A sort of 18 F-labeled ethinyl estradiol and its preparation method and application
A technology of ethinyl estradiol and 18F, which is applied in the field of 18F-labeled ethinyl estradiol and its preparation, can solve problems such as harsh reaction conditions and serious adverse reactions, achieve fast reaction rate, simplify labeling steps and purification time, and solve reaction time long effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
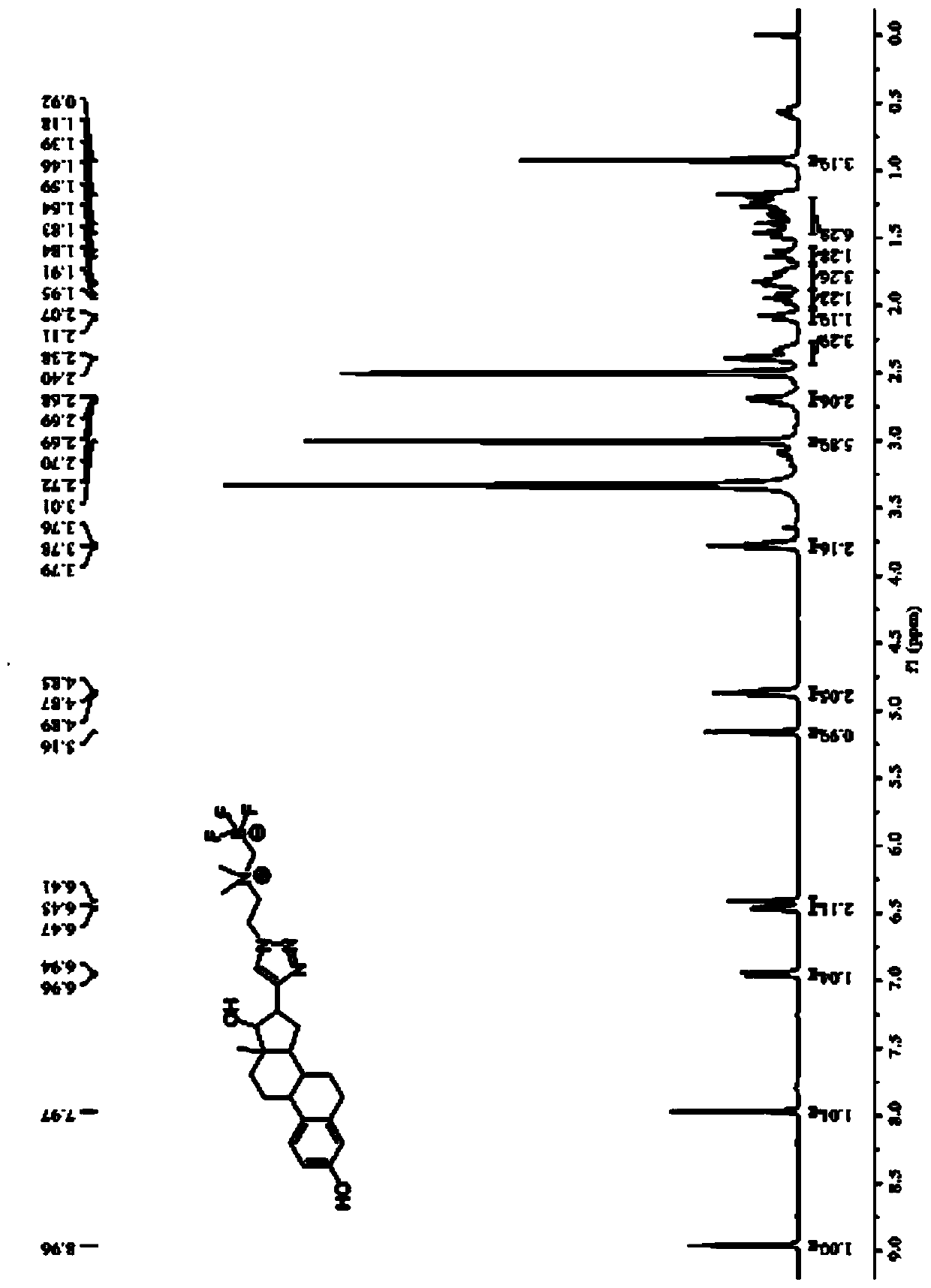
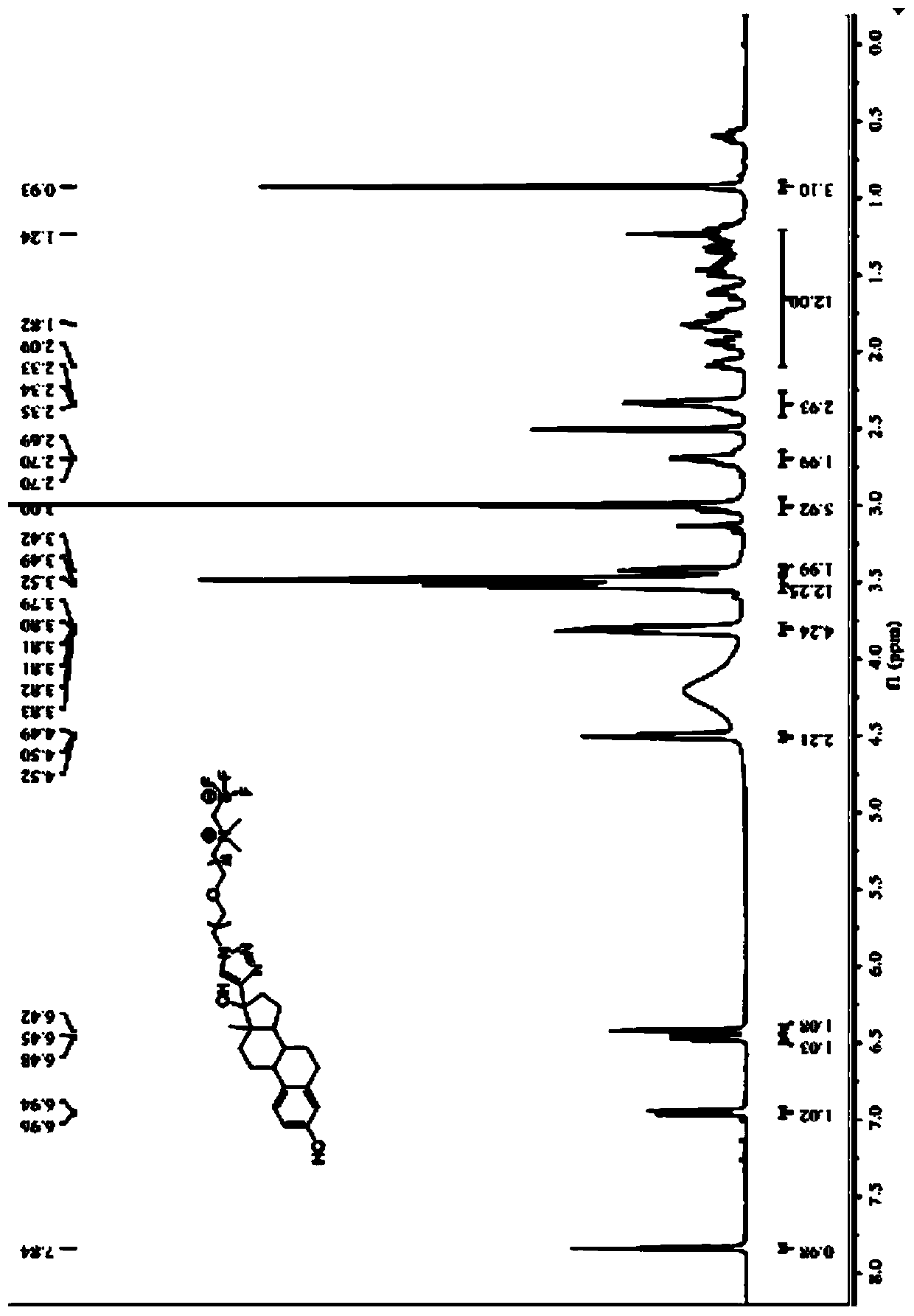
[0076] The compound described in this embodiment has the structure of formula (I) 18 F-labeled ethinyl estradiol, such as figure 1 shown, where R 1 Is ethylene, has formula (I-a) structure:
[0077]
[0078] The synthetic route of compound (I-a) is:
[0079]
[0080] The synthetic steps of the compound shown in above-mentioned formula I-a comprise:
[0081] Q1, mix 2.33g (10mmol) of compound 2-(dimethylamino) bromoethane hydrobromide shown in formula X-1 with 2.60g (40mmol) of sodium azide, add water 50ml to dissolve, nitrogen protection, 90 React at ℃ for 16h, add Na at the end of the reaction 2 CO 3 Adjust the pH value of the obtained reaction solution to neutral, carry out TLC detection on the above reaction solution, develop with dichloromethane:methanol=10:1 (v / v), the chromogenic agent is iodine simple substance, Rf=0.5, using dichloromethane Methane extraction, the obtained organic phase was dried over anhydrous sodium sulfate, and spin-dried to obtain an oi...
Embodiment 2
[0088] The structure shown in the formula (I) described in this embodiment 18 F-labeled ethinyl estradiol, such as figure 1 shown, where R 1 Is ethylene, has formula (I-a) structure:
[0089]
[0090] The synthetic route of compound (I-a) is:
[0091]
[0092] The synthetic steps of the compound shown in above-mentioned formula I-a comprise:
[0093] Q1, mix 4.66g (20mmol) of compound 2-(dimethylamino)bromoethane hydrobromide shown in formula X-1 with 4.9g (76mmol) of sodium azide, add water 100ml to dissolve, nitrogen protection, 93 React at ℃ for 15h, add Na at the end of the reaction 2 CO 3 Adjust the pH value of the obtained reaction solution to neutral, carry out TLC detection on the above reaction solution, develop with dichloromethane:methanol=10:1 (v / v), the chromogen is simple iodine, Rf=0.5, using dichloromethane Methane extraction, the obtained organic phase was dried over anhydrous sodium sulfate, and spin-dried to obtain an oily liquid, which was the c...
Embodiment 3
[0099] The structure shown in the formula (I) described in this embodiment 18 F-labeled ethinyl estradiol, such as figure 1 shown, where R 1 Is ethylene, has formula (I-a) structure:
[0100]
[0101] The synthetic route of compound (I-a) is:
[0102]
[0103] The synthetic steps of the compound shown in above-mentioned formula I-a comprise:
[0104] Q1. Mix 3493.5mg (15mmol) of compound 2-(dimethylamino)bromoethane hydrobromide shown in formula X-1 with 4095.6mg (63mmol) of sodium azide, add 75ml of water to dissolve, nitrogen protection, 87 React at ℃ for 17h, add Na at the end of the reaction 2 CO 3 Adjust the pH value of the obtained reaction solution to neutral, carry out TLC detection on the above reaction solution, develop with dichloromethane:methanol=10:1 (v / v), the chromogen is simple iodine, Rf=0.5, using dichloromethane Methane extraction, the obtained organic phase was dried over anhydrous sodium sulfate, and spin-dried to obtain an oily liquid, which ...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap