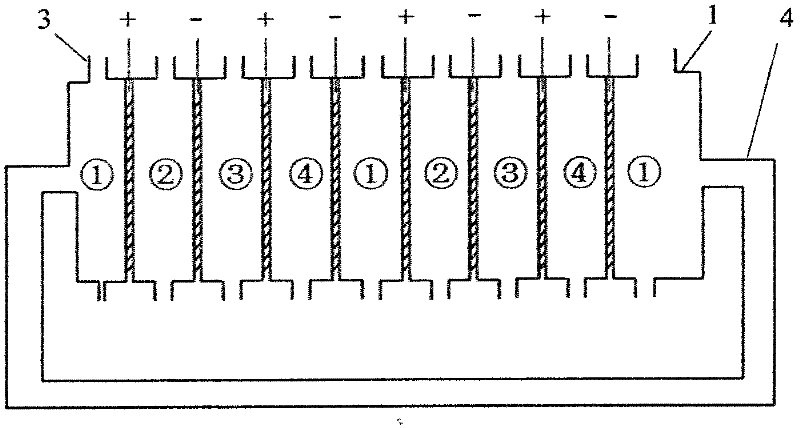
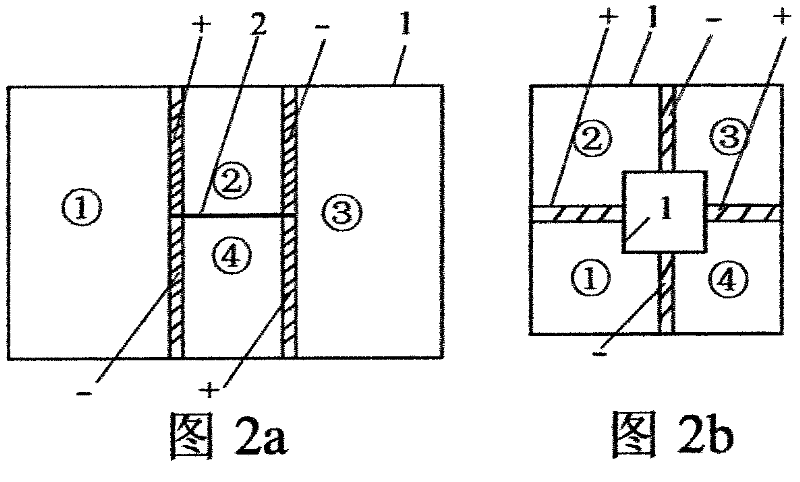
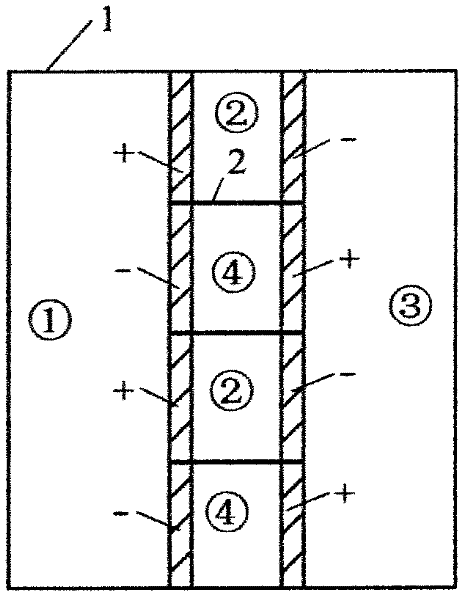
Method for realizing and controlling electrolyte metathesis reaction
A metathesis reaction, electrolyte technology, applied in the field of chemistry, can solve the problem of not being able to carry out metathesis reaction
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 2
[0131] Example 2, utilizing gypsum, that is, calcium sulfate containing two waters of crystallization and water to carry out metathesis reaction to produce sulfuric acid. Since calcium sulfate is slightly soluble in water, if it undergoes metathesis reaction with water directly, its chemical reaction rate is very slow, and the concentration of sulfuric acid solution produced is low. Therefore, sulfuric acid can be produced by indirect production method using medium medium. The selected medium is strong acid, strong alkali, strong salt, etc.
[0132] If the medium is a strong acid, such as hydrochloric acid, the chemical reaction equation is as follows:
[0133]
[0134] If the medium is a strong base, such as sodium hydroxide, the chemical reaction equation is as follows:
[0135]
[0136] If the medium is strong salt, such as using gypsum, salt, and water to produce sulfuric acid, hydrochloric acid, and caustic soda through metathesis reactions, the preparation method...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com