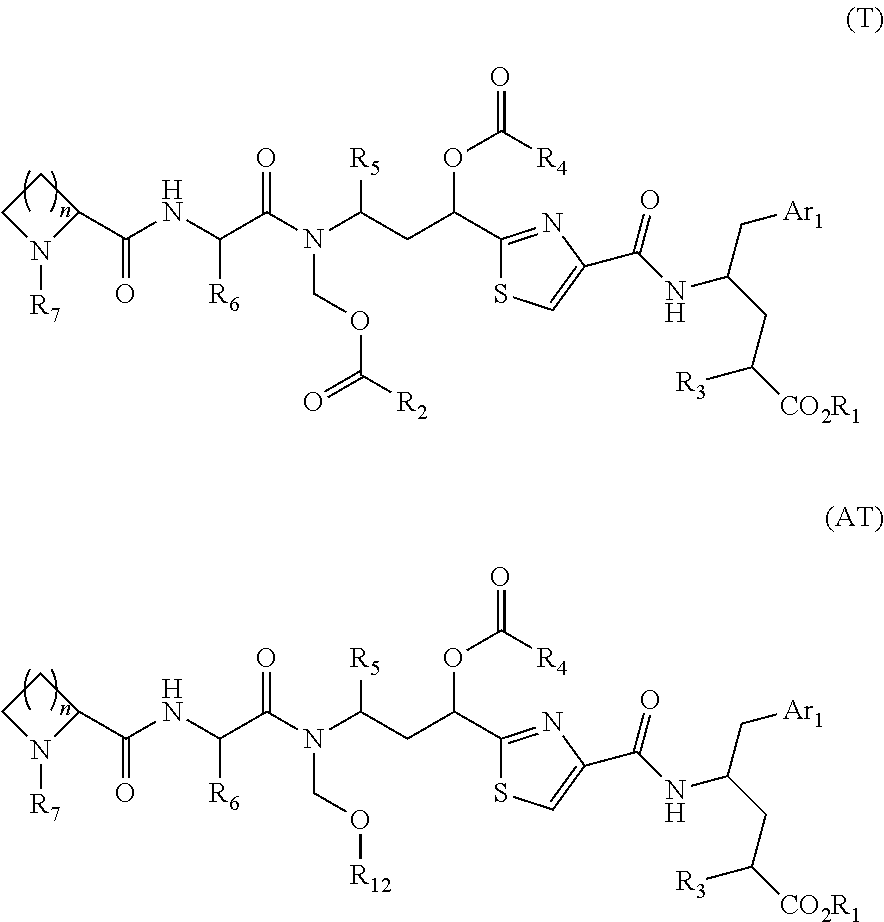
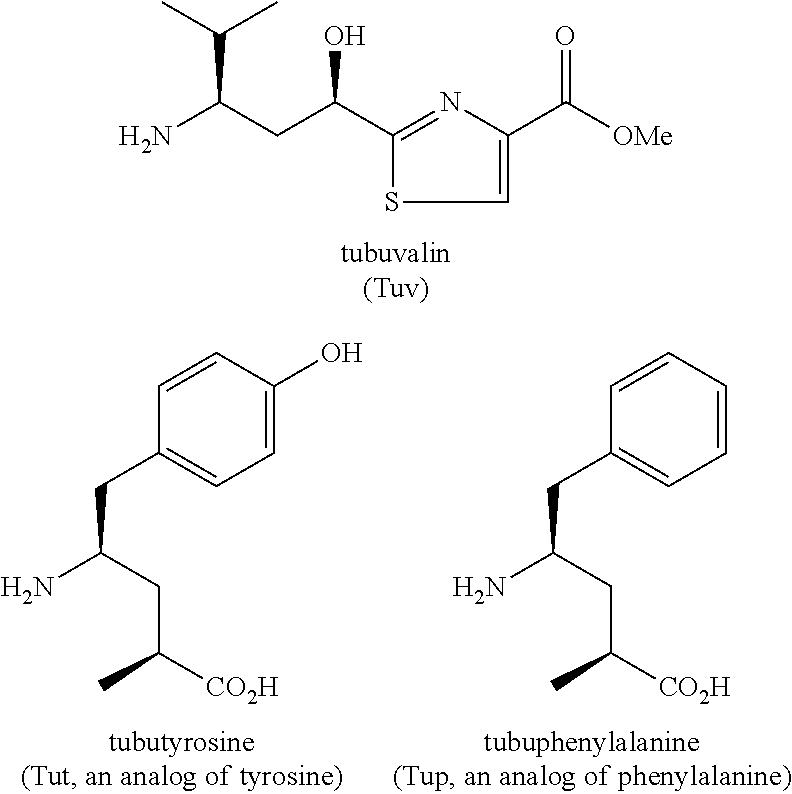
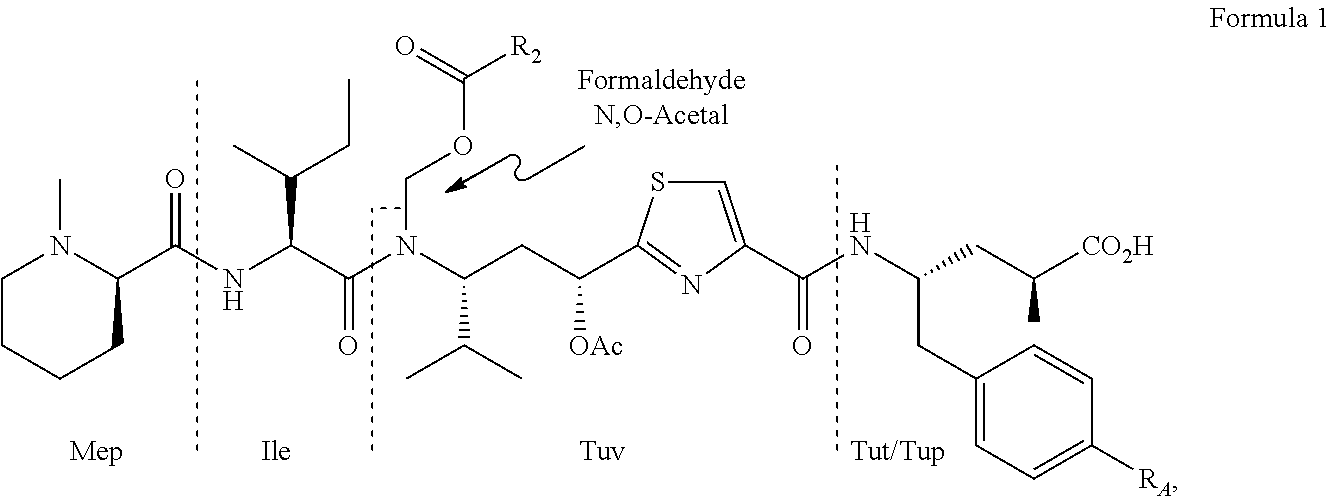
Processes for preparing tubulysins
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
examples
[0213]
Example
Synthesis of Dipeptide 3
[0214]4.9 g of dipeptide 1 (11.6 mmol) was dissolved in 60 mL dichloromethane, imidazole (0.87 g, 12.7 mmol) was added to the resulting solution at 0° C. The reaction mixture was warmed slightly to dissolve all solids and re-cooled to 0° C. TESCl (2.02 mL, 12.1 mmol) was added drop-wise at 0° C., the reaction mixture was stirred under argon and warmed to room temperature over 2 h. TLC (3:1 hexanes / EtOAc) showed complete conversion. The reaction was filtered to remove the imidazole HCl salt, extracted with de-ionized water, and the aqueous phase was back-washed with dichloromethane, the combined organic phase was washed with brine, dried over Na2SO4, filtered to remove the Na2SO4, concentrated under reduced pressure, co-evaporated with toluene and dried under high-vacuum overnight to give 6.4 g of crude product 2 (vs 5.9 g of theoretical yield).
[0215]The crude product 2 was co-evaporated with toluene again and used without further purification. TE...
example
Synthesis of EC1662 (Scheme 3)
[0246]Step 1: Anhydrous DCM (5.0 mL) was added to a mixture of EC1623 (II: R=n-pentyl. 114 mg), pentafluorophenol (PFP, 67.3 mg), and DCC-resin (2.3 mmol / g, 396 mg) and the suspension was stirred at ambient temperature under argon for 23 hours. The resin was filtered off and washed with anhydrous DCM (3.0 mL) and the combined filtrates were concentrated under reduced pressure to give a residue, which was vacuumed at ambient temperature for 1 hour prior to use in Step 3.
[0247]Step 2: EC1426 (114 mg) was dissolved in anhydrous DCM (1.5 mL) and to which was added TFA (0.50 mL). The resulting solution was stirred at ambient temperature under argon for 70 minutes and concentrated under reduced pressure to give a residue, which was co-evaporated with anhydrous DCM (2.0 mL×3) and vacuumed at ambient temperature for 9 hours prior to use in Step 3.
[0248]Step 3: The residue from Step 1 was dissolved in anhydrous DCM (1.5 mL) and to this solution was added DIPEA (...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Temperature | aaaaa | aaaaa |
| Temperature | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More