Polypeptide with anti-bacterial function and preparation method and application thereof
A technology with multiple functions and antibacterial drugs, applied in the antibacterial field, can solve the problems of large molecular weight of natural antibacterial peptides, high purification or production costs, and easy interference of long-chain polypeptides, and achieve small molecular weight peptides, reduce bacterial drug resistance, and structure Stable and Simple Effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
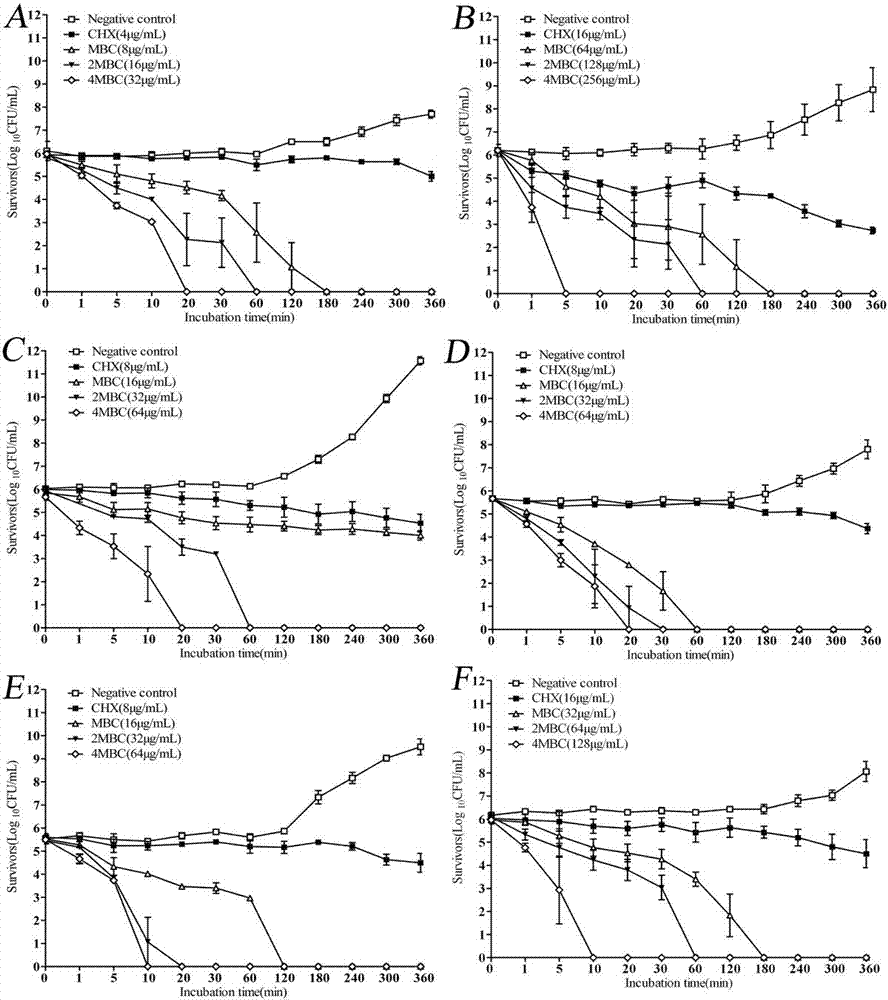
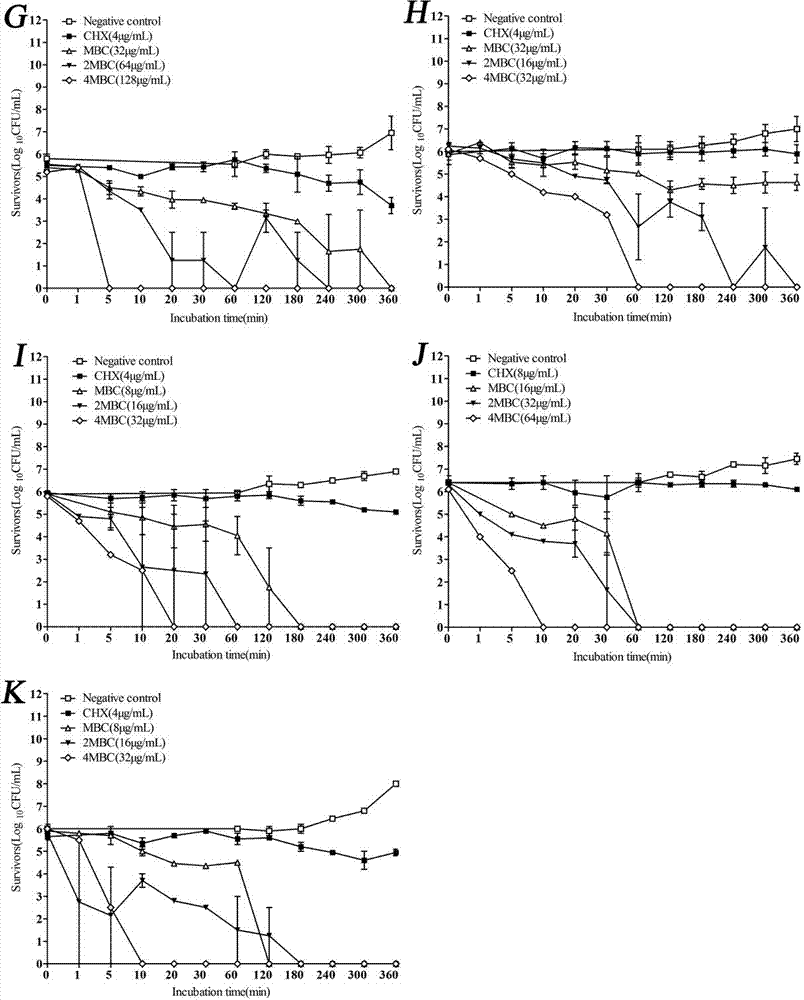
Examples
Embodiment 1
[0034] 1. Select Fmoc-His (Trt)-Wang Resin as the resin (carrier);
[0035] 2. Fully swell the resin with DCM;
[0036] 3. Use an appropriate concentration of DBLK (hexahydropyridine + DMF) to remove the Fmoc-protecting group;
[0037] 4. Wash several times with DMF to remove DBLK;
[0038] 5. Weigh the appropriate condensing agent and activator (HBTU, NMM) and the second Fmoc-protected amino acid (Fomc-Leu-OH) at the C-terminal for coupling;
[0039] 6. The ninhydrin detection method is used for detection to ensure that the connection is relatively complete;
[0040] 7. Wash several times with DMF to wash away the remaining residues and activator condensation agent;
[0041] 8. Carry out coupling according to the amino acid sequence of SEQ ID NO.1, the method refers to steps 3-7;
[0042] 9. After connecting all the amino acids, use steps 3 and 4 to remove the final Fmoc-protecting group;
[0043] 10. Cleavage with TFA cutting solution to remove resin and amino acid prot...
Embodiment 2
[0049] Circular dichroism analysis of embodiment 3 polypeptide
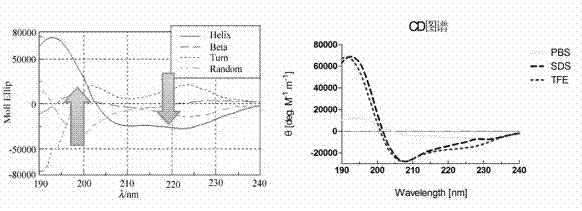
[0050] Circular dichroism (CD) spectroscopy is a fast, simple and accurate method to study protein conformation in dilute solution. This experiment is used to measure the structure of the polypeptide to verify the validity of its design principle. According to literature review, circular dichroism analysis shows that the α-helical structure has a positive band near the ultraviolet wavelength of 192nm, and two negative characteristic shoulder bands at 222 and 208nm. The appearance of the α-helical structure can be confirmed by observing the circular dichroism spectral bands.
Embodiment 3
[0051] laboratory apparatus
[0052] Jasco J-1500 CD Spectrometer (Japan)
[0053] Experimental procedure
[0054] Under the condition of 25°C, the light transmission length of the measurement container is 1mm, and the wavelength range of ultraviolet light is 190nm to 240nm. The GH12 samples prepared in Examples 1 and 2 were detected respectively, and each sample was scanned 10 times to obtain the average value. The sample peptide concentration is 0.2mg / ml, which are dissolved in solution A: 0.02M PBS solution; B: 0.02M PBS solution containing 25mM SDS; C: 0.02M PBS solution containing 50% TFE (v / v).
[0055] The obtained data is calculated by the formula of molar ellipticity draw a graph such as figure 1 shown.
[0056] It can be seen from the image curve that with the participation of surfactant (SDS) and common chemical solvents (TFE), the peptide spectrum presents a typical α-helix structure, indicating that the secondary structure of the polypeptide in solution is ...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap