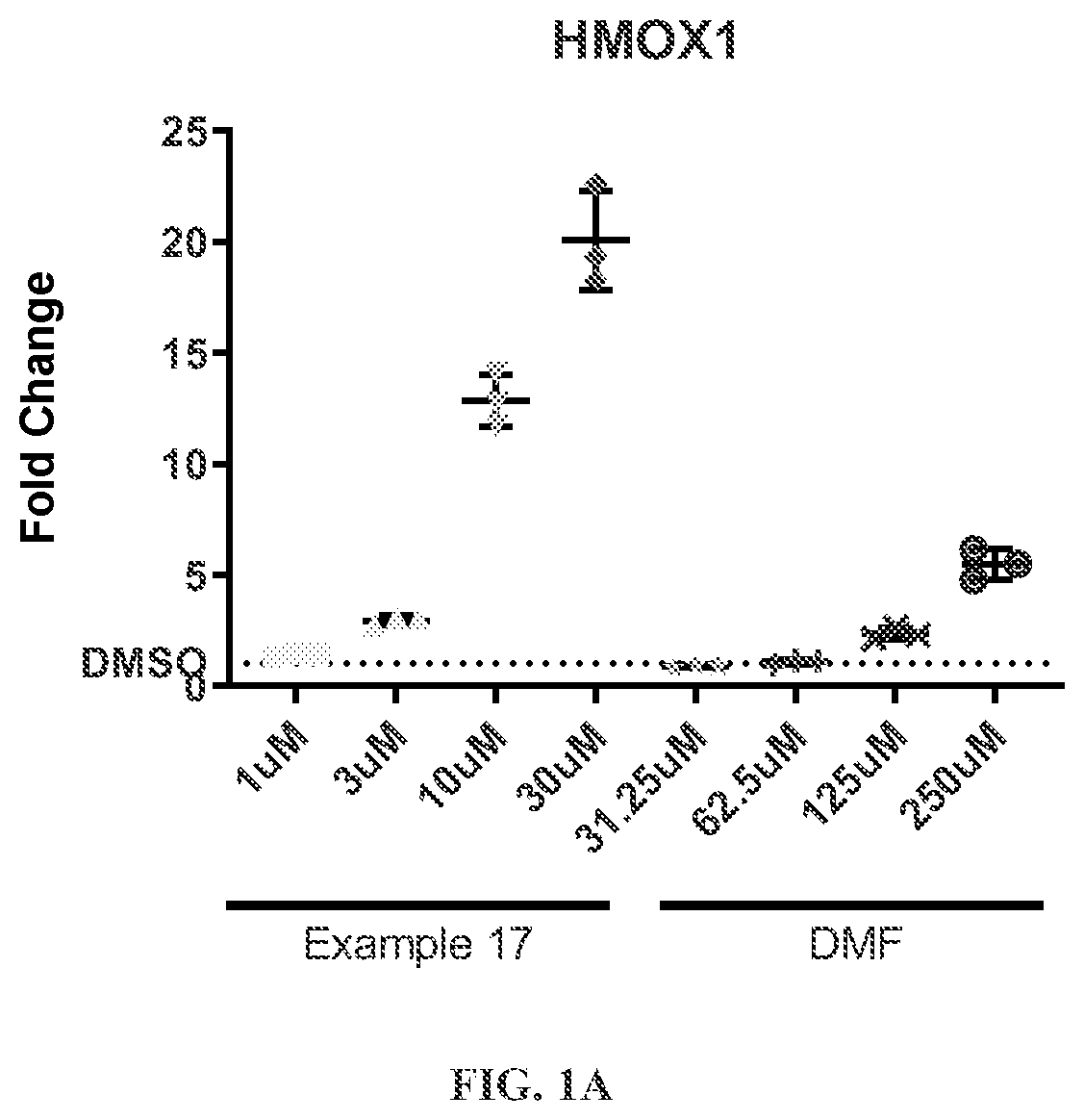
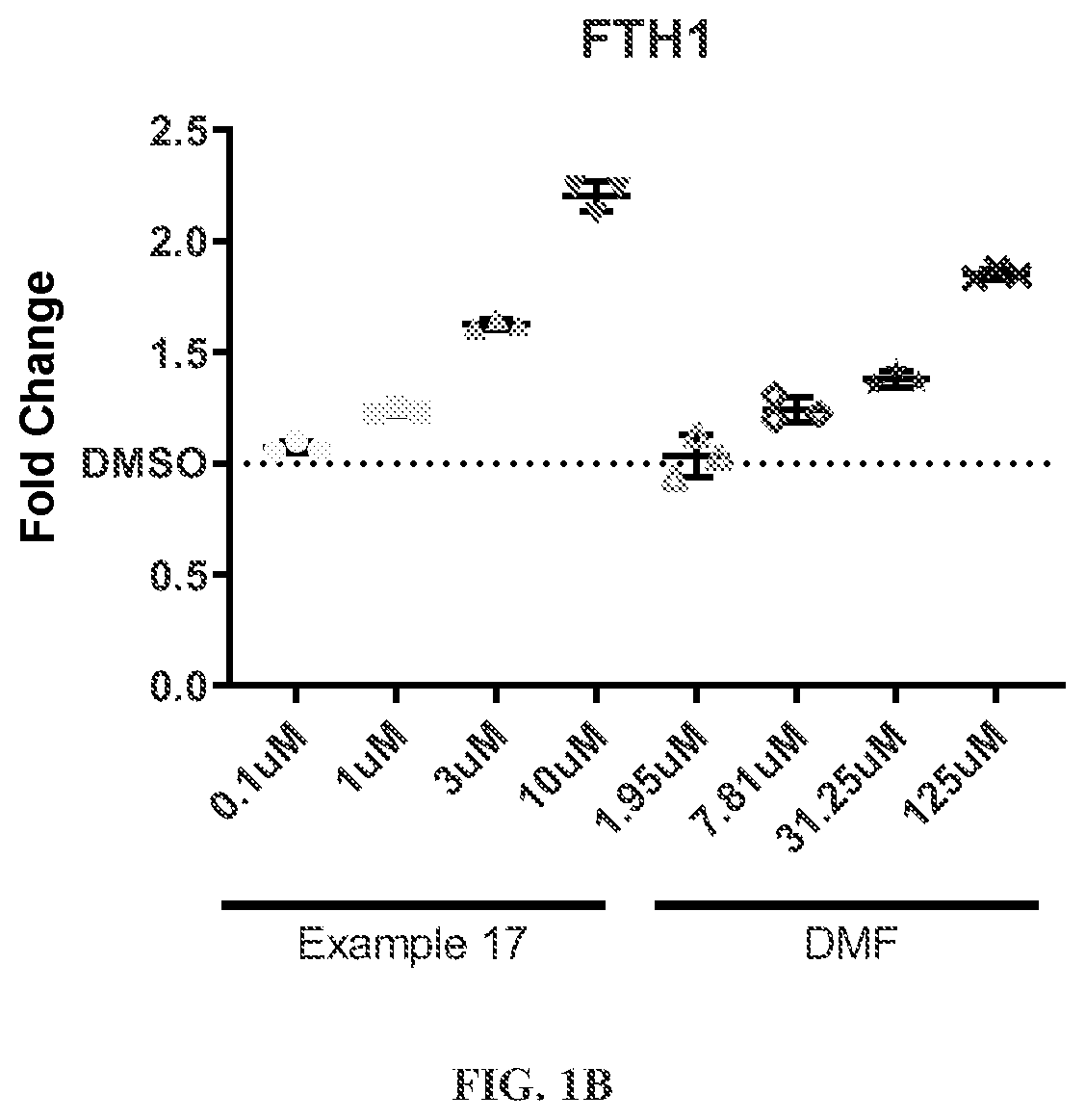
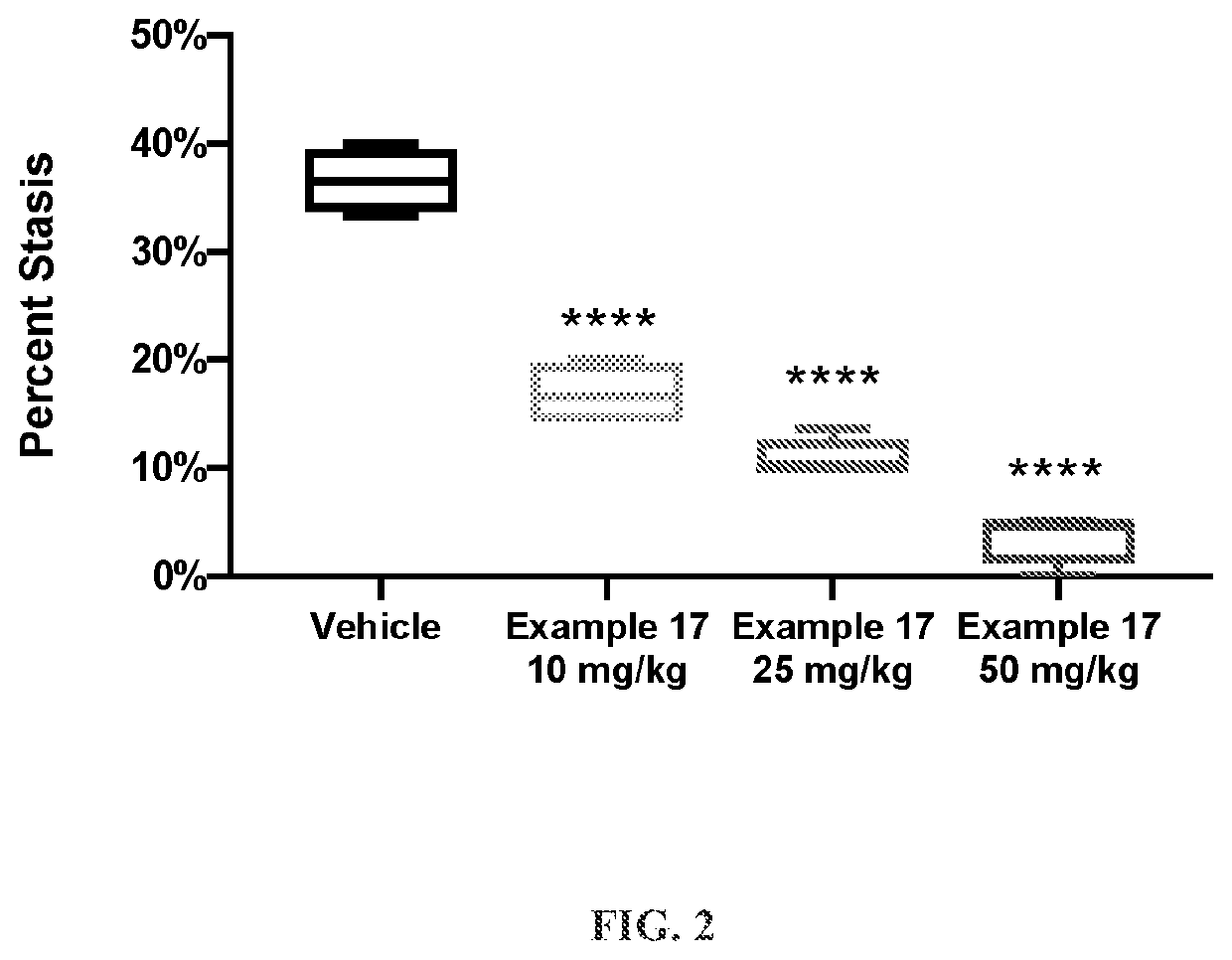
Hmox1 inducers
a technology of heme oxygenase and inducers, which is applied in the field of hmox1 (heme oxygenase 1) inducers, can solve the problems of oxidative stress imbalance, deleterious effects, damage to components such as lipids, proteins, polysaccharides, etc., and achieves reduced herg inhibition, strong influence on hmox1 protein expression, and good solubility
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
examples 1h and 1
i. Synthesis of 2-(benzo[d]oxazol-2-ylamino)-1-methyl-1H-benzo[d]imidazole-5-carboxylic acid and Synthesis of 2-(benzo[d]oxazol-2-ylamino)-N-(2-methoxyethyl)-1-methyl-1H-benzo[d]imidazole-5-carboxamide
[0143]
Step-a: Synthesis of benzo[d]oxazole-2-thiol (1a)
[0144]To a solution of 2-aminophenol (4.0 g, 36.7 mmol) in ethanol (80 mL) at RT was added powdered potassium hydroxide (3.59 g, 64.2 mmol) and carbon disulfide (20 mL, 330.3 mmol) and the reaction mixture was refluxed for 16 h. The reaction mixture was concentrated, diluted with water (100 mL) and acidified with 1 N HCl. The solid obtained was filtered and dried under vacuum to afford the title compound (4.0 g, 72%); 1H NMR (400 MHz, DMSO-d6): δ 13.85 (bs, 1H), 7.51-7.49 (m, 1H), 7.32-7.23 (m, 3H); LC-MS: m / z 152.0 (M+1)+.
