Conveniently implantable sustained release drug compositions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
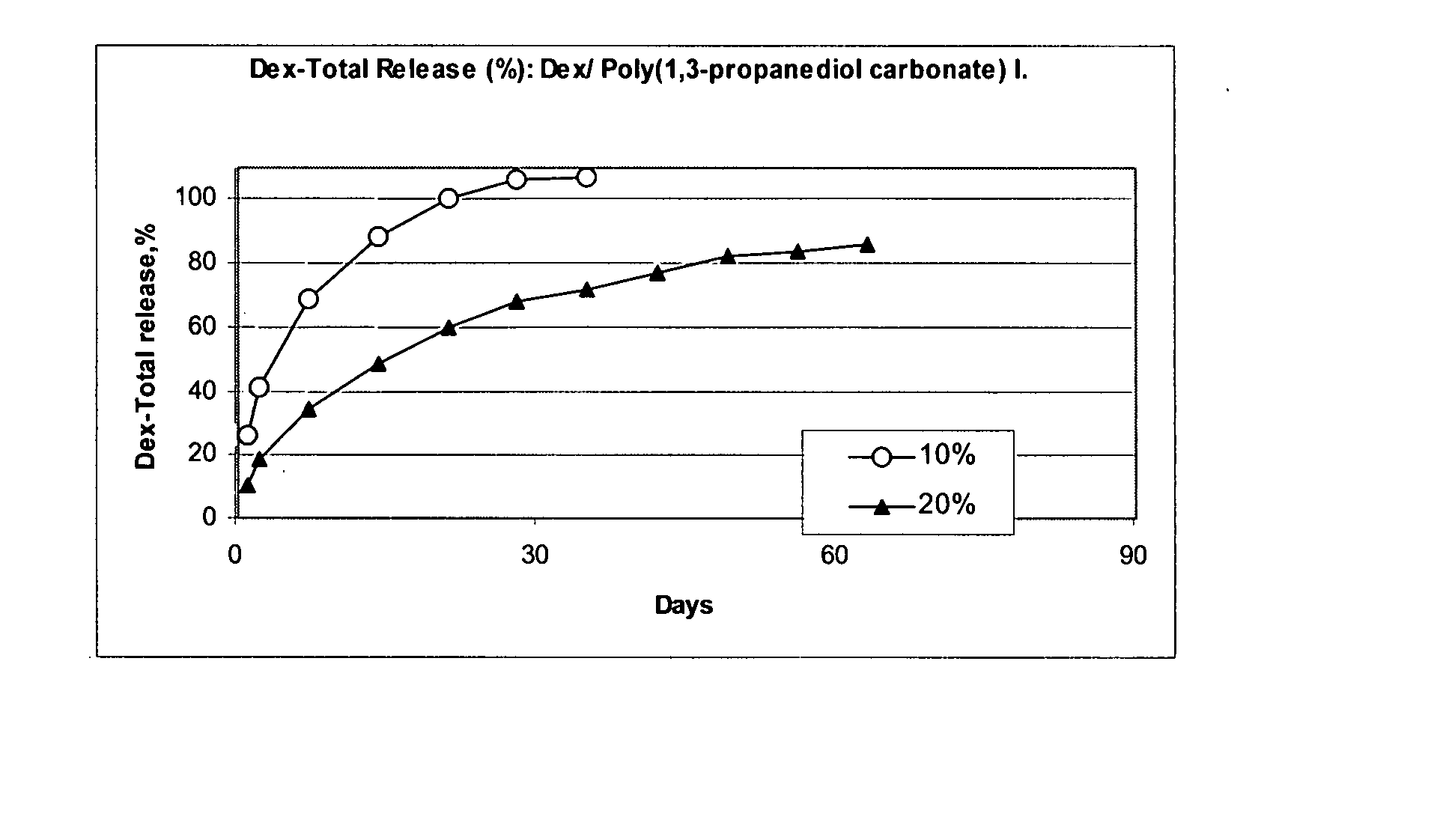
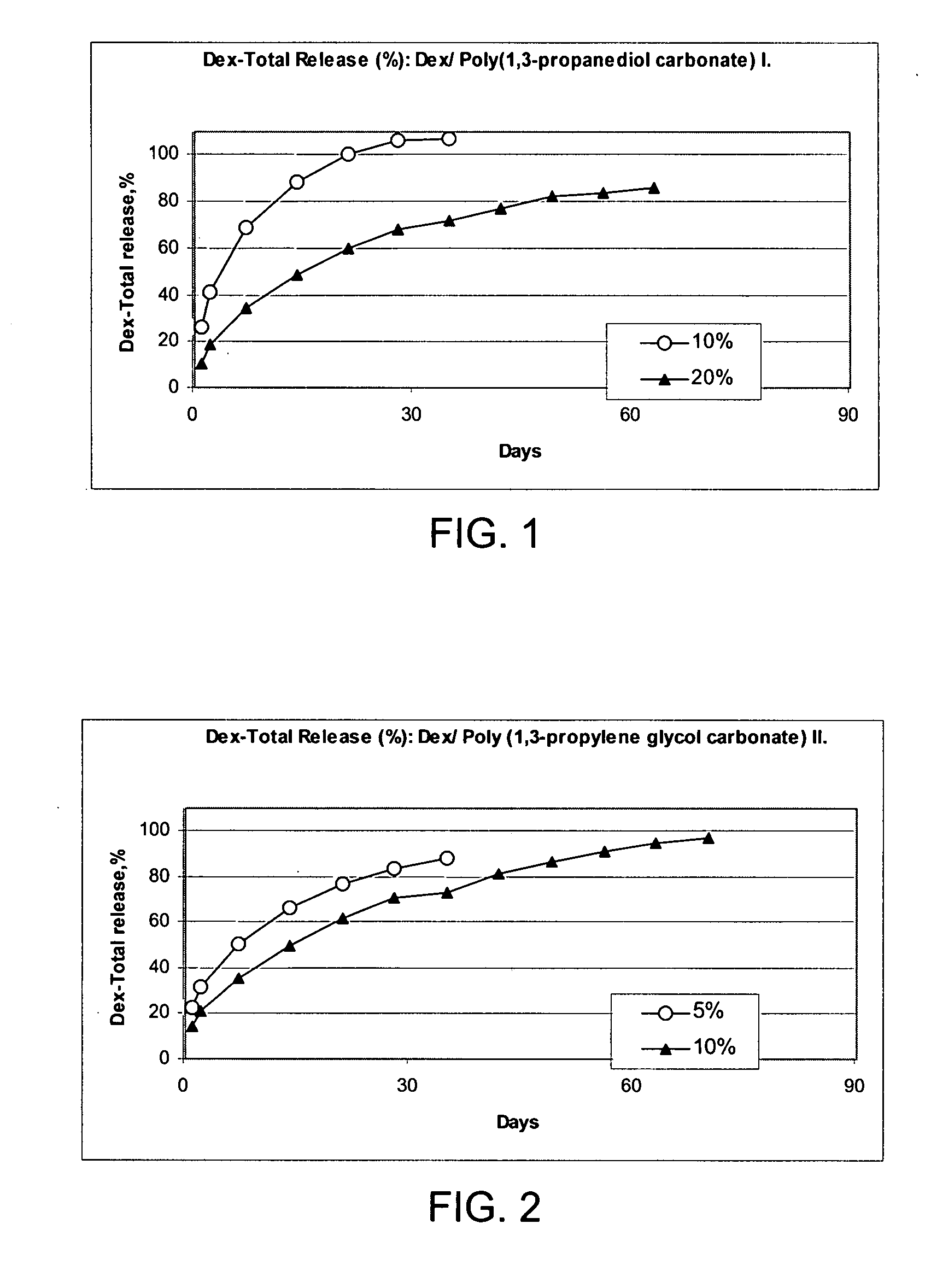
Preparation of a Poly(1,3-propanediol carbonate) I from 1,3-propanediol at 65° C. and 96 hours
[0240] To 23.6 g (0.2 mole) diethyl carbonate (b.p. 128° C.) was added 15.2 g (0.2 mole) 1,3-propanediol containing 0.05 g (1.25 mmole) of metallic Na to give two liquid phases. These reactants were placed in an open container in a 65° C. oven and were shaken occasionally. After 12 hours, the reactants were a homogeneous solution weighing 38.0 g. The theoretical weight for the loss of 0.4 moles (18.4 g) of ethanol in a complete reaction would be 20.4 g. The heating and occasional shaking were continued to give 27.0 g at 24 hours, 23.2 g at 48 hours, 21.4 g at 72 hours, and 17.4 g at 96 hours. The product oil was washed with 15 ml 5% aqueous acetic acid to two phases. The top phase was the water soluble phase. The 10.5 ml bottom phase was washed with 15 ml water to give 7.5 ml of a poly(1,3-propylene glycol carbonate) oligomer as a water-insoluble oil.
example 2
Preparation of Poly(1,3-propanediol carbonate) II from 1,3-propanediol at 110-150° C. and 26 hours.
[0241] A mixture of 76 g (1.0 mole) 1,3-propanediol containing 0.1 g metallic Na (2.5 mmole) and 118 g (1.0 mole) diethyl carbonate was heated at 110° C. As soon as the reactants reached 60° C. they formed a homogeneous solution. After heating 8 hours, the reactants had lost 48 g (52% of theoretical amount of ethanol). The temperature was then raised to 150° C. After 10 hours, the reactants lost another 46 g. A drop of this product completely dissolved in water. The resultant 97 g of oil was mixed with 6 g (0.05 moles) diethyl carbonate and the resultant solution was heated with occasional stirring at 150° C.
