Purification of Biological Molecules
a biological and molecule technology, applied in the field of biological molecule purification, can solve the problems of large vessel size, complex purification process, and high cost, and achieve the effect of reducing the number of vessels and reducing the number of purification steps
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Process for Purifying a Monoclonal Antibody
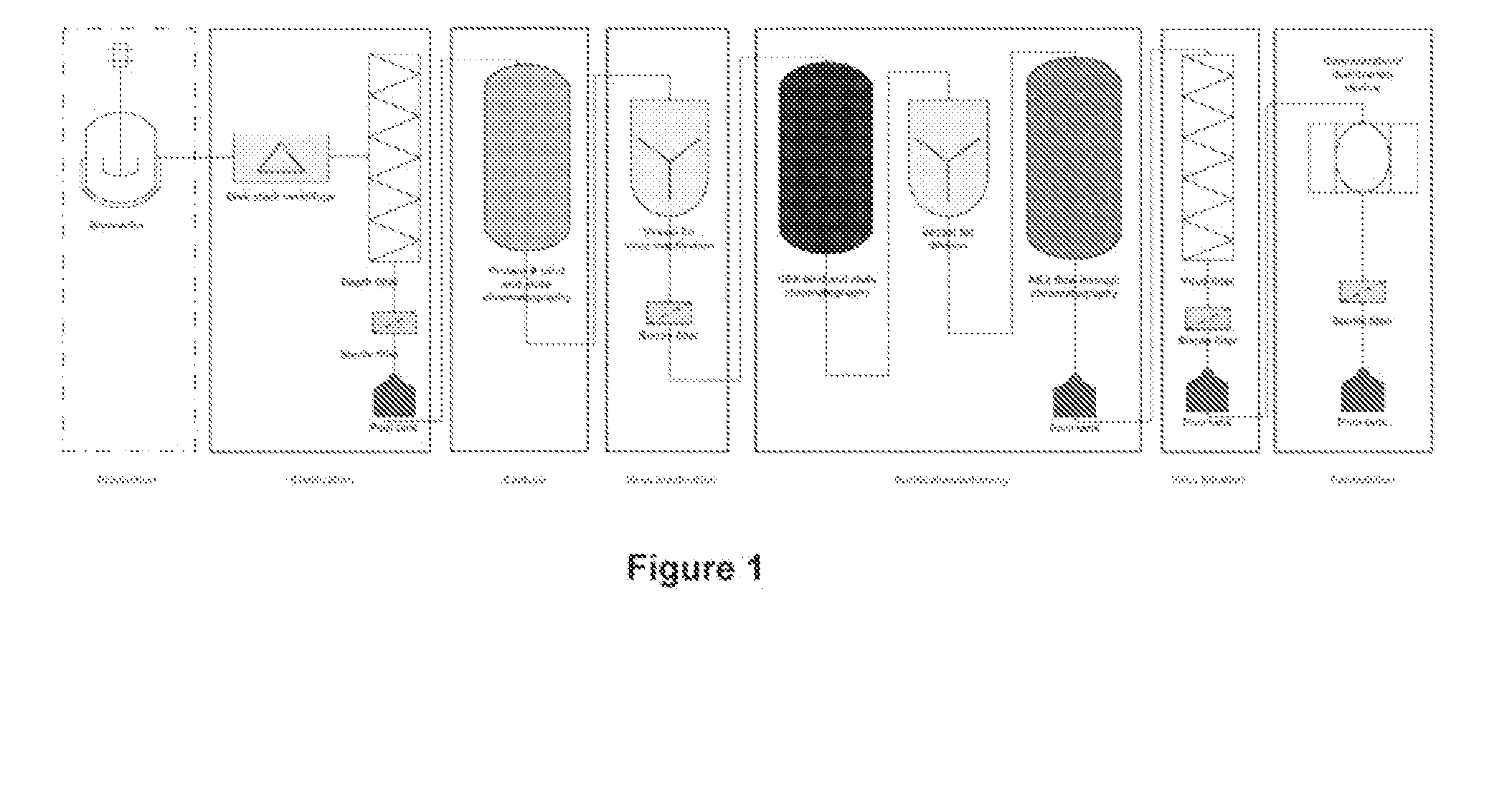
[0288]In this representative example, the purification of a monoclocal antibody is achieved using a purification process in a continuous manner, where various unit operations are connected in a manner to operate continuously. An exemplary process is depleted in FIG. 2.
[0289]The representative example described below includes the following steps performed in the sequence listed: clarification using depth filtration; use of one or more in-line static mixers to change solution conditions: Protein A bind and elute chromatography using continuous multicolumn chromatography which employs two separation units; pH adjustment of the output using, one or more static mixers; and flow-through purification which employs depth filtration followed by activated carbon followed by anion exchange chromatography followed by pH adjustment using a static mixer followed by cation exchange chromatography followed by virus filtration.
[0290]In this example a CHO-ba...
example 2
Process for Purifying a Monoclonal Antibody
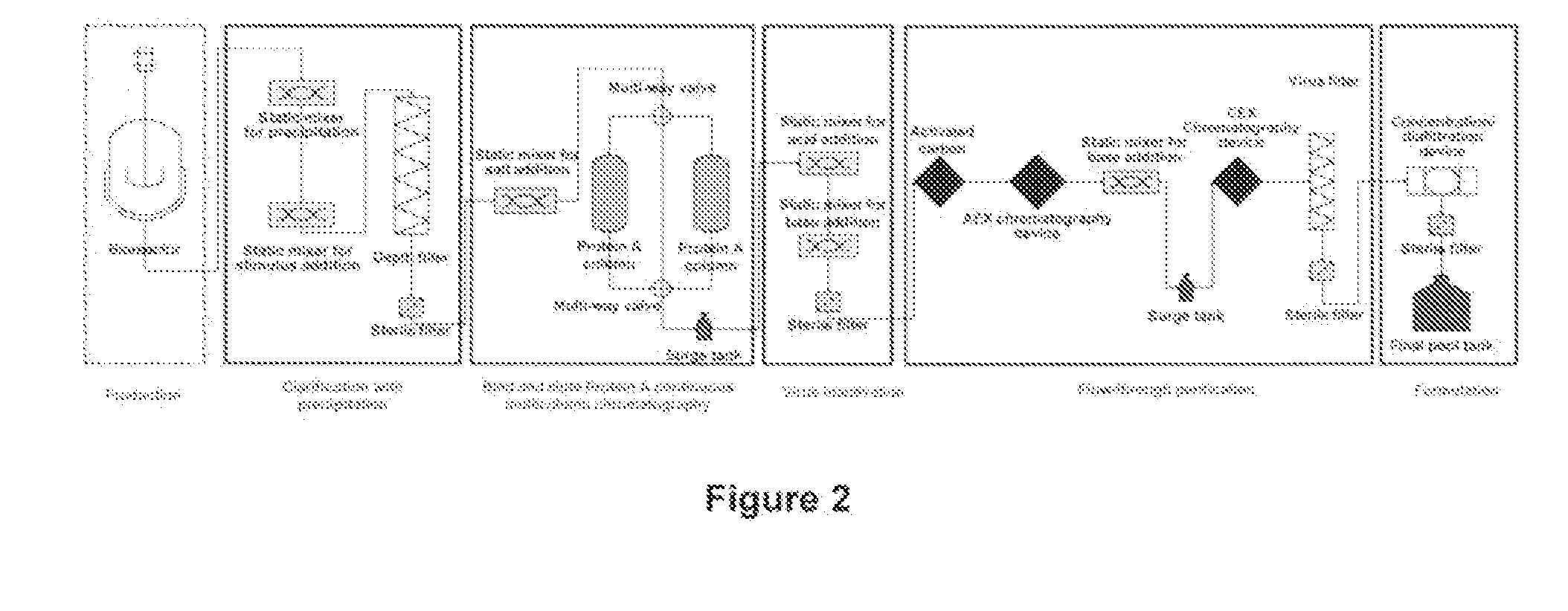
[0295]In this representative example, the purification of a monoclocal antibody is achieved using a purification process, where various unit operations are connected in the sequence described below.
[0296]The representative example described below includes the following steps performed in the sequence listed: clarification using stimulus responsive polymer following centrifugation; contacting the supernatant with salt; Protein A bind and elute chromatography using continuous multicolumn chromatography which employs two separation units; pH adjustment of the output using one or more static mixers; and flow-through purification which employs depth filtration followed by activated carbon followed by anion exchange chromatography followed by pH adjustment using a static mixer followed by cation exchange chromatography followed by virus filtration.
[0297]In this example, a CHO-based monoclonal antibody (MAb05) is produced in a fed-batch bioreactor...
example 3
Flow-Through Purification Process Following Batch Protein A Chromatography
[0307]In this representative experiment, a monoclonal antibody solution previously purified by batch protein A is further purified using flow-through purification to meet final purity and yield targets. This is done by performing the following steps in a flow-through manner: activated carbon; anion exchange chromatography; in-line pH change; cation exchange chromatography and virus filtration.
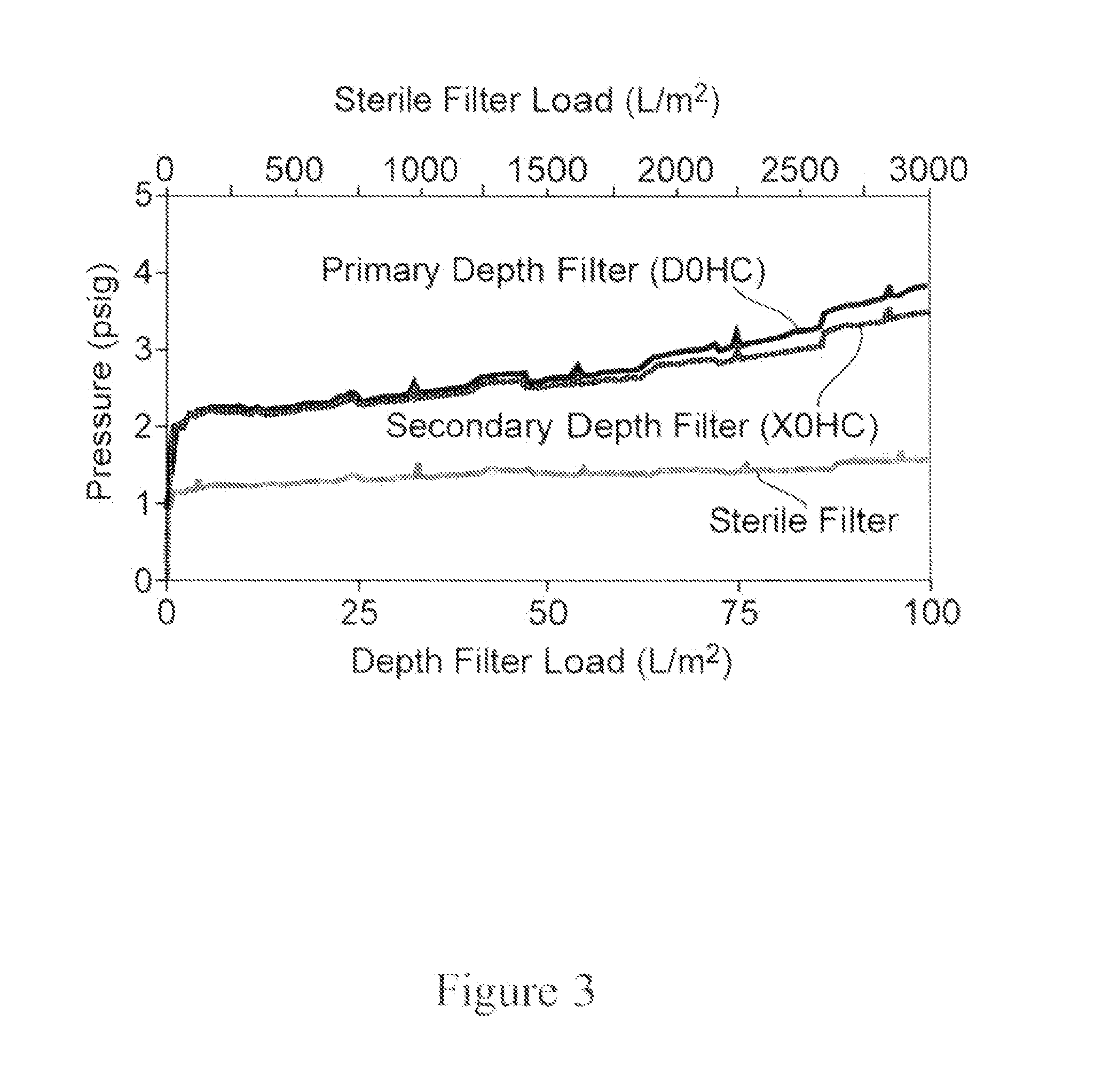
[0308]The set-up, equilibration and run is similar to Example 2 except for some minor modifications. The starting material is a protein A eluate from a batch protein A process. Specifically, the MAb feed processed for this run is 102 mL of 135 mg / mL MAb05 at a flow rate of 0.6 mL / min. A depth filter is not used in this study as the feed is filtered through a sterile 0.22 μm filter prior to performing the flow-through purification. A 2.5 mL activated carbon column is used which corresponds to a loading of 0.55 kg / L. Two an...
PUM
| Property | Measurement | Unit |
|---|---|---|
| time | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More