Nucleic acid, kit and method for simultaneously detecting three viruses of Congo fever virus, Copo fever virus type 1 and 2
A technology of Kupo heat and Congo heat, applied in biochemical equipment and methods, microbiological determination/inspection, DNA/RNA fragments, etc., can solve problems such as laborious, high cost, and time-consuming, and achieve rapid screening and low cost Low, good detection effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0053] The design of embodiment 1 primer, probe
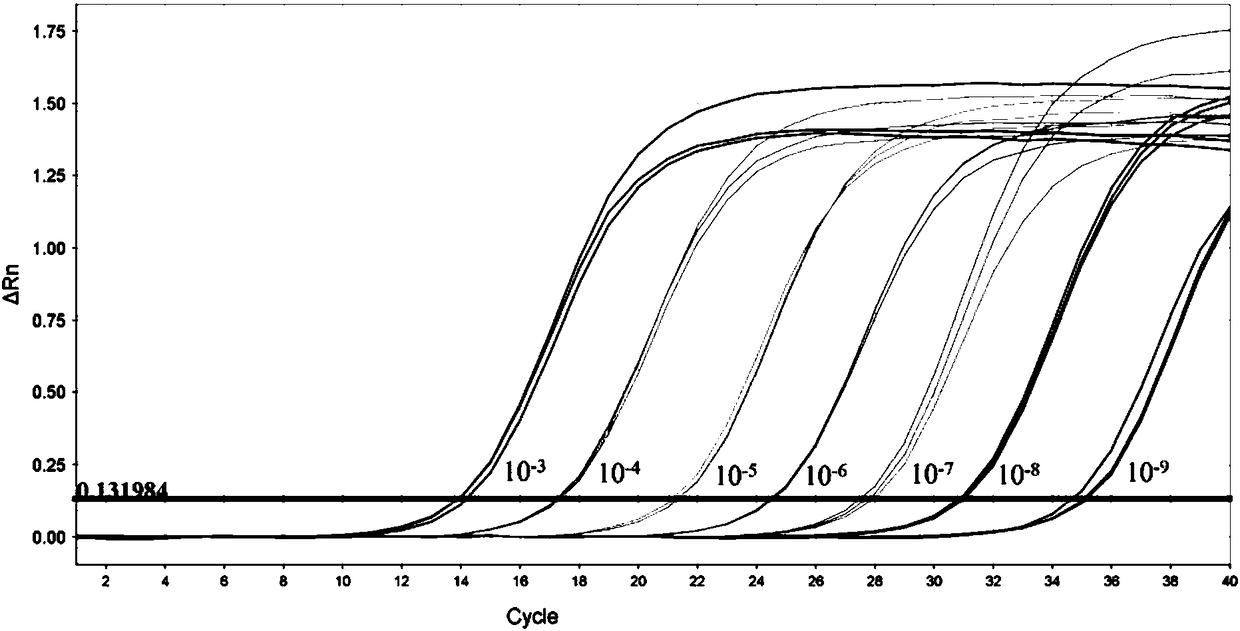
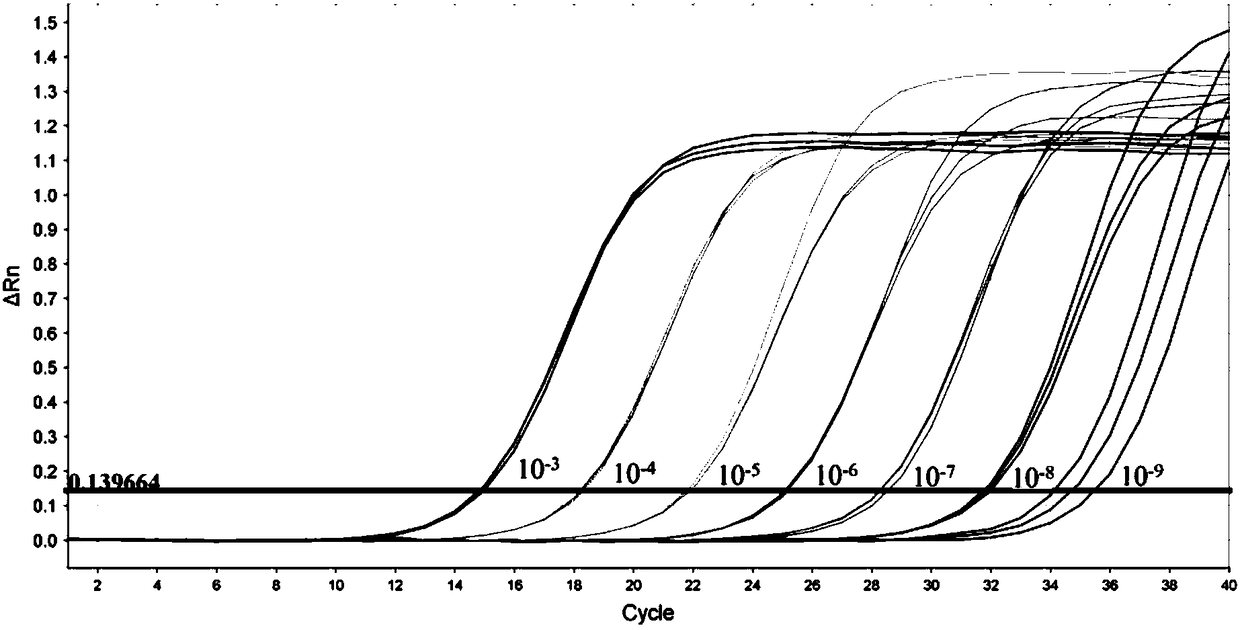
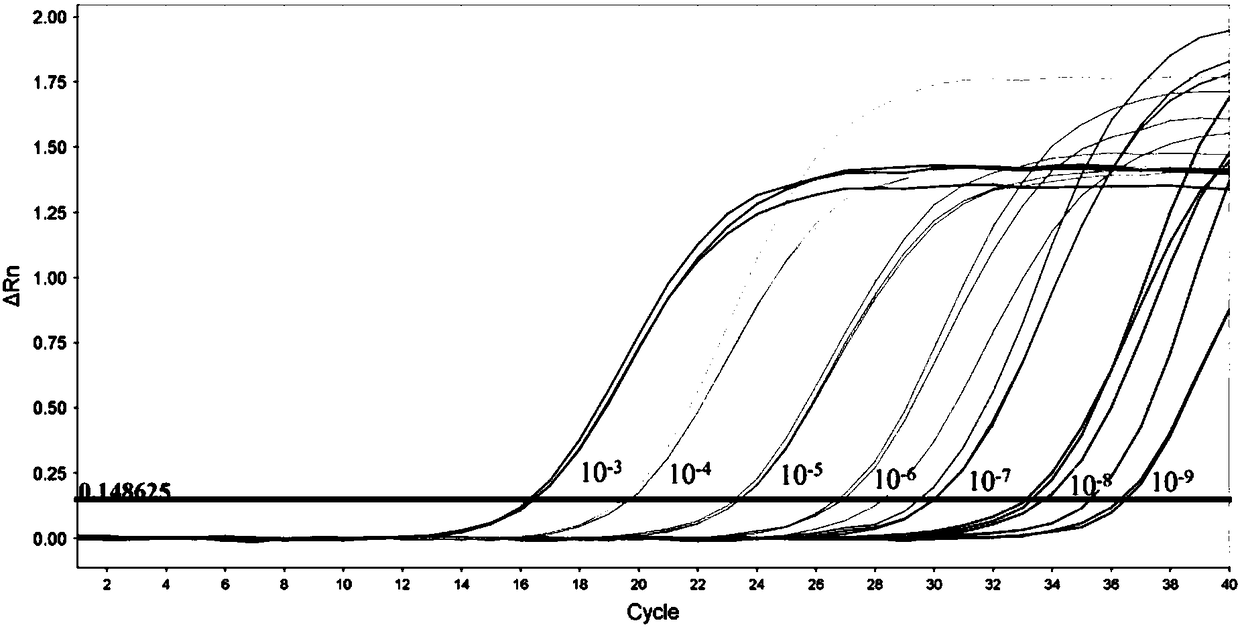
[0054] Fluorescent quantitative PCR detection is based on ordinary PCR detection, further through a specific fluorescent probe, the probe is an oligonucleotide, and the two ends are respectively labeled with a reporter fluorescent group and a quencher fluorescent group. When the probe is intact, the fluorescent signal emitted by the reporter group is absorbed by the quencher group; during PCR amplification, the 5'-3' exonuclease activity of Taq enzyme degrades the probe, so that the reporter fluorescent group and the quencher group The fluorescent group is separated, so that the fluorescence monitoring system can receive the fluorescent signal, that is, every time an RNA strand is amplified, a fluorescent molecule is formed, and the accumulation of the fluorescent signal is completely synchronized with the formation of the PCR product. Therefore, the premise of fluorescent quantitative PCR detection is to carry out PCR amplific...
Embodiment 2
[0081] Real-time fluorescent PCR detection kit of embodiment 2 Congo fever virus, Kupo fever virus type 1 and Kupo fever virus type 2
