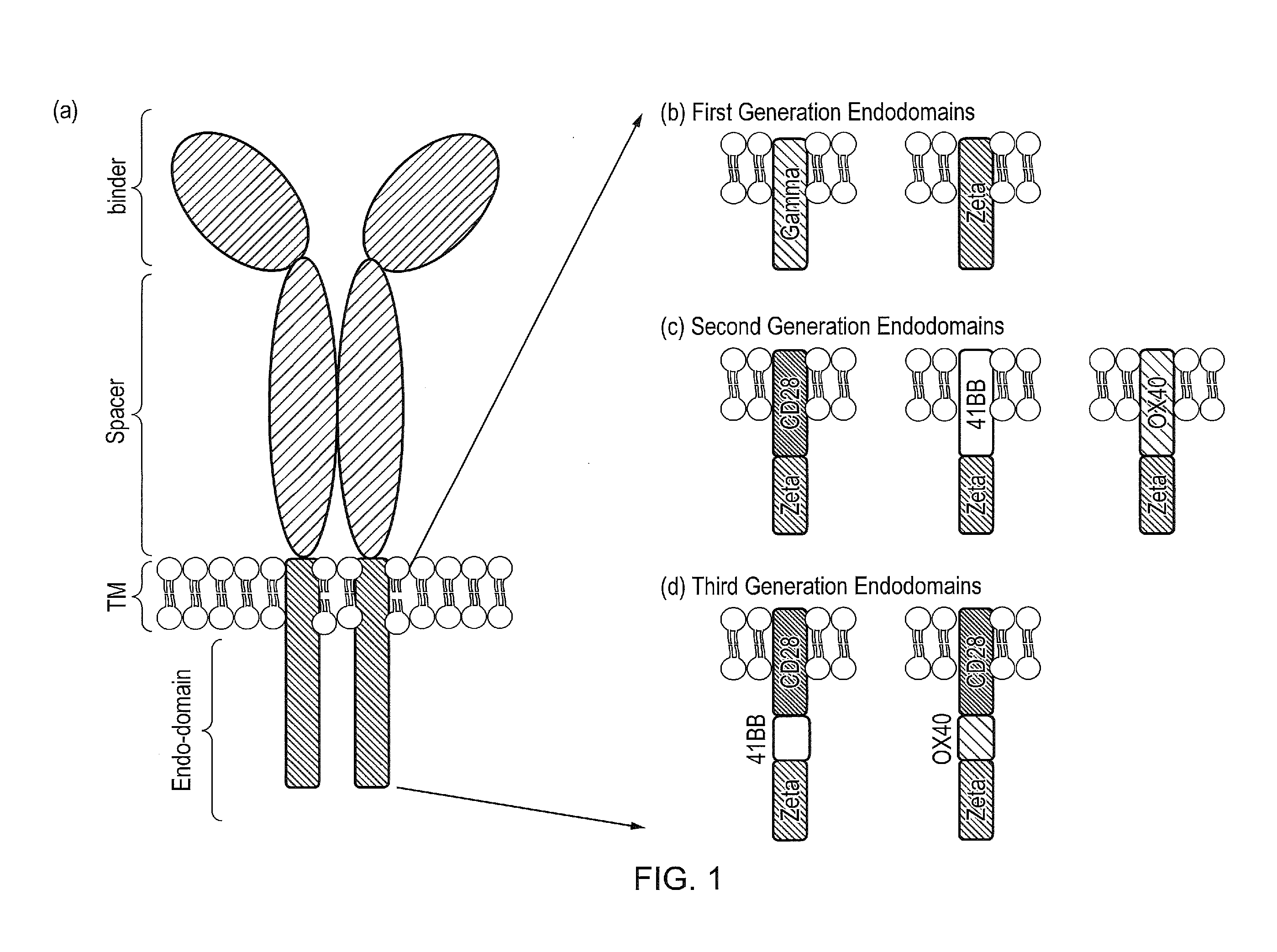
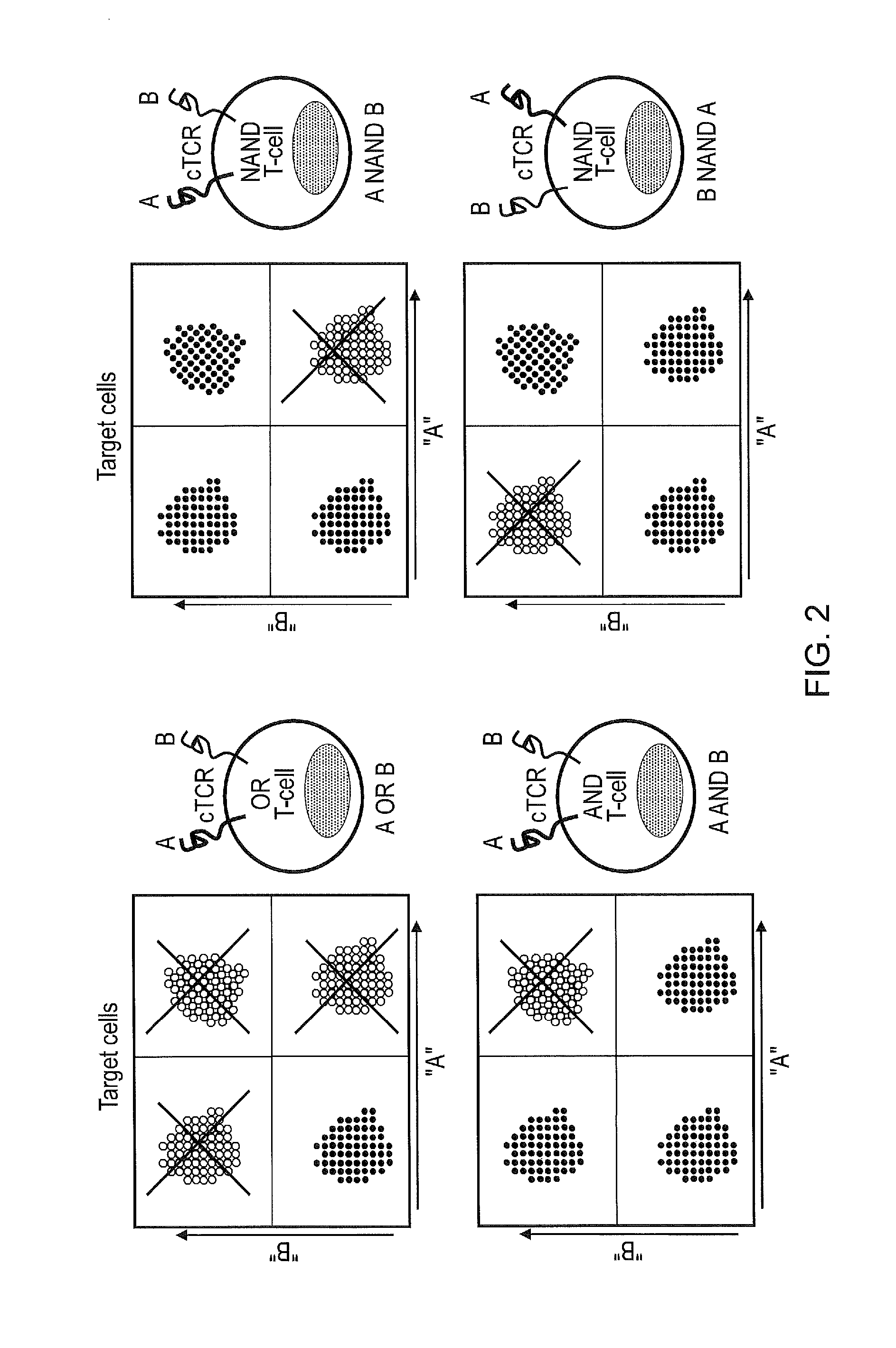
Cell
a cell and cell technology, applied in the field of cells, can solve the problems of reducing the efficacy of known immunotherapeutics, reducing tumor escape by modulation, and introducing significant challenges in designing effective treatment strategies, so as to avoid the problem of cancer escape, overcome the spatial problem, and improve the effect of therapeutic efficacy
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
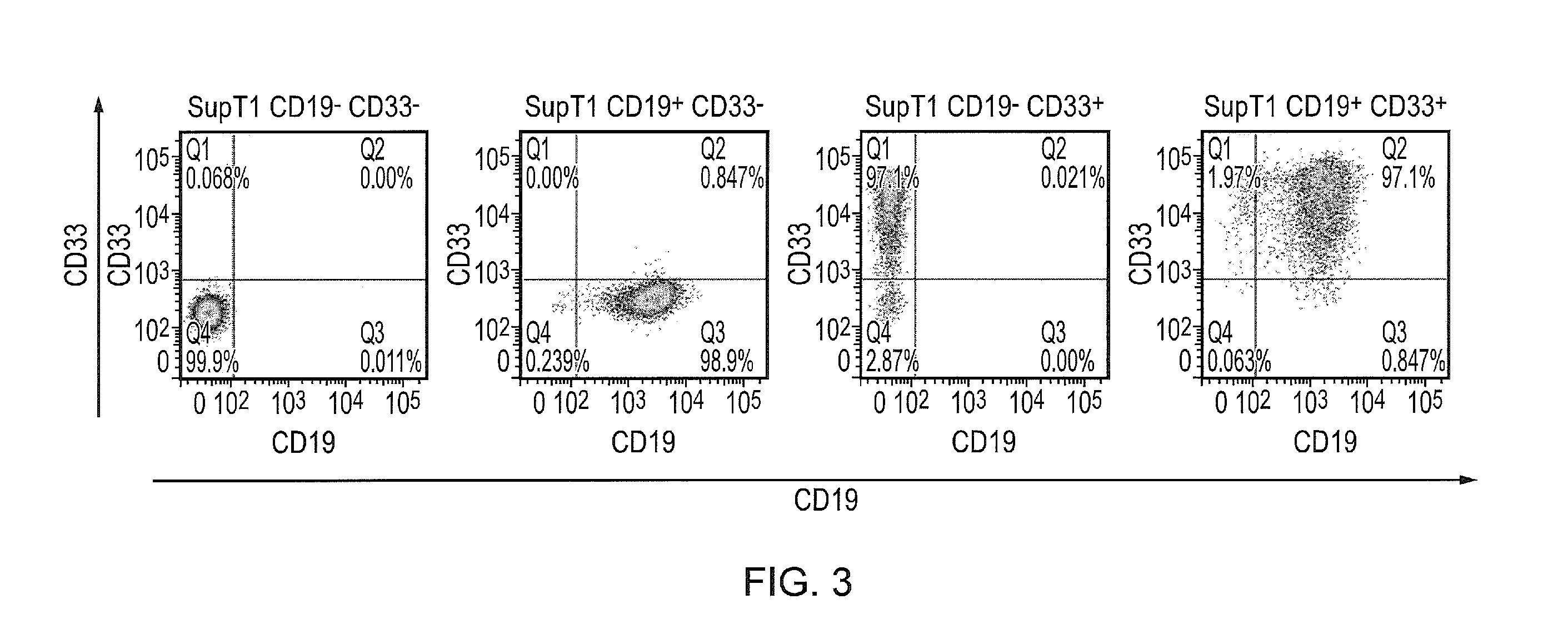
Creation of Target Cell Populations
[0362]For the purposes of proving the principle of the invention, receptors based on anti-CD19 and anti-CD33 were arbitrarily chosen. Using retroviral vectors, CD19 and CD33 were cloned. These proteins were truncated so that they do not signal and could be stably expressed for prolonged periods. Next, these vectors were used to transduce the SupT1 cell line either singly or doubly to establish cells negative for both antigen (the wild-type), positive for either and positive for both. The expression data are shown in FIG. 3.
example 2
Design and Function of the OR Gate
[0363]To construct the OR gate, a pair of receptors recognizing CD19 and CD33 were co-expressed. Different spacers were used to prevent cross-pairing. Both receptors had a trans-membrane domain derived from CD28 to improve surface stability and an endodomain derived from that of CD3 Zeta to provide a simple activating signal. In this way, a pair of independent 1st generation CARs were co-expressed. The retroviral vector cassette used to co-express the sequences utilizes a foot-and-mouth 2A self-cleaving peptide to allow co-expression 1:1 of both receptors. The cassette design is shown in FIG. 4, and the protein structures in FIG. 5. The nucleotide sequence of homologous regions was codon-wobbled to prevent recombination during retroviral vector reverse transcription.
example 3
Testing the OR Gate
[0364]Expression of both CARs was tested on the T-cell surface by staining with cognate antigen fused to Fc. By using different species of Fc domains (mouse for CD19 and rabbit for CD33), co-expression of both CARs was determined on the cell surface by staining with different secondary antibodies conjugated with different fluorophores. This is shown in FIG. 6.
[0365]Functional testing was then carried out using the mouse T-cell line BW5147. This cell line releases IL2 upon activation allowing a simple quantitative readout. These T-cells were co-cultured with increasing amounts of the artificial target cells described above. T-cells responded to target cells expressing either antigen, as shown by IL2 release measured by ELISA. Both CARs were shown to be expressed on the cell surfaces and the T-cells were shown to respond to either or both antigens. These data are show in FIG. 7.
PUM
| Property | Measurement | Unit |
|---|---|---|
| nucleic acid sequence | aaaaa | aaaaa |
| nucleic acid | aaaaa | aaaaa |
| heterogeneity | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More