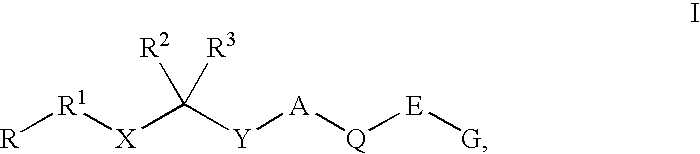
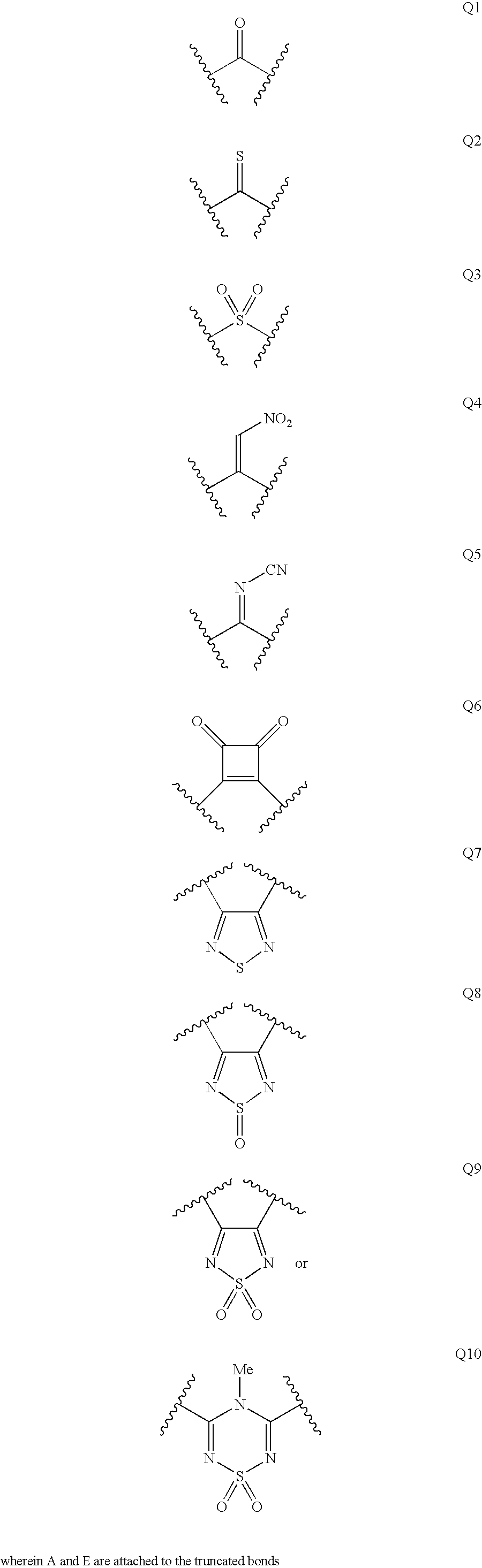
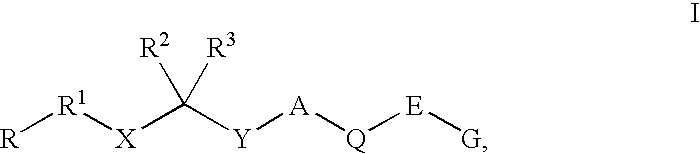
Renin inhibitors
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
preparation 1
Weinreb Amide
(R)-tert-butyl 3-(N-methoxy-N-methylcarbamoyl)piperidine-1-carboxylate
[0200]
[0201](R)-1-(tert-butoxy carbonyl)piperidine-3-carboxylic acid (25 g, 0.11 mol, 1.0 equiv), N,O-dimethylhydroxylamine hydrochloride, (10.5 g, 0.14 mol, 1.25 equiv), EDC.HCl (26.3 g, 0.14 mol, 1.25 equiv) and DIEA (48 mL, 0.28 mol, 2.5 equiv) were dissolved in CH2Cl2 (400 mL) and stirred overnight at rt. The reaction mixture was diluted with EtOAc, washed with 5% aq HCl (2×150 mL), satd aq NaHCO3 (150 mL), brine (100 mL), and dried over Na2SO4. Concentration afforded (R)-tert-butyl 3-(N-methoxy-N-methylcarbamoyl)-piperidine-1-carboxylate (24.42 g, 82%) as a clear oil.
preparation 2
Halodiphenyl Ethers from Halophenols and Benzeneboronic Acids
1-(3-Fluorophenoxy)-2-bromobenzene
[0202]
[0203]To a stirring solution of 3-fluorophenylboronic acid (2.10 g, 15 mmol), 2-bromophenol (1.77 g, 10 mmol) and Cu(OAc)2 (0.93 g, 5 mmol) in anhydrous CH2Cl2 (25 mL) is added activated 4 Å molecular sieves (˜0.1 g), followed by anhydrous Et3N (3.5 mL, 25 mmol). The resulting dark green solution is stirred at rt for 48 h. The mixture is evaporated under reduced pressure and the residue washed several times with Et2O (˜150 mL). The Et2O solution is washed with satd aq NH4Cl and 1 N aq HCl. The organic layer is evaporated and the crude product is purified by flash column chromatography to give 1-(3-fluorophenoxy)-2-bromobenzene (1.28 g, 48%) as clear oil.
[0204]The following halodiphenyl ethers were prepared following the procedure described above.
Halodiphenyl etherPhenolBoronic Acid1-bromo-3-chloro-2-[(2-2-bromo-6-chlorophenol2-methylphenylboronic acidmethylphenyl)oxy]benzene1-bromo-3...
preparation 3
Halodiphenyl Ethers from Phenoxyanilines
1-(O-tolyloxy)-2-iodobenzene
[0205]
[0206]To a solution of 2-(o-tolyloxy)aniline (40 g, 0.2 mol) in 1N aq HCl (400 mL, 0.4 mol, 2 equiv) cooled to 0° C. was added dropwise a solution of NaNO2 (18 g, 0.26 mol, 1.3 equiv) in water (520 ml). The mixture was stirred for 1 h at 0° C. and a solution of KI (83 g, 0.5 mol, 2.5 equiv) in water (500 mL) was added dropwise with vigorous stirring. After 0.5 h the mixture was warmed to 90-100° C. for 1 h, cooled to rt and washed with satd NaHSO3 until the aqueous layer became clear. The mixture was extracted with EtOAc (3×200 mL) and the combined organic layers were washed with aq Na2S2O4 and dried over Na2SO4. After evaporation of the solvent, the solution was passed through a short silica gel column to afford 1-(o-tolyloxy)-2-iodobenzene (40.0 g, 65%).
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap