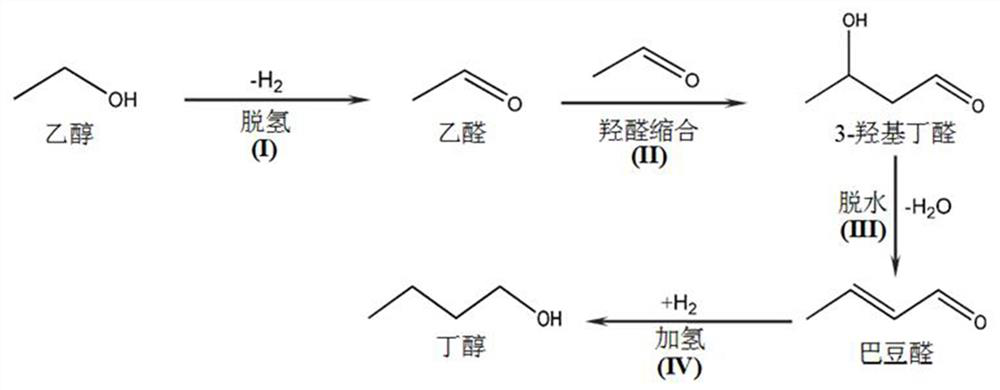
[0003] In published literature, complex homogeneous catalysts such as iridium, ruthenium, and manganese have been used in the reaction of ethanol dehydrogenation and condensation to produce n-butanol, and have achieved high selectivity and yield of butanol, but their preparation is complicated. , use sodium hydroxide, sodium ethoxide and other soluble strong bases as catalysts in the aldol condensation step of acetaldehyde, especially in the use of tank reactors, the separation of catalysts is difficult, and the reaction cannot be carried out continuously, which is not conducive to the large-scale use of butanol fuel in the future Scale production [Dowson, G.R.M., Haddow, M.F., Wass, D.F., Catalytic conversion of ethanol into an advanced biofuel: unprecedented selectivity for n-butanol, Angew. Chem. Int. Ed., 2013, 52, 9005-9008; Chakraborty, S.P, Piszel, E.P., Cassandra, E.H., Jones, W.D., Highly selective formation of n-butanol from ethanol through the Guerbet process: A tandemcatalytic approach, J.Am.Chem.Soc., 2015, 137, 14264-14267; Wingad, R.L., Gates, P.J., Street, S.T.G., Wass, D.F., Catalytic conversion of ethanol to n-butanolusing ruthenium P-N Ligand complexes, ACS Catal., 2015, 5, 5822-5826; Fu S.M., ShaoZ.H., Wang Y.J., Liu Q.,Manganese-catalyzed upgrading of ethanol into 1-butanol.J.Am.Chem.Soc.,2017,139(34):11941-11948]
Solid catalysts such as hydrotalcite, apatite, strontium phosphate, alumina-loaded copper and nickel are used in the reaction of ethanol dehydrogenation condensation to prepare n-butanol, and there are also many public reports, but the yield of its butanol is generally low (generally lower than 10wt%), and the reaction temperature and pressure are high (>300°C,>4.0MPa) [Carvalho, D.L., de Avillez, R.R., Borges, L.E.P., Mg and Al mixed oxides and the synthesis of n-butanol from ethanol, Appl. Catal.A.,2012,415-416,96-100; Tsuchida,T.,Sakuma,S.,Takeguchi,T.,Ueda,W.,Yoshioka,T.,Reaction of ethanolover hydroxyapatite affected by Ca / P ratio of catalyst, J. Catal., 2008, 259, 183-189; OgO, S., Onda, A., Yanagisawa, K., Selective synthesis of 1-butanol from ethanolover strontium phosphate hydroxyapatite catalysts, Appl. Catal. A., 2011, 402,188-195; OgO,S.,Onda,A.,Yanagisawa,K.,Iwasa,Y.,Hara,K.,Fukuoka,A.,1-Butanolsynthesis from ethanol over strontium phosphate hydroxyapatite catalysts with various Sr / P ratios, J.Catal.,2012,296,24-30; Riittonen,T.,Toukoniitty,E.,Madnani,D.K.,Leino,A.R.Kordas.,One-pot liquid-phase catalytic conversion ofethanol to 1-butanol over aluminum oxide— the effect of the active metal on the selectivity, Catalysts, 2012, 2, 68-84; Dziugan, P., Jastrzabek, K.G., Binczarski, M., K arski,S.Continuous catalytic coupling of raw bioethanol into butanol and higher homologues,Fuel,2015,158,81-90; Jordison,T.L.,Lira,C.T.,Miller.D.J.,Condensed phase ethanol conversion to higher alcohols,Ind.Eng.Chem .Res.,2015,54,10991-11000; Riittonen,T.,Eranen,K.,Maki-Arvela,P.,Shchukarev,A.,Rautio,A.R.,Continuous liquid-phase valorization of bio-ethanol towards bio- butanolover metal modified aluminum, Renew. Energy, 2015, 74, 369-378]
Copper supported by cerium oxide with a large specific surface exhibits a high ethanol conversion rate of 67% and a butanol yield of 30% in a supercritical carbon dioxide medium (260 ° C, 10 MPa), but the reaction pressure of up to 10 MPa increases the reaction pressure. Requirements for equipment and its materials, and the production capacity of butanol per unit volume reactor is low, so its industrial application is also restricted to a certain extent [Earley J.H., BourneR.A., Watson M.J., Poliakoff M., Continuous catalytic upgrading of ethanol to n-butanol and>C4products over Cu / CeO2catalysts in supercritical CO2.GreenChem.,2015,17:3018-3025]
However, in the latest literature, activated carbon-supported copper-cerium oxide catalysts were used in the catalytic ethanol upgrading to butanol reaction, and achieved 46% ethanol conversion and nearly 20wt% butanol yield under relatively mild reaction conditions. rate, but its catalyst activity has a certain degree of decline in long-term evaluation[Jiang D.H., WuX.Y., Mao J., Ni J., Li X.N., Continuous catalytic upgrading ethanol to n-butanolover Cu-CeO2 / AC catalysts ,Chem.Commun.,2016,52:13749-13752; Activated carbon supported copper-metal oxide catalyst and its preparation method and application, Chinese invention patent, 201610399455.4]
 Login to View More
Login to View More