Quality control method for Chinese medicinal preparation Qinggong Shoutao pills
A technology of traditional Chinese medicine preparations and detection methods, which is applied in measuring devices, instruments, scientific instruments and other directions to achieve the effect of improving quality control
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0034] The following examples are used to further illustrate the present invention. The following examples are illustrative and not restrictive. The following examples cannot be used to limit the protection scope of the present invention.
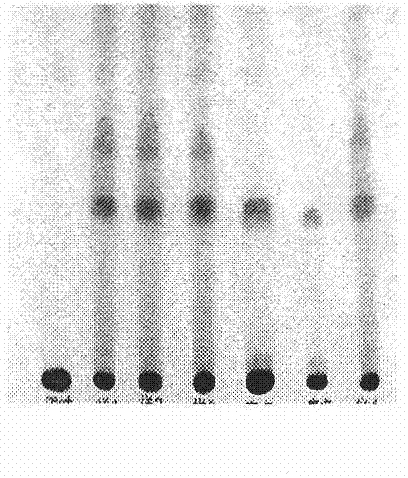
[0035] The quality control method of traditional Chinese medicine preparation Qinggongshoutao Pills, the method steps are:
[0036] (1) Microscopic identification:
[0037] Observed under a microscope: the seed coat stone cells are light yellow to yellow, thick, wavy and curved, and the cell cavity contains brown material (medium berry). The thick-walled cells of the endothelium are yellowish-brown or reddish-brown, with thick walls, non-lignified, and the cell cavity contains siliceous blocks (Yizhi). Seed coat palisade cells are in slices, yellow-brown or light reddish-brown, polygonal-like, closely arranged, long columnar in lateral view, 60~145μm in length (sour date kernel). The undigested mesophyll tissue fragments contain many small clust...
PUM
| Property | Measurement | Unit |
|---|---|---|
| diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More