Immunostimulatory polynucleotide/immunomodulatory molecule conjugates
a technology of immunomodulatory molecule and immunonucleotide, which is applied in the field of immunonucleotide/immunomodulatory molecule conjugates, can solve the problems of general absence or weakness
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example ii
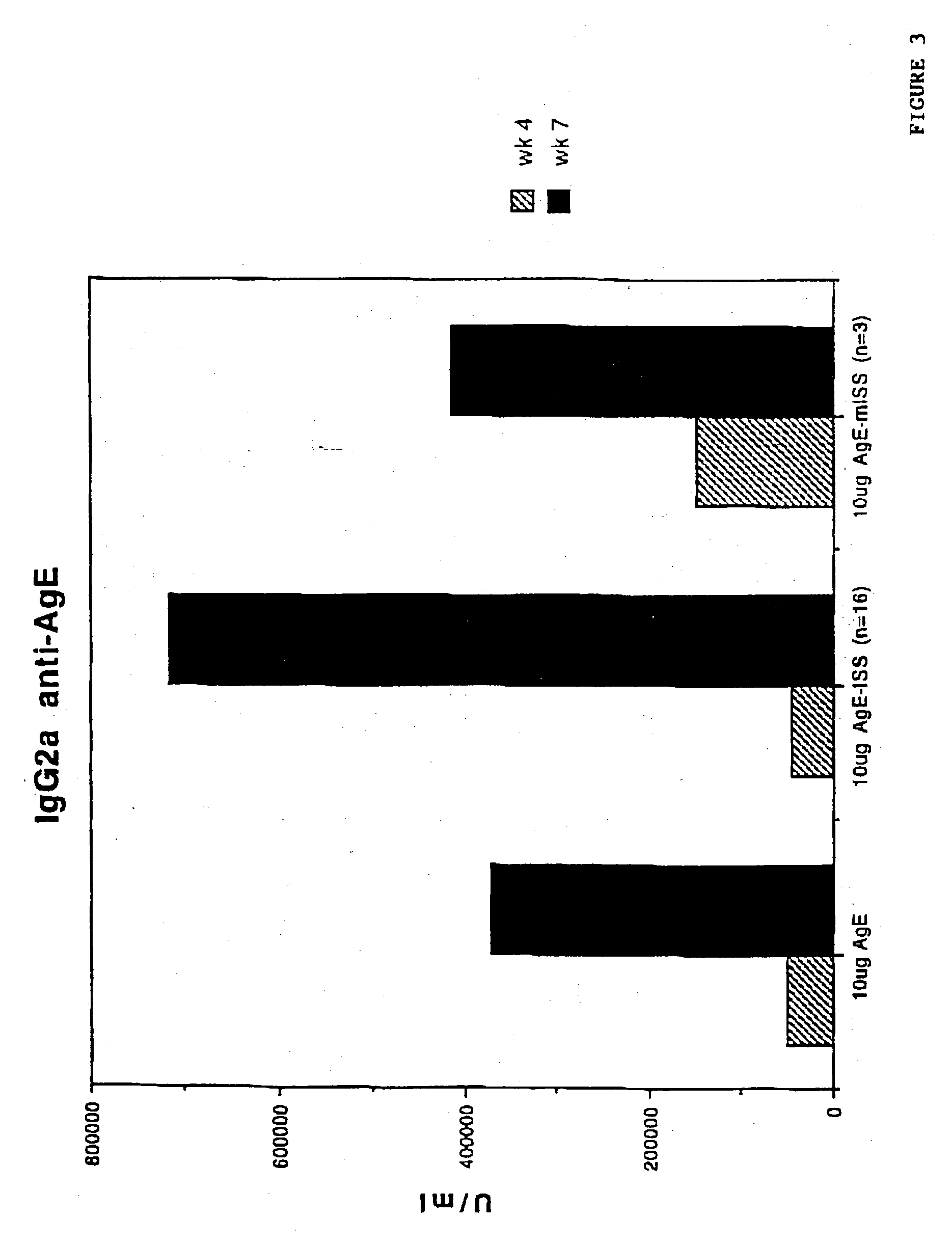
[0142] Suppression of IgE Antibody Response to Antigen by Immunization with ISS-PN / IMM
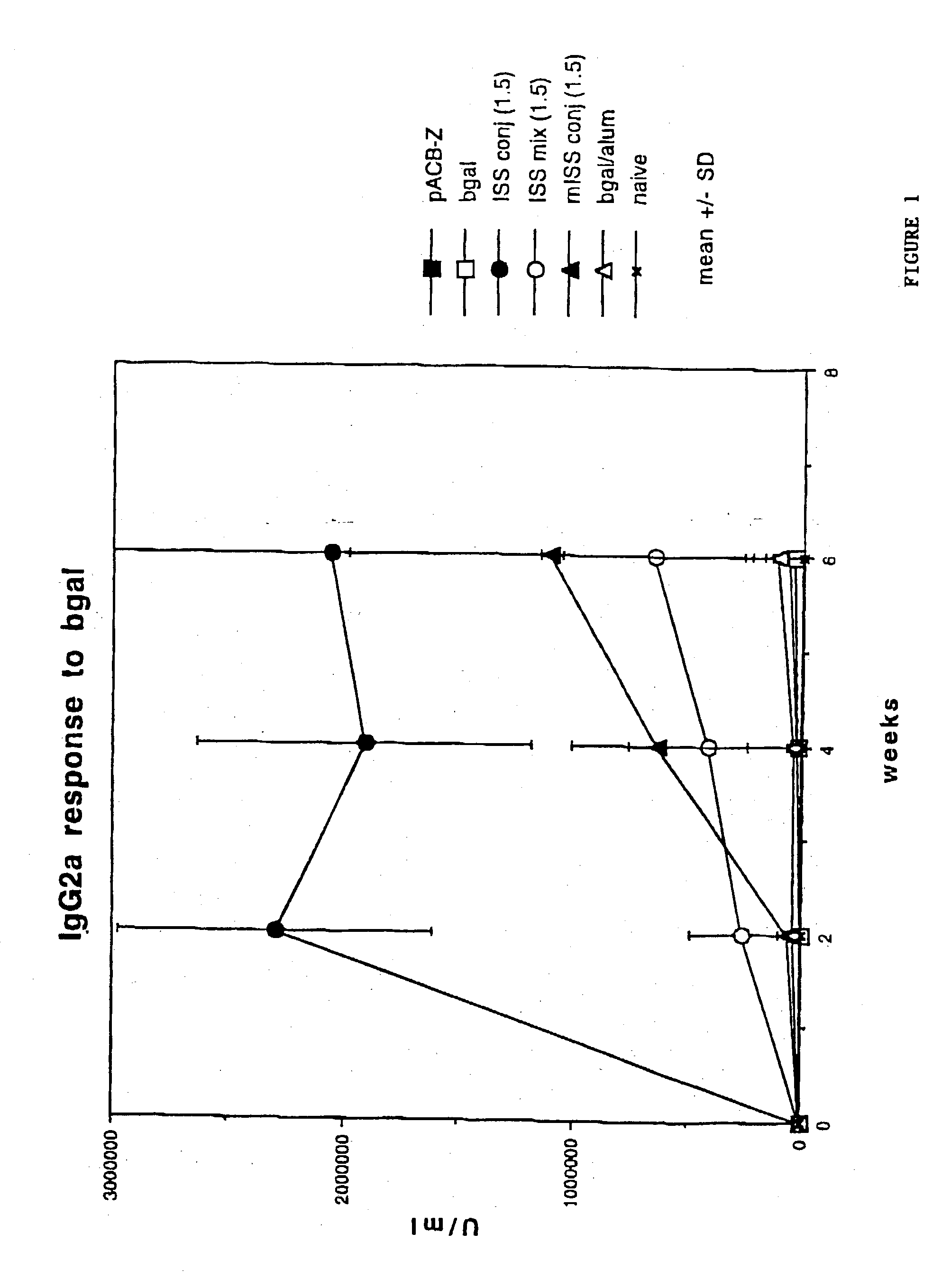
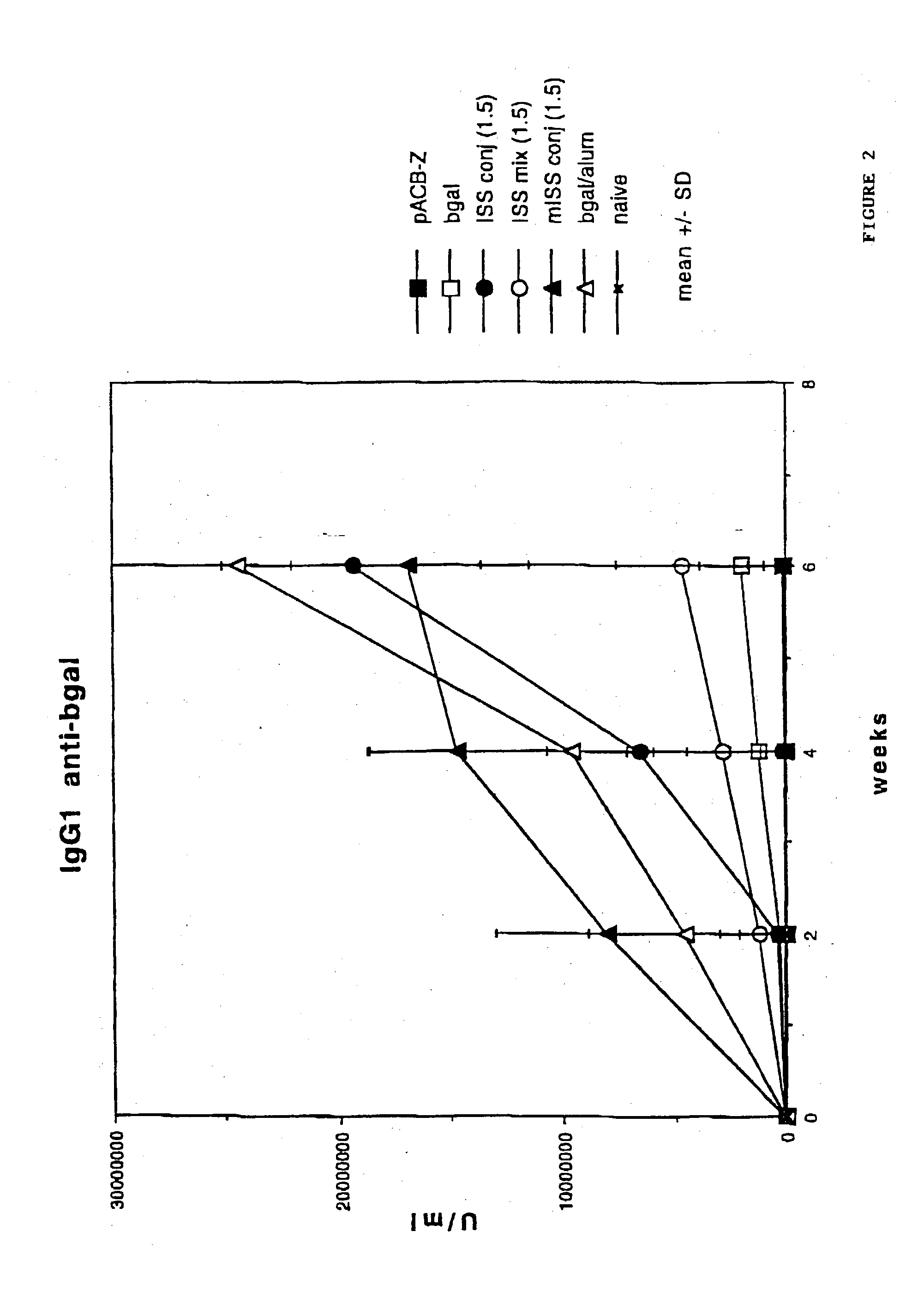
[0143] To demonstrate the IgE suppression achieved through stimulation of a Th1 type cellular immune response in preference to a Th2 type cellular immune response, five to eight week old Balb / c mice were immunized with AgE as described in the previous Example.
[0144] IgE anti-Age were detected using a solid phase radioimmunoassay (RAST) in a 96 well polyvinyl plate (a radioisotopic modification of the ELISA procedure described in Coligan, "Current Protocols In Immunology", Unit 7.12.4, Vol. 1, Wiley & Sons, 1994), except that purified polyclonal goat antibodies specific for mouse e chains were used in lieu of antibodies specific for human Fab. To detect anti-AgE IgE, the plates were coated with AgE (10 .mu.g / ml). The lowest IgE concentration measurable by the assay employed was 0.4 ng of IgE / ml.
[0145] Measuring specifically the anti-antigen response by each group of mice, as shown in FIG. 5, anti-Ag...
example iii
INF.gamma. Levels in Mice After Delivery of ISS-PN / IMM
[0147] BALB / c mice were immunized with .beta.gal as described in Example I then sacrificed 24 hrs later. Splenocytes were harvested from each mouse.
[0148] 96 well microtiter plates were coated with anti-CD3 antibody (Pharmingen, La Jolla, Calif.) at a concentration of 1 .mu.g / ml of saline. The anti-CD3 antibody stimulates T cells by delivering a chemical signal which mimicks the effects of binding to the T cell receptor (TCR) complex. The plates were washed and splenocytes added to each well (4.times.10.sup.5 / well) in a medium of RPMI 1640 with 10% fetal calf serum. Supernatants were obtained at days 1, 2 and 3.
[0149] Th1 cytokine (INF.gamma.) levels were assayed with an anti-INF.gamma. murine antibody assay (see, e.g., Coligan, "Current Protocols in Immunology", Unit 6.9.5., Vol. 1, Wiley & Sons, 1994). Relatively low levels of INF-.gamma. would be expected in mice with a Th2 phenotype, while relatively high levels of INF-.gamma...
example iv
Boosting of CTL Responses by ISS-PN / IMM
[0151] A mixture of lymphoytes was obtained and contacted with .beta.gal antigen alone or as part of the constructs and mixtures described in Example I. As shown in FIG. 6, CTL production in response to ISS-PN / IMM was consistently higher than the response to antigen delivered in other forms; even twice as high than in animals treated with an unconjugated mixture of ISS-PN and IMM antigen.
[0152] In the experiment, the higher values for the mice treated with M-ISS-PN / IMM after antigen challenge as compared to the conventionally immunized mice is most likely owing to the antigen carrier properties of DY1019.
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| frequency | aaaaa | aaaaa |
| Structure | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More