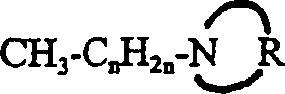Microbicidal compositions and methods using combinations of propiconazole and N-alkyl heterocycles and salts thereof
A heterocyclic compound and antimicrobial technology, applied in the field of propiconazole, can solve the problem of high concentration treatment cost
Inactive Publication Date: 2002-10-16
BUCKMAN LAB INT INC
View PDF11 Cites 3 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
The concentration of traditional microbicides mentioned above and the corresponding treatment costs are relatively high
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1
[0064] 1 BUSAN 1292 is an emulsifiable concentrate available from Buckman Laboratories, Inc. containing propiconazole (24.7%), emulsifier and water;
[0065] 2 Pzole / DDM: 10% propiconazole, 40% dodecylmorpholine (DDM).
Embodiment 2
[0068] 1 BUSAN 1292 is an emulsifiable concentrate available from Buckman Laboratories, Inc. containing propiconazole (24.7%), emulsifier and water;
[0069] 2 Pzole / DDM: 10% propiconazole, 40% dodecylmorpholine (DDM).
Embodiment 3
[0072] 1 BUSAN 1292 is an emulsifiable concentrate available from Buckman Laboratories, Inc. containing propiconazole (24.7%), emulsifier and water;
[0073] 2 Pzole / DDM: 10% propiconazole, 40% dodecylmorpholine (DDM).
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
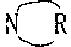
An improved microbicide propiconazole, (RS)-1-[2-(2,4-dichlorophenyl)-4-propyl-1,3-dioxolane-2-ylmethyl] -1H-Method of microbicidal efficacy of 1,2,4-triazole. In this method, propiconazole and a synergist, an N-alkyl heterocyclic compound, a salt thereof or a mixture thereof, are applied to a matrix or aqueous system that is prone to microorganisms. The dosage of N-alkyl heterocyclic compound, its salt or its mixture can effectively improve the microbicidal activity of the microbicide. N-alkyl heterocyclic compounds have the structure of formula (I), CH3-CnH2n- (I), where the parameter n is 5-17, and the heterocyclic ring defined as (II) is a substituted or unsubstituted 4-8 membered ring. The present invention describes microbicidal compositions wherein propiconazole and an N-alkyl heterocyclic compound, a salt thereof, or a mixture thereof are present in a total amount effective to control the growth of at least one microorganism. Methods of controlling microbial growth on various substrates and in various aqueous systems are also described. In the leather industry, wood industry, paper industry, textile industry, agricultural industry, coating industry and industrial process water, the microbicidal composition of propiconazole and N-alkyl heterocyclic compounds, their salts or their mixtures is particularly useful. Microbicides.
Description
field of invention [0001] The present invention relates to compositions and methods for controlling the growth of microorganisms on various substrates and in aqueous systems. More specifically, the present invention relates to propiconazole, also known as (RS)-1-[2-(2,4-dichlorophenyl)-4-propyl-1,3-dioxolane-2 -Alkylmethyl]-1H-1,2,4-triazole, and the composition of N-alkyl heterocyclic compound, its salt or its mixture, wherein, N-alkyl heterocyclic compound, its salt or its mixture Enhanced microbicidal activity of propiconazole. Background of the invention [0002] A large number of commercial, industrial, agricultural and wooden materials or products are susceptible to attack or degradation by microorganisms, which reduces or destroys their economic value. Examples of such materials or products include surface coatings, wood, seeds, plants, leather and plastics. The various temperatures at which the aforementioned materials or products are manu...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): A01N25/00A01N43/36A01N43/40A01N43/46A01N43/50A01N43/653A01N43/76A01N43/78A01N43/80A01N43/84
CPCA01N43/653A01N43/84A01N43/80A01N43/76A01N43/50A01N43/46A01N43/40A01N43/36A01N2300/00
Inventor 戴维·奥庞玛里琳·S·怀特摩尔M·谢尔登·埃利斯罗伯特·H·米勒周向东迈克尔·E·埃尔莫尔
Owner BUCKMAN LAB INT INC