[0242] After 8 hours, the resultant syrup was found to be partially insoluble in water. The product was washed with 100 ml 5% aqueous acetic acid followed by four washings with 100 ml portions of water to give 46.1 g slightly yellow viscous oil (46.1 / 102=45% yield).
example 3
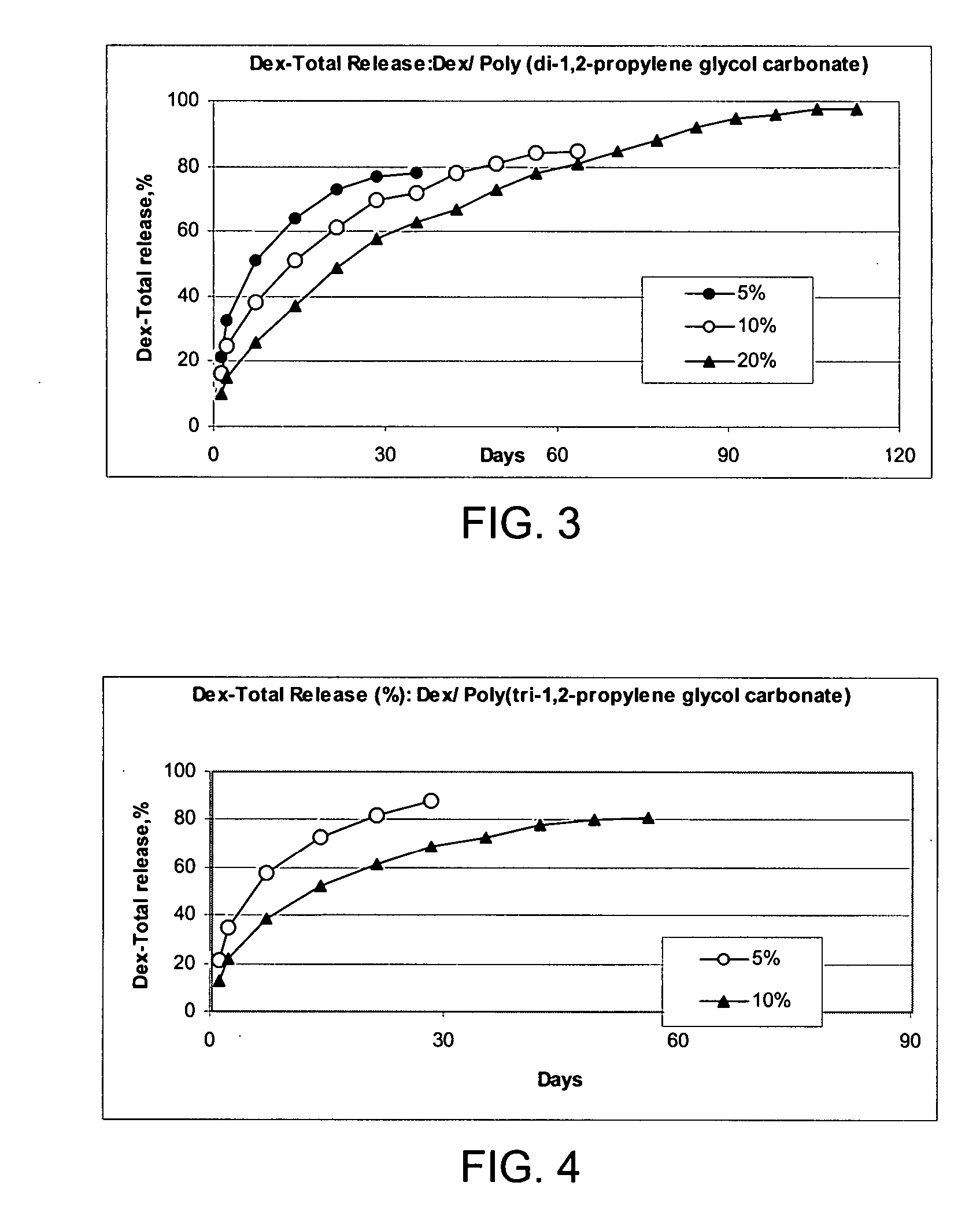
Preparation of a Poly(di-1,2-propylene glycol carbonate) from di-1,2-propylene glycol
[0243] To 590 g (0.5 moles) diethyl carbonate was added 67.0 g (0.5 moles) di-1,2-propylene glycol which had been reacted with 0.02 g Na to form a homogeneous solution. The reactants were placed in an open flask at 100° C. After 12 hours, the solution lost 23.4 g (about 50% of the theoretical 46 g ethanol). After another 15 hours at 150° C. the reactants had lost a total of 53.2 g to give a syrup that was partially insoluble in water. The product was washed with 100 ml 5% aqueous acetic acid followed by four washing with 100 ml portions of water to give 25.2 g colorless viscous, water insoluble liquid poly(di-1,2-propylene glycol carbonate) oligomer.
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More