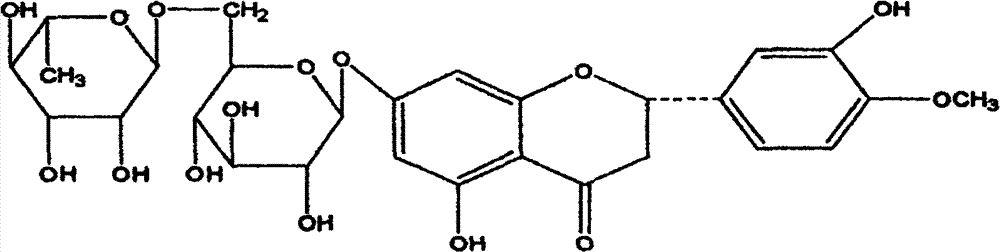
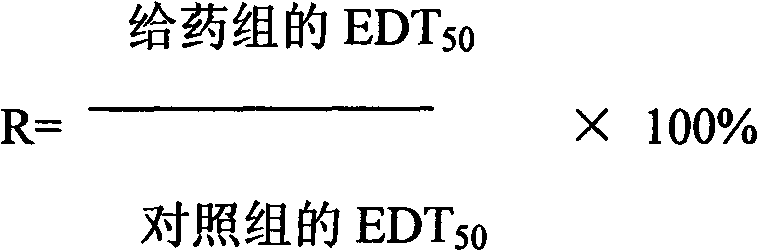Dihydromyricetin medicine treating acute and chronic bronchitis and its preparation method
A technology for dihydromyricetin and bronchitis, which is applied in the preparation of sugar derivatives, drug combinations, pharmaceutical formulations, etc., can solve the problems of lack of dihydromyricetin, inconvenience to carry, and high dosage of medicines, and achieves the treatment of acute and chronic diseases. Bronchitis, inhibit the deterioration of liver cells, safe and effective effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0015] Example 1. Antitussive pharmacological experiment of dihydromyricetin medicine
[0016] 1. Experimental animals: NIH mice, half male and half female, clean-grade standard, a total of 45 mice, sorted by body weight, selected healthy animals weighing 18-22 g, randomly divided into three groups, 15 mice in each group. Experimental groups such as negative control, positive control and dihydromyricetin drug samples were set up.
[0017] 2. Sample source and preparation:
[0018] (1) Dihydromyricetin sample group: an appropriate amount of dedihydromyricetin is fixed in a volumetric flask with normal saline, the concentration of dihydromyricetin is 2.0mg / ml, and the dihydromyricetin sample used is as described above Methods The dihydromyricetin monomer extracted from Chinese herbal medicines has a purity of over 95%.
[0019] (2) Methorphan: tablet, the content of methorphan is 15mg / tablet, take 2 tablets and dissolve in 20ml of normal saline to make a suspension, the concen...
Embodiment 2
[0033] Embodiment 2. The toxicology experiment of dihydromyricetin medicine
[0034] At a temperature of 24° C. and a humidity of 60±5%, 30 healthy clean-grade NIH mice were selected, half male and half female, weighing 18-22 g. The feed and water were disinfected, and the animals were raised under normal feed conditions before the test and during the observation period of the test.
[0035] Dihydromyricetin was dissolved in 0.5% Tween80 at a concentration of 900 mg / ml, and the liquid was orally administered to mice at a dosage of 0.2 ml / 20 g of mouse body weight. Observe 1, 4, 8, and 12 hours after administration, and observe every 12 hours thereafter. The death situation was observed, and the body weight change and other symptoms of the mice were recorded every day. On the 10th day, the mice were killed by neck dislocation, and the organs were taken for pathological examination.
[0036] On the 10th day, all the mice survived, and no toxic reaction was observed with 900 m...
Embodiment 3
[0037] Embodiment 3. Dihydromyricetin capsule preparation
[0038] Prepare gelatin capsules according to the following ingredient ratios
[0039] Dihydromyricetin 100
[0040] dry starch 40
[0041] Micronized silica gel 10
[0042] Mix the auxiliary materials and dihydromyricetin evenly, put them into transparent capsules, and get ready. Capacity: 150mg / capsule.
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap