Crystalline polymorphs of a muscarinic acetylcholine receptor agonist
a technology of muscarinic acetylcholine and crystalline polymorphs, which is applied in the field of crystalline polymorphs of muscarinic acetylcholine receptor agonists, can solve the problems of unacceptable safety margins of xanomeline and other related muscarinic agonists
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
ne Form I Compound A
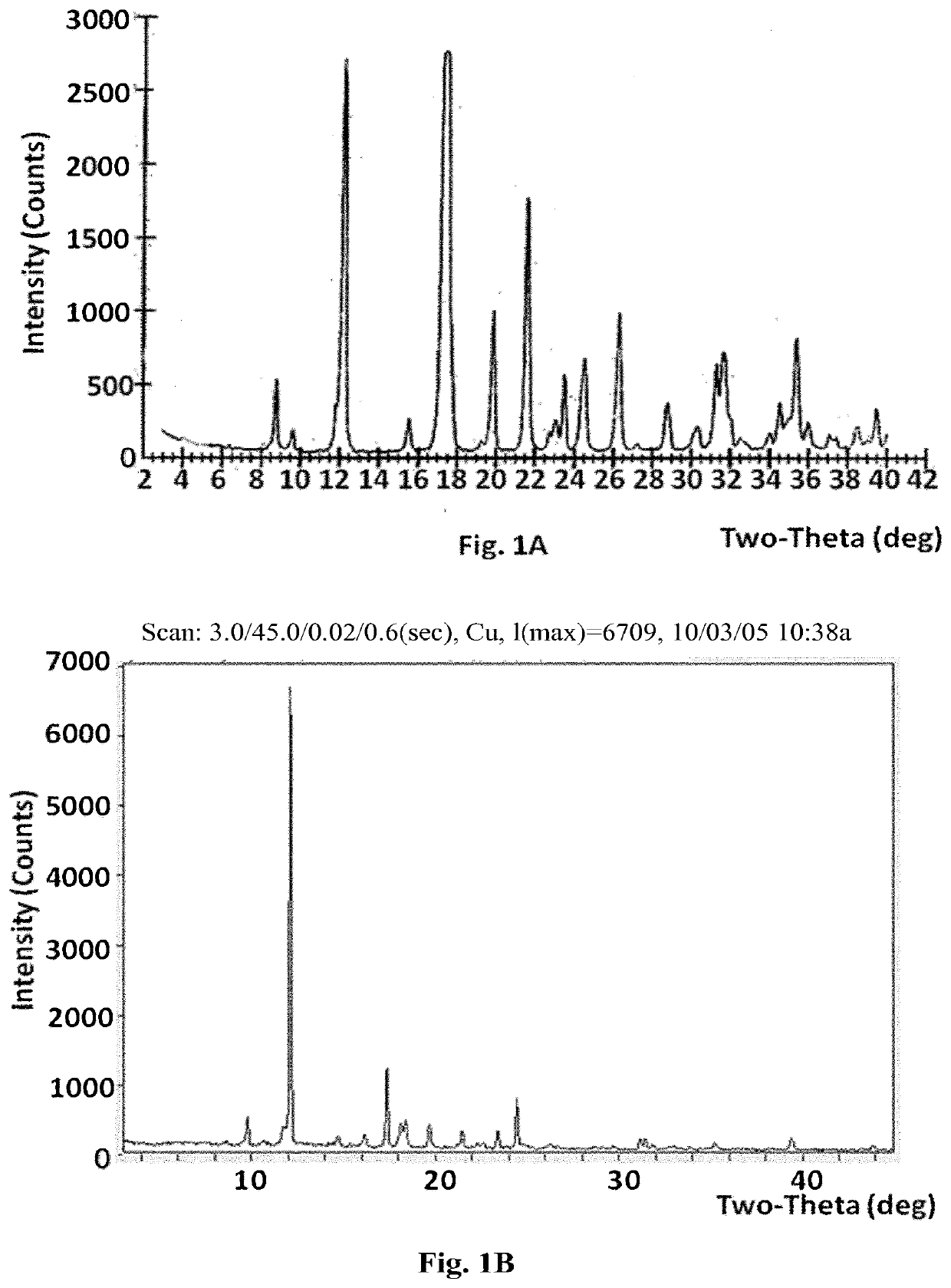
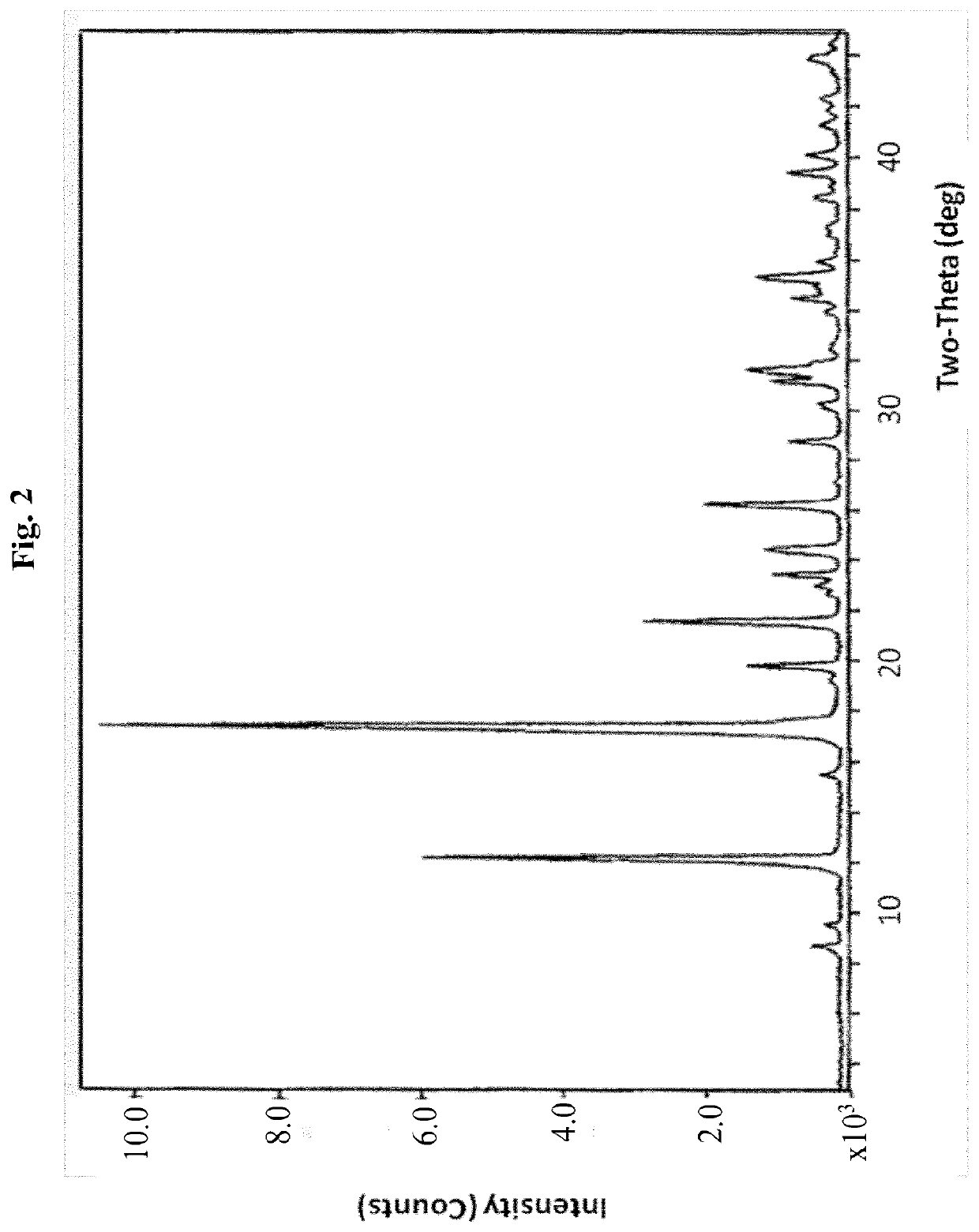
[0201]a) To Compound A (1.37 g) was added ethyl acetate (7 ml), and the mixture was heated to dissolve the compound. The clear solution was cooled at room temperature overnight. Solid material which appeared to the naked eye to be crystalline formed in the solution. This material was collected by filtration and dried at room temperature under vacuum to give 1.06 g of solid Compound A. X-ray powder diffraction, which yielded the following 2-theta values, d-spacings and relative intensities, confirmed that the material was crystalline (FIG. 1):
2 θ (±0.1)d (Å, unrounded)I / I0 8.810.069 5.3 9.69.1821.812.37.22451.915.65.6992.217.55.067100.019.34.6110.619.94.4649.321.64.11016.022.73.9081.123.03.8611.823.53.7835.124.53.6275.826.33.3859.127.23.2750.228.83.1013.030.32.9481.131.32.8585.431.62.8265.932.52.7560.634.02.6360.834.52.5983.035.42.5377.036.02.4951.837.12.4260.737.42.4030.638.52.3371.439.02.3100.439.42.2832.6
[0202]Solid-state CP / MAS 13C NMR of the crystalline mater...
example 2
ne Form I Compound A
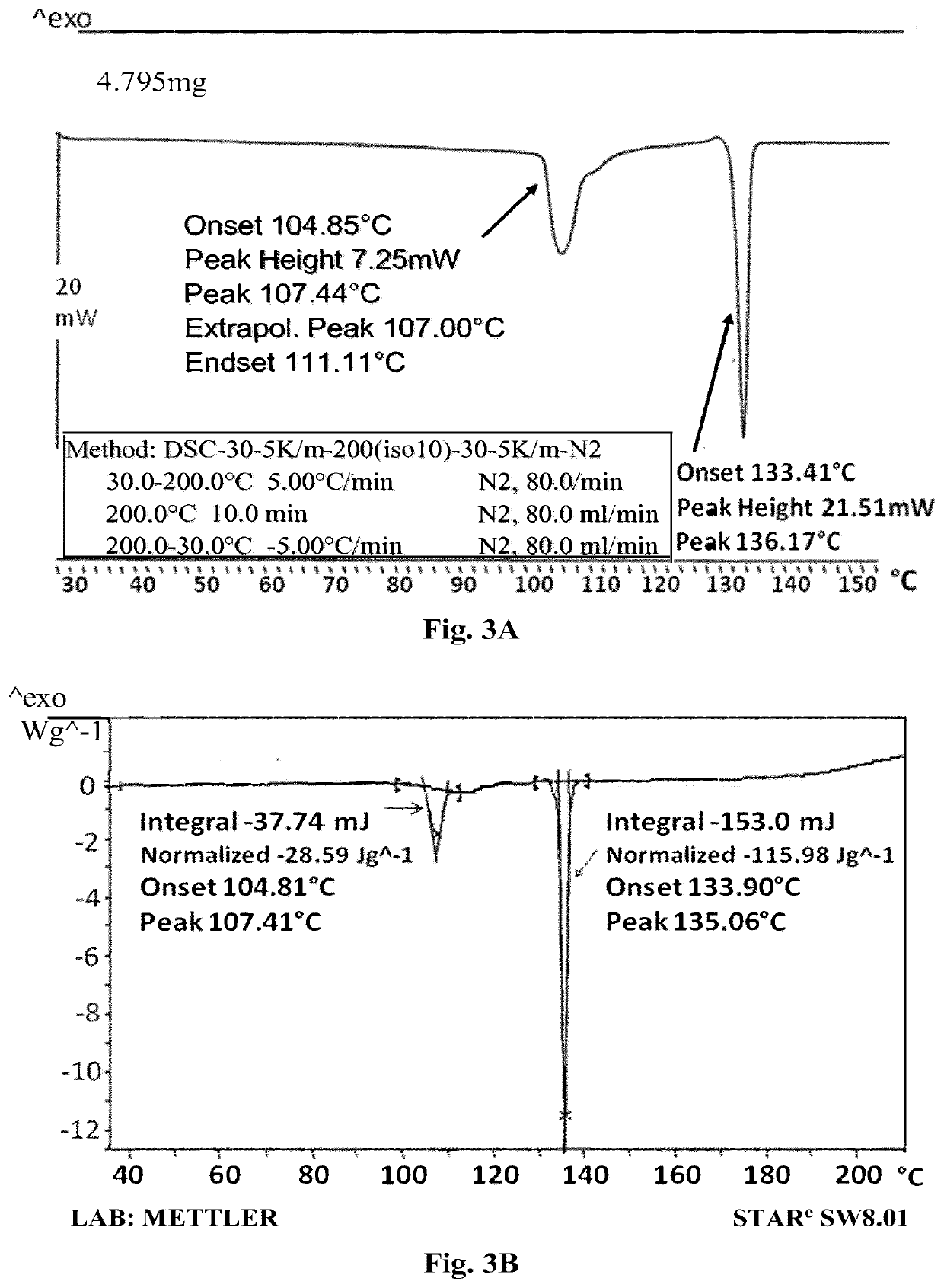
[0206]a) To Compound A (1.07 gr.) was added water (25 ml), and the mixture was heated to dissolve the compound. The clear solution was cooled at room temperature overnight and the precipitated crystals were collected by filtration. The resulted crystals were dried to give 0.82 gr. of Compound A. m.p. 134.7-135.2° C.
