Primer pair and kit for detecting polymorphism of Etanercept-medication-related genes
A technology of gene polymorphism and etanercept, which is applied in the field of primer pairs and kits for detecting etanercept drug-related gene polymorphisms, can solve the problems that cannot meet the needs of typing
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0113] Embodiment 1: the preparation of kit (30 tests / box)
[0114] 1. Design and synthesis of primers and probes
[0115] For human SLCO1C1 gene, PTPRC gene, STAT4 gene, HLA-E gene, TRAF1 gene and KLRD1 gene, select specific mutation sites rs3794271, rs10919563, rs7574865, rs1264457, rs3761847 and rs2302489, and select primers and probes designed at the mutation sites Points and nearby conserved regions, avoiding SNPs in the primer-binding region (retrieve the SNP of the target gene sequence through the online NCBI website), perform PrimerBlast through the online NCBI website, design allele-specific PCR amplification primers, and confirm the primers For the specific amplification of the PCR primer, when there is a mismatch between the base at the 3' end of the PCR primer and its template DNA, the amplification efficiency will drop sharply, and the PCR amplification signal will only appear when the 3' base of the primer is paired with the template. The probe is located in the...
Embodiment 2
[0174] Embodiment 2: the use of kit
[0175] 1. Sample testing
[0176] Prepare the system according to the number of templates: take the PCR reaction tube, add the corresponding primer solution, PCR premix solution, sterilized purified water, add sample DNA, sterilized purified or positive control substance as template to form a PCR reaction system. Perform PCR amplification according to the PCR reaction procedure.
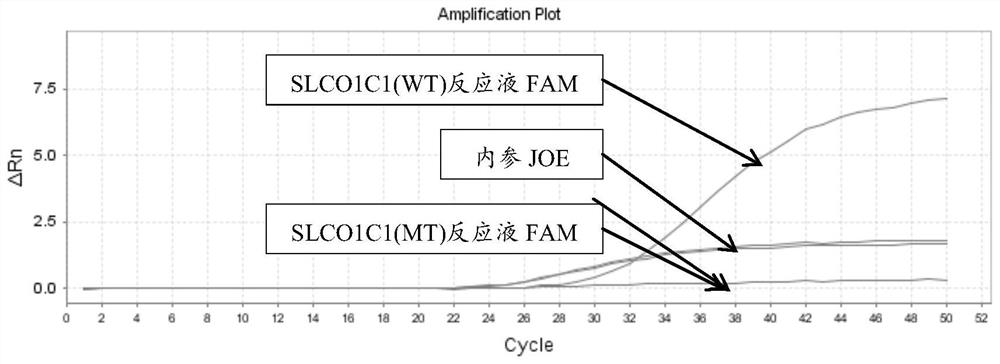
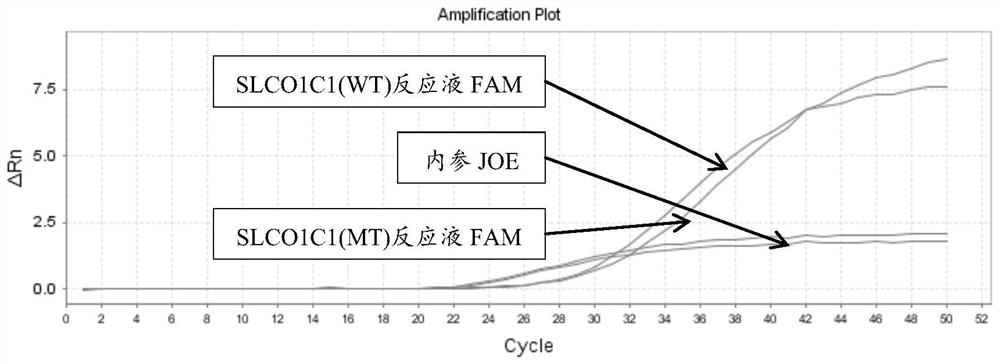
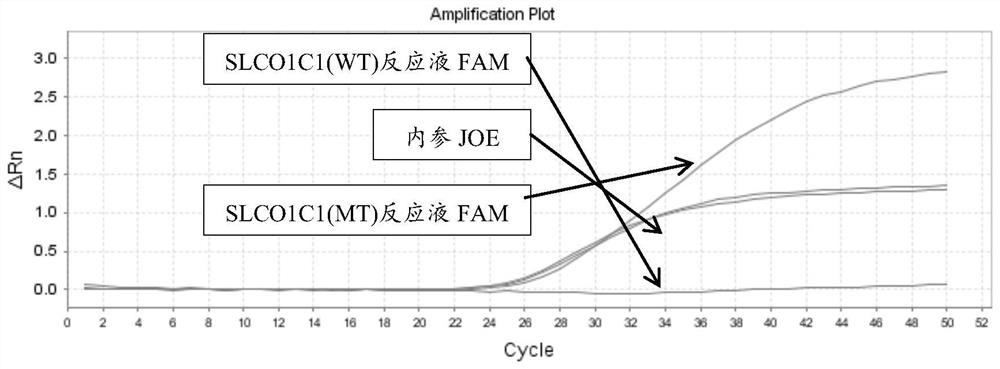
[0177] There are two kinds of reaction solutions for each site: wild (WT) and mutant (MT), and there are 12 kinds of reaction solutions for 6 sites. Each reaction solution was prepared as follows:
[0178] Table 18. Preparation composition of each reaction solution
[0179]
[0180] The system reaction procedure is as follows:
[0181] Table 19. PCR reaction program
[0182]
[0183] 2. ABI7500 fluorescent quantitative PCR
[0184] Press the power button on the right to start ABI 7500. After starting up, the "power" indicator on the left end of the m...
Embodiment 3
[0219] Embodiment 3: Large sample verification of the kit
[0220] 1. According to the preparation method shown in Example 1, the relevant components of the kit were prepared and stored at -20°C for later use.
[0221] 2. Take 30 cases of known whole blood samples, use "nucleic acid extraction or purification reagents" (record number: Xiangchang Machinery Equipment 20160167) to extract sample DNA, and use a nucleic acid protein analyzer to detect DNA sample concentration, 30 samples A260 / 280 All are between 1.6 and 2.0.
[0222] 3. According to the steps shown in Example 2, add DNA samples and perform detection on an ABI 7500 fluorescent quantitative PCR instrument.
[0223] 4. According to the interpretation standard shown in embodiment 2, the results are interpreted and counted (the test result coincidence rate statistics), the sample coincidence rate is 100%; the test result specific information is as follows:
[0224]
[0225]
[0226]
[0227]
[0228]
...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap