Pseudotyped oncolytic viral delivery of therapeutic polypeptides
An oncolytic virus and therapeutic technology, applied in the field of pseudotyped oncolytic virus delivery of therapeutic polypeptides, can solve the problem of suboptimal anti-tumor efficacy
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0361]Example 1: Preparation of pseudotyped VSV-G
[0362] The following protocol was used to prepare an exemplary pseudotyped VSV-G by combining VSV-glycoprotein (VSV-GP) with HIV1-gag and rev proteins.
[0363] Cell culture and transfection: DNA from the following packaging plasmids was mixed and prepared for transfection into 293T cells: pMDLg / pRRE expressing HIV-1 GAG / POL; pRSV / REV expressing HIV-1 REV; and pMD2.G 5 60 5.8 VSV glycoproteins. Add the DNA mixture to 500 μL of pre-warmed Optimem II medium. Prepare a working stock of polyethyleneimine transfection reagent (PEI) at 1 μg / μL in 1xPBS (pH 4.5) and add 88 μL of the working stock to the mixture, maintaining a 4:1 v / w ratio of PEI:DNA . The mixture was vortexed briefly and left at room temperature for 5-10 minutes to form the PEI:DNA transfection complex. Will add up to 2.5x 10 6 Low passage (less than P20) 293T cells were seeded per 15 cm dish in 15 mL of DMEM supplemented with 10% serum and 1% Pen / Strep. Two ...
Embodiment 2
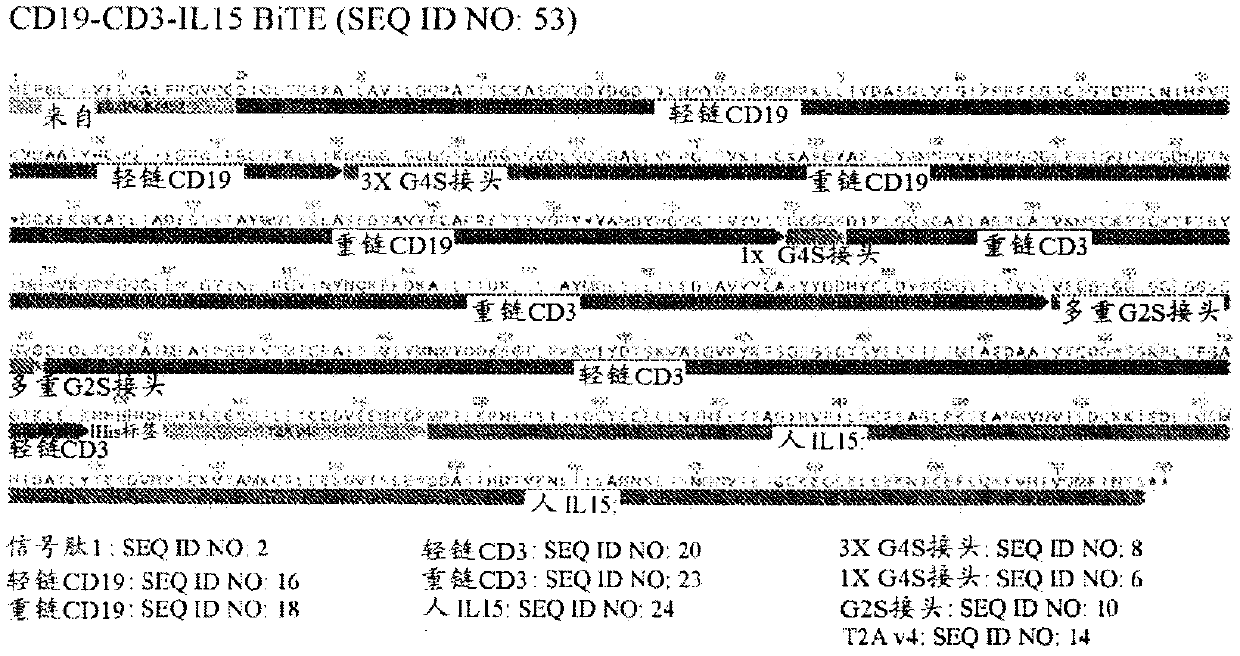
[0365] Example 2: Construction of pseudotyped VSV-G expressing CD28-CA125 bispecific antibody adapter molecule
[0366] Pseudotyped VSV-G was prepared as described in Example 1 and further processed to express nucleic acid encoding an adapter polypeptide comprising an activation domain comprising an anti-CD28 molecule and an antigen recognition domain comprising an anti-CA125 molecule , and a nucleic acid encoding an anti-PD1 immunomodulatory peptide. The resulting oncolytic virus was a pseudotyped oncolytic VSV-G virus encoding a CD28-CA125 adapter molecule and an anti-PD1 therapeutic molecule (CD28-CA125-PD1 VSV-G).
Embodiment 3
[0367] Example 3: CD28-CA125-PD1 VSV-G activates human T cells and exhibits antitumor activity
[0368] Human T cells were infected with pseudotyped CD28-CA125-PD1 VSV-G virus. Between 24 and 48 hours after viral infection, T cell culture media were collected and examined for the presence of pro-inflammatory cytokines. These results will show that T cells are activated by CD28-CA125-PD1 VSV-G, as shown by the presence of pro-inflammatory cytokines such as IFN-β and Evidenced by IL-2.
[0369] EphA2-overexpressing gastric cancer cells from the KATO3 cell line were infected with pseudotyped CD28-CA125-PD1 VSV-G or non-pseudotyped CD28-CA125-PD1 VSV virus, and cell proliferation was assessed. These results will show that cell proliferation is significantly reduced in KATO3 cells infected with pseudotyped CD28-CA125-PD1 VSV-G compared to KATO3 cells infected with non-pseudotyped CD28-CA125-PD1 VSV virus.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More