[0207]x-ray powder diffraction (XRPD): [2 Th, d (A), I / I0] (8.8, 10.052, 6), (9.6, 9.174, 4), (12.3, 7.223, 73.9), (15.6, 5.696, 4.9), (17.3, 5.126, 81.3), (17.5, 5.066, 100), (19.3, 4.6, 1.8), (19.9, 4.461, 20.3), (21.6, 4.106, 39.8), (23.1, 3.857, 5.6), (23.5, 3.782, 6.3), (24.5, 3.632, 12.2), (26.3, 3.388, 20.9), (27.2, 3.276, 0.5), (28.8, 3.097, 8.4), (30.3, 2.945, 3.6), (31.2, 2.865, 9.8), (31.6, 2.83, 14.1), (32.5, 2.751, 2.6), (34, 2.634, 3.3), (34.5, 2.597, 5.9), (35, 2.564, 4.8), (35.4, 2.533, 9.3), (36, 2.493, 5.9), (37.4, 2.405, 2.4), (38.5, 2.338, 4.1), (39.4, 2.285, 3.8).
[0208]Solid-state CP / MAS 13C NMR chemical shifts (δc in ppm) 13.289, ...
example 3
ne Form I Compound A
[0213]To Compound A (1.0 g) was added isopropanol (3.5 ml), and the mixture was heated to dissolve the compound. The clear solution was cooled at room temperature overnight. Solid material which appeared to the naked eye to be crystalline formed in the solution. This material was collected by filtration and dried at room temperature under vacuum to give 0.5 g of solid Compound A.
[0214]Solid-state CP / MAS 13C NMR of the crystalline material yielded a spectrum having the following chemical shifts (δc in ppm, unrounded): 13.423, 30.301, 43.729, 41.122, 52.289, 54.237, 46.739, 67.222, 174.457.
[0215]ATR-FT-IR yielded a spectrum having the following absorption peaks (cm−1, unrounded): 725, 775, 809, 827, 890, 945, 985, 1012, 1027, 1071, 1111, 1144, 1195, 1278, 1292, 1353, 1371, 1389, 1427, 1438, 1468, 1672, 2845, 3021, 3158, 3427.
[0216]This form is defined as Form I because of characteristic peaks in CP / MAS 13C NMR & ATR-FT-IR.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More