonistic effect of naloxone given by injection, still subject to patient misuse and abuse, etc., and achieves the effect of reducing the tendency of aqueous dispersion and enhancing the elasticity of the film formed
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 2
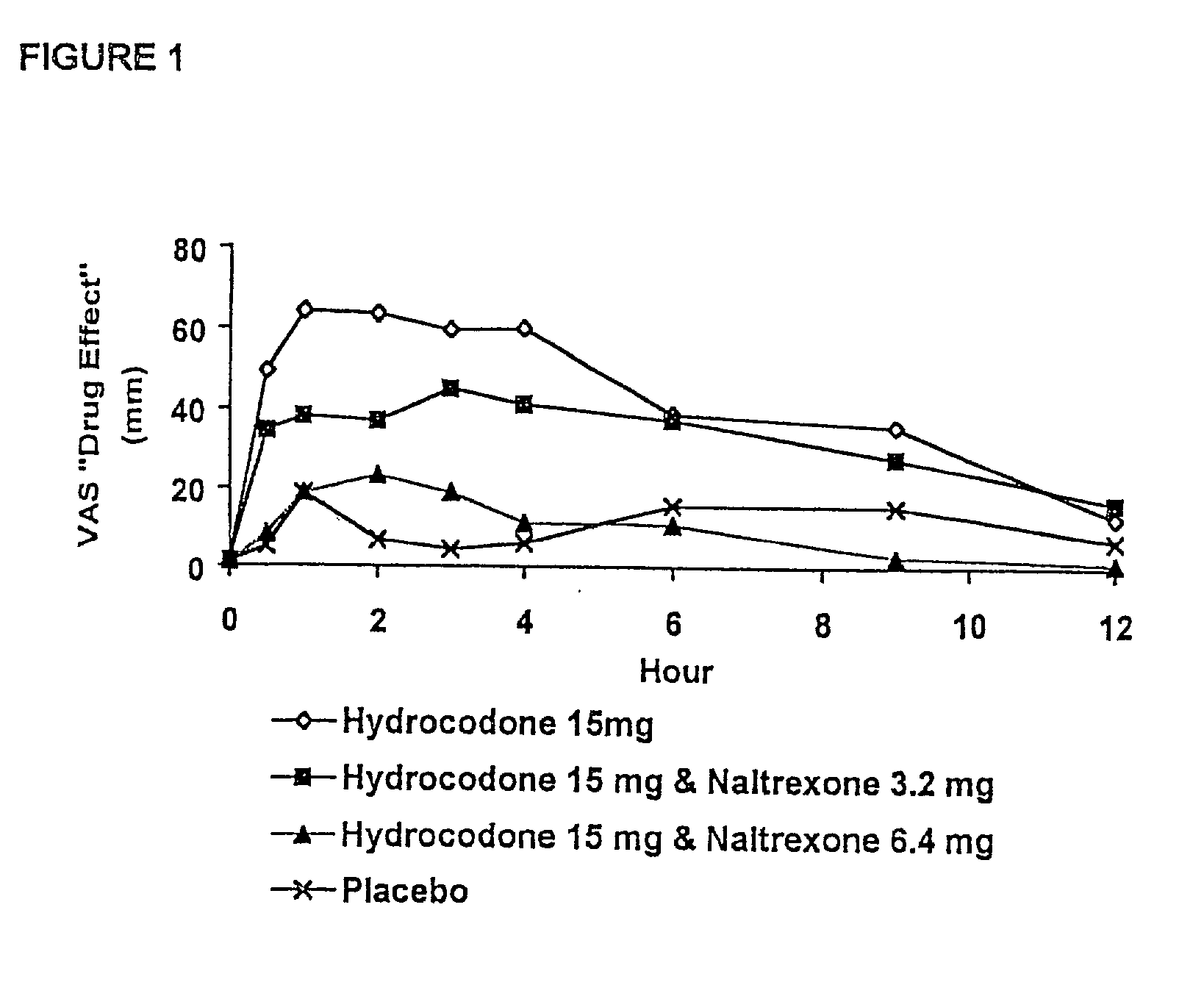
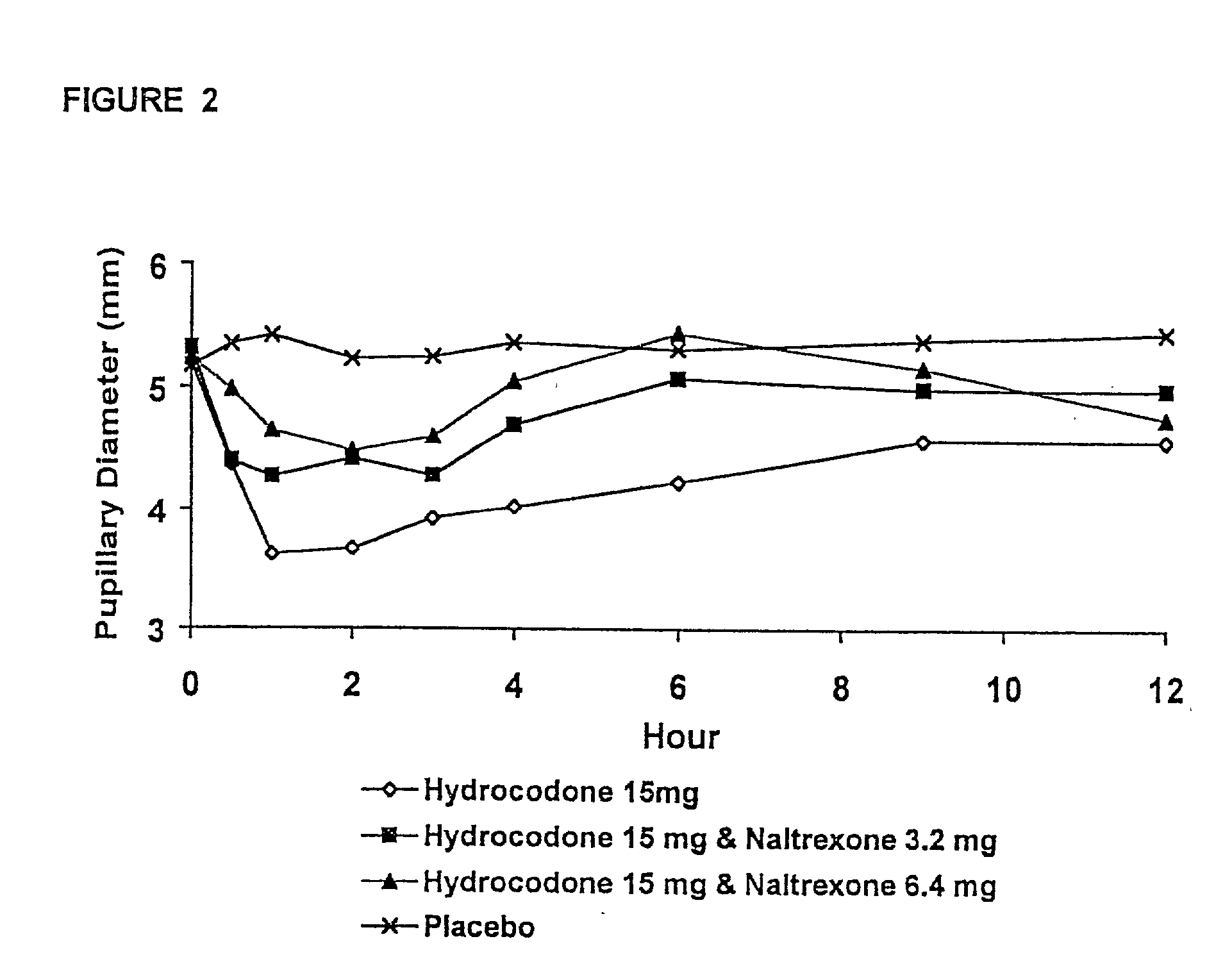
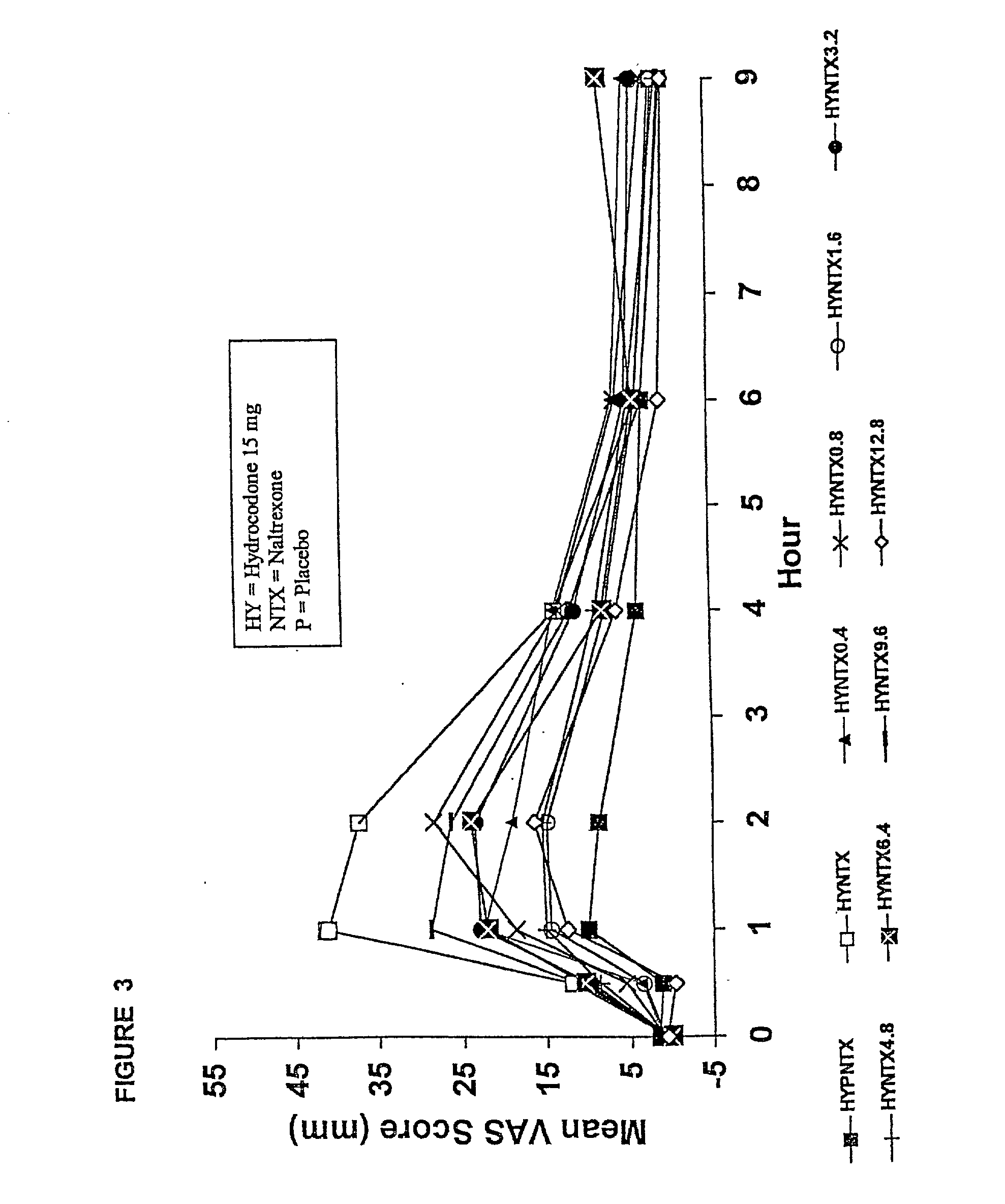
[0216] In Example 2, a ten period, randomized, crossover, single-blind study evaluating the ratio of oral naltrexone to oral hydrocodone that would nominally minimize the opioid agonist effects was conducted in normal, healthy, female volunteers. Twenty-one subjects enrolled in the study, and 16 completed the study. The ten treatments included HYIR / APAP (2 tablets of hydrocodone 7.5 and acetaminophen 750 mg per tablet, Vicodin ES.RTM.) with the following doses of naltrexone oral solution: 0.4 mg / 10 mL, 0.8 mg / 10 mL, 1.6 mg / 10 mL, 3.2 mg / 10 mL, 4.8 mg / 10 mL, 6.4 mg / 10 mL, 9.6 mg / 10 mL, 12.8 mg / 10 mL, and placebo naltrexone oral solution, as well as hydrocodone immediate-release comparator tablets (2.times.750 mg Trilisate.RTM. tablets) with placebo naltrexone oral solution. All treatments were administered under fasted conditions. A 48-hour washout period occurred between doses. Subjects were randomly assigned to ten treatment sequences of the ten treatment groups. Subjects reported ...
example 3
[0237] Example 3 presents the results of a study evaluating precipitated withdrawal in morphine dependent volunteers receiving hydrocodone immediate-release tablets and naltrexone oral solution. The study was a single-blind, single-dose, placebo-controlled naltrexone dose ascending study in subjects physically dependent on opioids. The experimental subjects (5) were opioid-dependent as determined by Narcan challenge, Addiction Severity Index scores, physical examination, observation and urine drug screen results, and were not currently seeking treatment for their addiction. To evaluate precipitated withdrawal following the coadministration of hydrocodone immediate release and naltrexone, a 30 mg dose of hydrocodone immediate release was selected to simulate a dose level used by individuals who abuse hydrocodone. This is also a dose which is considered to be equianalgesic to other commonly used opioids in opioid naive patients. The relative analgesic potency of hydrocodone is believe...
PUM
| Property | Measurement | Unit |
|---|---|---|
| time period | aaaaa | aaaaa |
| time | aaaaa | aaaaa |
| time | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More