Processes and compositions for the preparation of noribogaine from voacangine
A technology of voacangine and covalent binding, applied in organic chemistry and other directions, can solve the problem of incorrect chemical formula of ibogaine
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
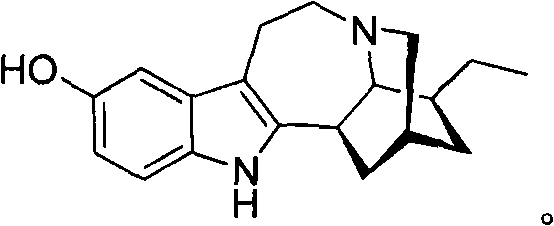
[0061] Preparation and Purification of Noribogaine
[0062] Voacangine (methyl 12-methoxyibogamine-18-carboxylate) is an alkaloid found mainly in the root bark of the African Voacanga africana tree and in other plants such as ibogamia, African horseshoe Found in Tabernaemontana africana, Trachelospermum jasminoides and Ervatamia yunnanensis. Voacangine has previously been used as a semi-synthetic precursor for ibogaine (see US Patent No. 2,813,873).
[0063] This application covers a process for the preparation of noribogaine from voacangine without providing ibogaine as an intermediate. The method described is useful for a number of reasons. First, known methods for the preparation of noribogaine involve demethylation of ibogaine as a final step. This is unlikely to provide pure noribogaine, and since ibogaine is a Class 1 controlled substance and is known to induce severe hallucinations, ibogaine contamination is not expected. Second, ibogaine is isolated from the root of...
example
[0092] In the following examples, the following abbreviations have the following meanings. Where an abbreviation is not defined, it has its broadly accepted meaning.
example 1
[0093] Example 1 - Synthesis of Noribogaine from Voacangine
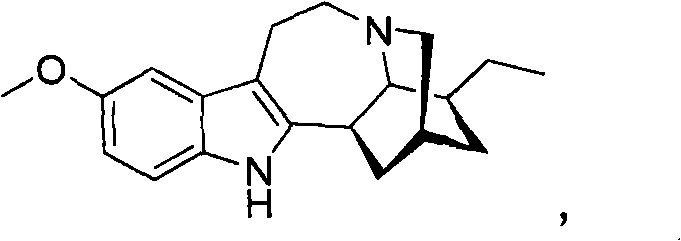
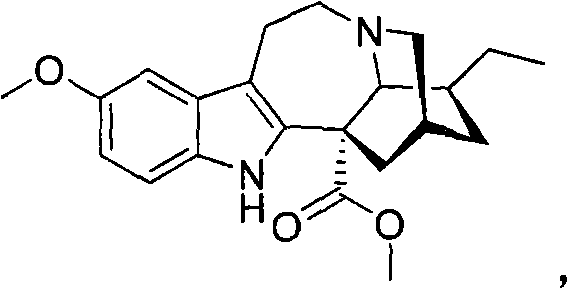
[0094] Example 1 illustrates a method for the synthesis and purification of noribogaine from voacangine, which follows Scheme 5 below.
[0095] Option 5
[0096]
[0097] Voacangine 1 can be taken up in dichloromethane / ethylene glycol solution and cooled to 0°C to -10°C (ice-salt bath). An excess (1-3 molar equivalents) of the appropriate Lewis acid (boron trichloride, boron tribromide or aluminum trichloride) is added in portions. The resulting mixture was stirred at 25°C to 50°C for 2 hours to 24 hours until sufficiently complete by TLC. The reaction mixture can then be diluted with fresh dichloromethane and washed with saturated NaHCO 3 The solution was washed, dried and evaporated under reduced pressure to provide the corresponding methyl 12-hydroxyibogamine-18-carboxylate 2, which could then be purified by silica gel column chromatography using a gradient of hexane and ethyl acetate or without Purificati...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com