[0082] The fluorescent quantitative PCR detection kit for the detection of Congo fever virus, Kupo virus type 1 and Kupo fever virus type 2 consists of the following components:
[0083] 2×RT-PCR buffer;
[0084] 25×RT-PCR Enzyme Mix
[0085] The upstream primer sequence of Congo fever virus is shown in SEQ ID No.1, and the downstream primer sequence is shown in SEQ ID No.2, and the probe sequence is shown in SEQ ID No.3, and the 5' mark of the probe SEQ ID No.3 TEXRED, 3' tagged BHQ2;
[0086] The upstream primer sequence of Kupovirus type 1 is shown in SEQ ID No.4, SEQ ID No.4, the downstream primer sequence is shown in SEQ ID No.5, and the probe sequence is shown in SEQ ID No.6. Wherein, the probe 5' mark FAM of SEQ ID No.6, 3' mark TAMRA;
[0087] The upstream primer sequence of Kupovirus type 2 is shown in SEQ ID No.7, the downstream ...
Embodiment 3
[0095] Embodiment 3 simultaneously detects the method for three kinds of virus fluorescent quantitative PCR
[0096] The method for simultaneously detecting three viruses of Congo fever virus, Kupo fever virus type 1 and Kupo fever virus type 2 by fluorescent quantitative PCR comprises the following steps:
[0097] (1) Pathogen RNA extraction
[0098] Roche's viral RNA kit can be used for extraction.
[0099] (2) Fluorescent quantitative PCR amplification
[0100] The mixed design of upstream and downstream primers and probes of Congo fever virus, Kupo fever virus type 1 and type 2, take equal volumes of primers and mix to form primer MIX, the final concentration of each primer is 3.3μmol / L, each take equal volumes The probes were mixed to form a probe MIX, and the final concentration of each probe was 1.0 μmol / L.
[0101] Using AgPath-ID One-step RT-PCR of ABI Company, 25 μL reaction system: primer MIX 2.0 μL; probe MIX 1.0 μL; 2×RT-PCR buffer 12.5 μL; 25×RT-PCR Enzyme Mix...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap