Morphine quantitative and micro detection method
A detection method, morphine technology, applied in the field of quantitative and trace detection of morphine, to achieve the effect of less sample consumption, convenient detection and short time
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0019] 1. Take 90mg of AgNO 3 Dissolved in 500mL of water to prepare AgNO with a mass percentage of 0.018% 3 Aqueous solution, when the aqueous solution was heated to 90°C, 10mL of trisodium citrate aqueous solution with a mass percentage of 1% was added dropwise, and stirred for 60min at the same time to obtain a green-yellow silver colloid.
[0020] 2. Put an appropriate amount of silver colloid into a centrifuge tube and centrifuge at high speed for 10 minutes, and remove the layer of high-concentration silver colloid for later use.
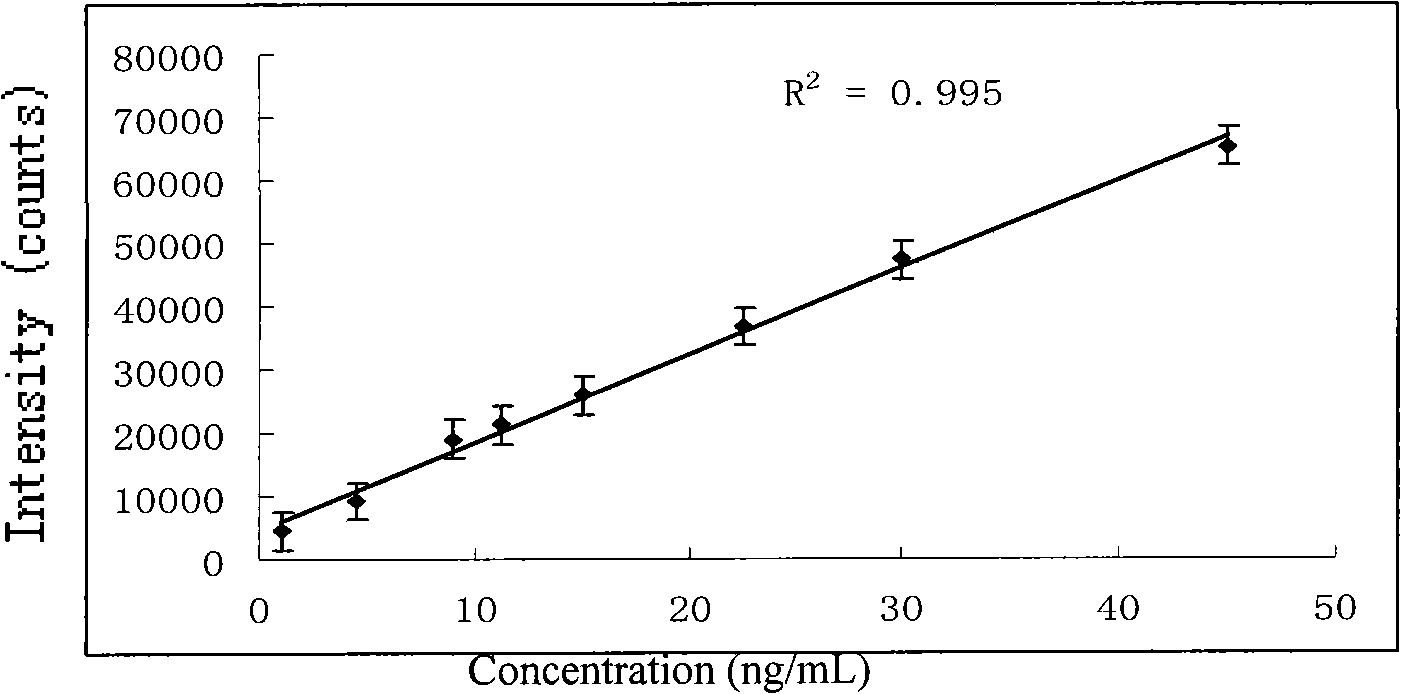
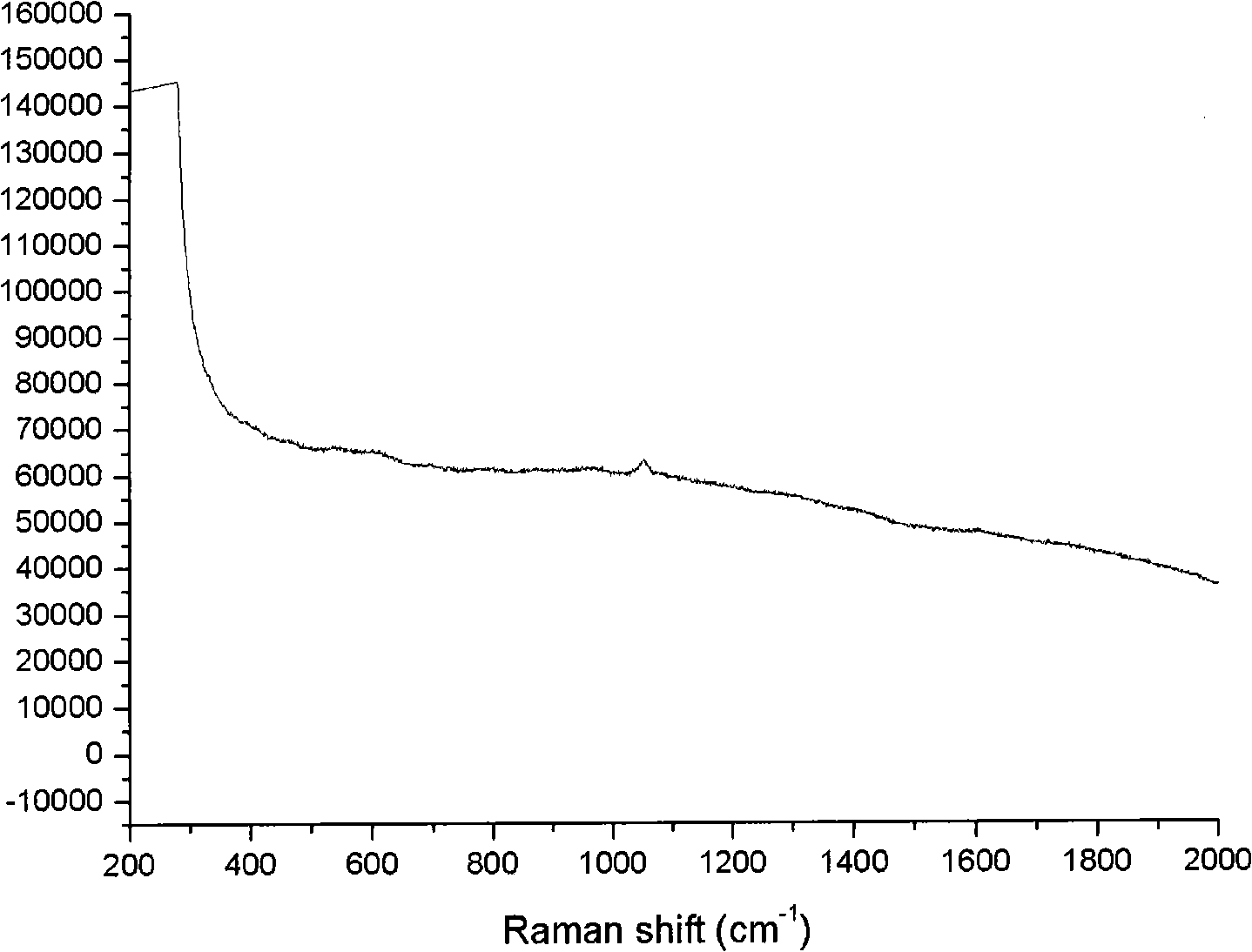
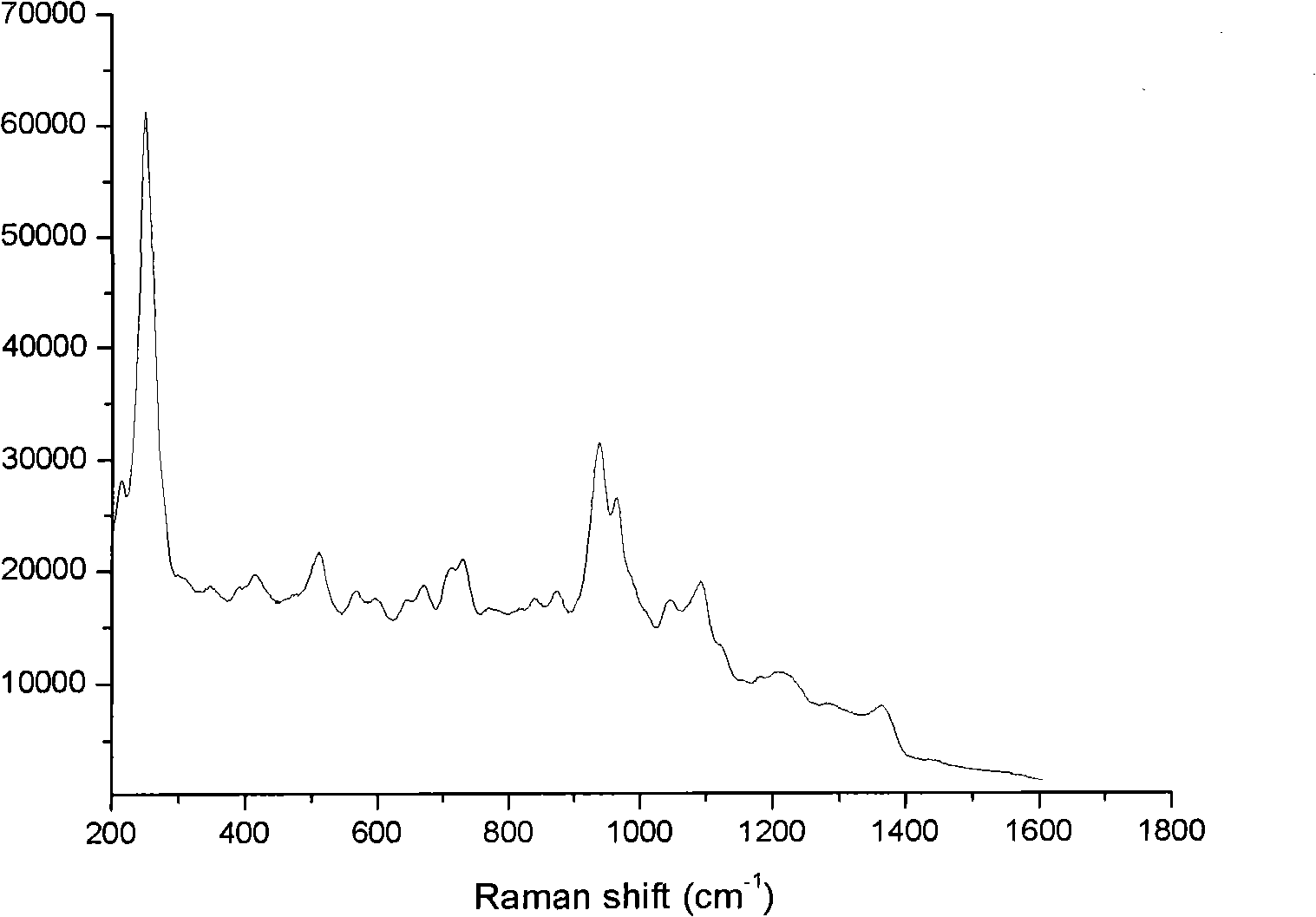
[0021] 3. Weigh 0.9mg of morphine, add it into a 1000mL volumetric flask, dilute to 1000mL with ultrapure water, and prepare a 900ng / mL morphine aqueous solution. Measure 5mL of the above-mentioned morphine solution and mix it with 5mL of high-concentration silver colloid. image 3 The morphine-silver colloidal SERS spectrum shown. After the morphine solution is mixed with the silver colloid, the obvious colloidal aggregation phenomenon in ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - Generate Ideas
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com