Step-b: Synthesis of 2-(methylthio)benzo[d]oxazole (1b)
[0145]To a solution of benzo[d]oxazole-2-thiol (3.0 g, 19.9 mmol) in acetonitrile (50 mL) at 0° C. was added potassium carbonate (3.01 g, 21.9 mmol) and meth...
examples 2d and 2e
trahydro-2H-pyran-4-yl)-2-((6-(trifluoromethoxy)benzo[d]thiazol-2-yl)amino)-1H-benzo[d]imidazole-5-carboxylic acid and Synthesis of N-(2-methoxyethyl)-1-(tetrahydro-2H-pyran-4-yl)-2-((6-(trifluoromethoxy)benzo[d]thiazol-2-yl)amino)-1H-benzo[d]imidazole-5-carboxamide
[0153]
Step-a: Synthesis of ethyl 3-nitro-4-((tetrahydro-2H-pyran-4-yl)amino)benzoate (2a)
[0154]To a stirred solution of ethyl 4-chloro-3-nitrobenzoate (1.0 g, 4.4 mmol) in DMSO (10 mL) was added potassium carbonate (1.2 g, 8.7 mmol) and tetrahydro-2H-pyran-4-amine (0.53 g, 5.2 mmol) at RT. Then the reaction mixture was heated to 70° C. and stirred for 16 h and then cooled to RT. The reaction mixture was diluted with cold water and the solid obtained was filtered and dried under vacuum to afford the title compound (1.1 g, 86%); 1H NMR (400 MHz, DMSO-d6): δ 8.62 (d, J=2.0 Hz, 1H), 8.23 (d, J=7.8 Hz, 1H), 7.97 (dd, J=1.9 Hz, J=8.8 Hz, 1H), 7.29 (d, J=9.3 Hz, 1H), 4.29 (q, J=7.3 Hz, 2H), 3.96-3.94 (m, 1H), 3.90-3.83 (m, 2H), ...
example 3d and 3e
-methylpiperidin-4-yl)-2-((6-(trifluoromethoxy)benzo[d]thiazol-2-yl)amino)-1H-benzo[d]imidazole-5-carboxylic acid and Synthesis of N-(2-methoxyethyl)-1-(1-methylpiperidin-4-yl)-2-((6-(trifluoromethoxy)benzo[d]thiazol-2-yl)amino)-1H-benzo[d]imidazole-5-carboxamide
[0159]
Step-a: Synthesis of ethyl 4-((1-methylpiperidin-4-yl)amino)-3-nitrobenzoate (3a)
[0160]To a stirred solution of ethyl 4-chloro-3-nitrobenzoate (500 mg, 2.2 mmol) in DMFA (5 mL) was added 1-methylpiperidin-4-amine (0.30 mL, 2.6 mmol) and N-ethyldiisopropyl amine (0.8 mL, 5.0 mmol). The reaction mixture was heated to 70° C. and continued stirring for 16 h at the same temperature. Reaction mixture was cooled to RT, diluted with water (50 mL) and extracted with EtOAc (2×100 mL). The combined organic layer was washed with water (50 mL), brine (30 mL), dried over anhydrous sodium sulfate and concentrated under vacuum to afford the title compound (620 mg, 92%); 1H NMR (400 MHz, DMSO-d6): δ 8.62 (s, 1H), 8.25 (d, J=8.0 Hz, 1H)...
PUM
| Property | Measurement | Unit |
|---|---|---|
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More