Medicinal composition for rapidly alleviating pain and eliminating swelling after operation and preparation method thereof
A pharmaceutical composition and rapid technology can be used in pharmaceutical formulations, pharmaceutical combinations, medical preparations containing active ingredients, etc., and can solve the problems of unsatisfactory anti-inflammatory and analgesic effects, differences in effects, and insignificant differences in beauty seekers. Reduced migration and phagocytosis, better efficacy, less gastrointestinal reactions and psychiatric symptoms
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
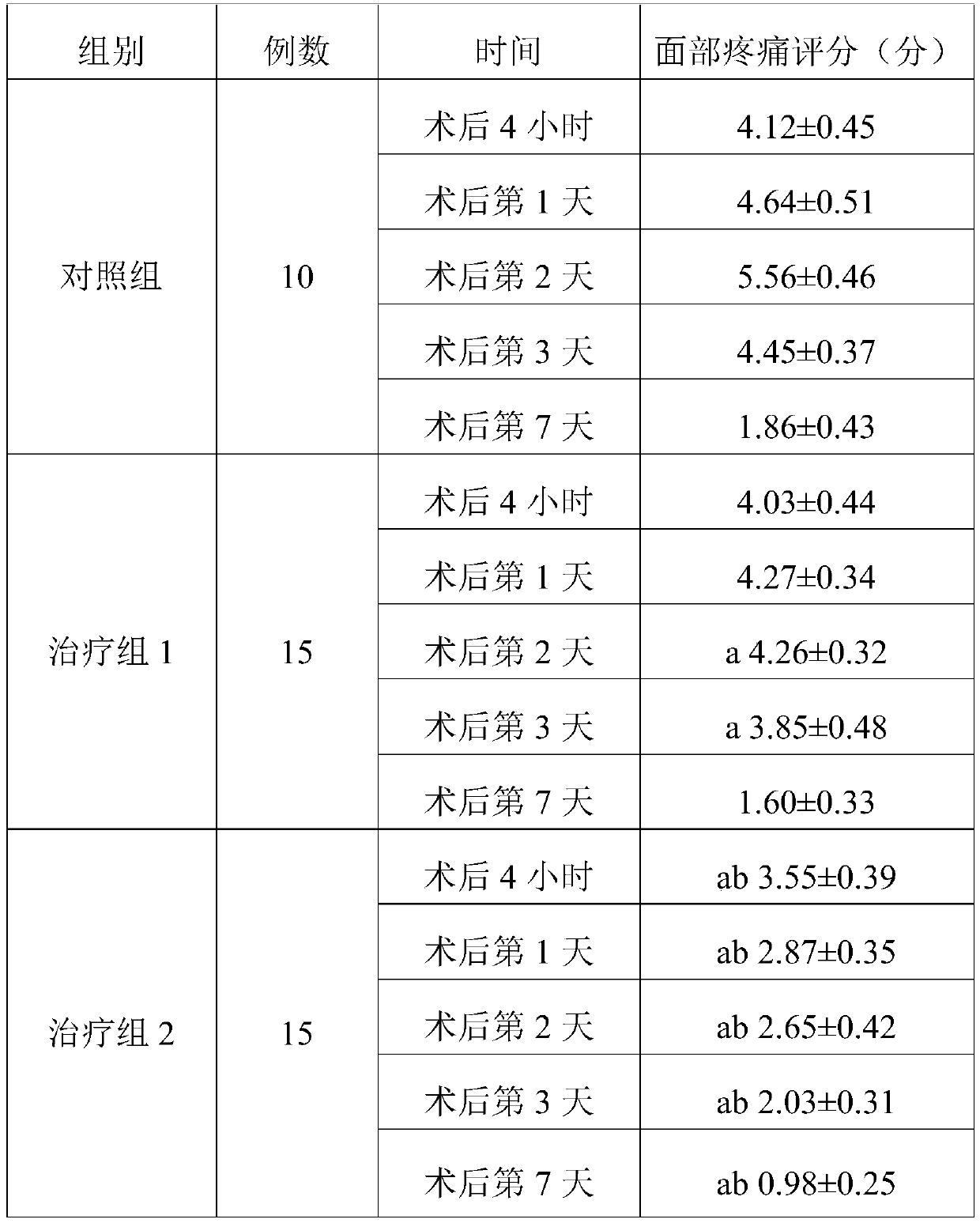
[0037] The invention provides a pharmaceutical composition for rapid postoperative pain relief and detumescence, which consists of non-steroidal anti-inflammatory drug injection, glucocorticoid injection, serotonin receptor antagonist injection, vitamin injection and normal saline composition.
[0038] Among them, non-steroidal anti-inflammatory drug injection: glucocorticoid injection: serotonin receptor antagonist injection: vitamin injection: normal saline = 25~50mg: 2.5~20mg: 2.5~15mg: 0.25~3.0g: 100ml .
[0039] In the present embodiment, the non-steroidal anti-inflammatory drug injection is selected from 100% flurbiprofen axetil injection; the glucocorticoid injection is selected from 100% dexamethasone injection; the serotonin receptor antagonism 100% tropisetron injection is selected for the dosage injection; 100% vitamin C injection is selected for the vitamin injection.
[0040] The flurbiprofen axetil injection used in the present invention is the prodrug of flurb...
Embodiment 2
[0073] As another embodiment of the present invention, another pharmaceutical composition for rapid postoperative pain relief and detumescence consists of the following components: non-steroidal anti-inflammatory drug injection, glucocorticoid injection, hemostatic freeze-dried powder, serotonin receptor antagonist injection, vitamin injection and normal saline. Among them, non-steroidal anti-inflammatory drug injection: glucocorticoid injection: hemostatic freeze-dried powder: serotonin receptor antagonist injection: vitamin injection: normal saline = 25~100mg: 2.5~20mg: 0.5~2KU: 2.5 ~15mg: 0.25~3.0g: 100ml.
[0074] Compared with Example 1, the composition of Example 2 also includes hemostatic freeze-dried powder. Wherein, the hemostatic freeze-dried powder is selected from snake venom hemocoagulase for injection, approved by the Chinese Medicine Standard H20041419, produced by Penglai Nuokang Pharmaceutical Co., Ltd.
[0075] In the operation and early postoperative perio...
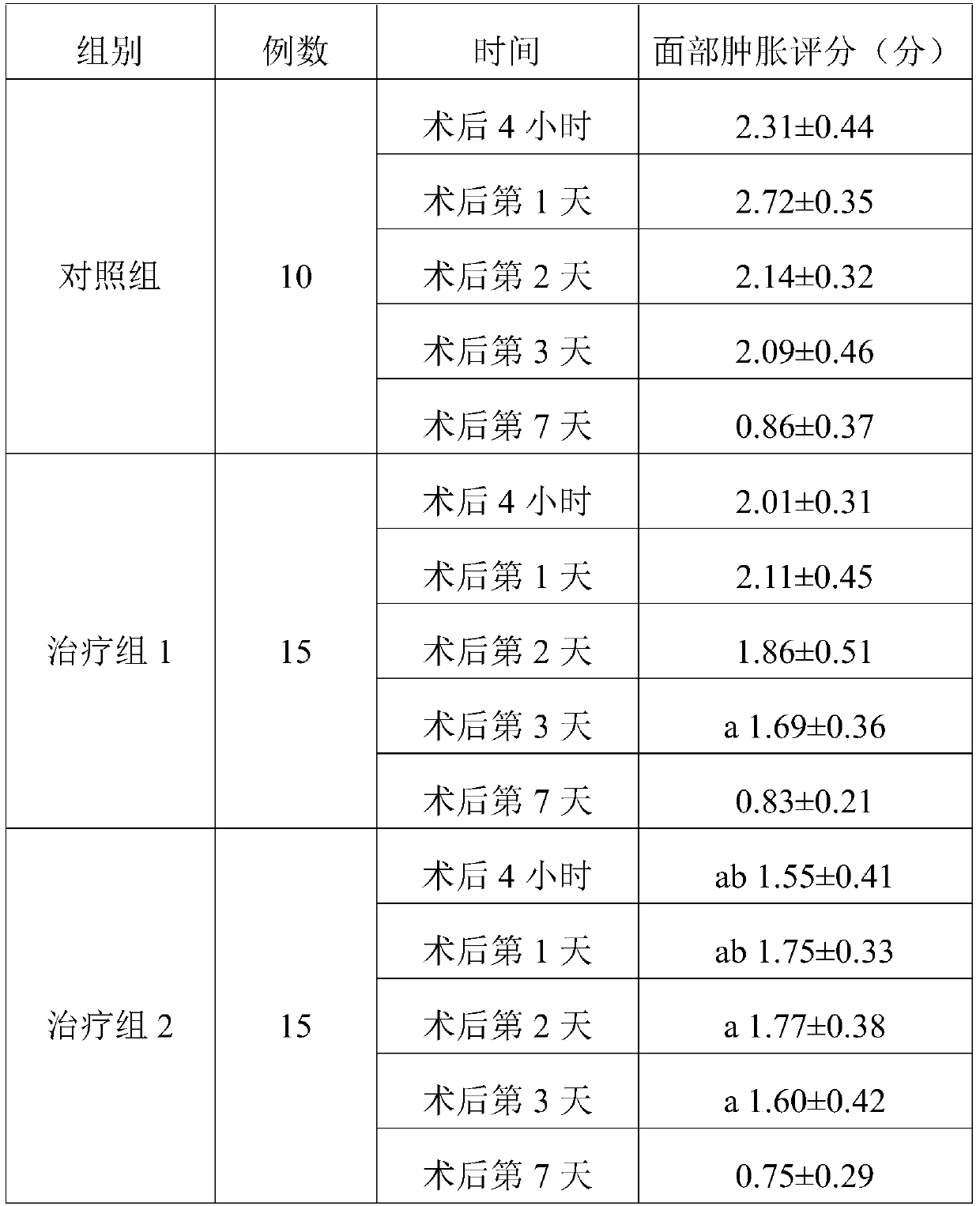
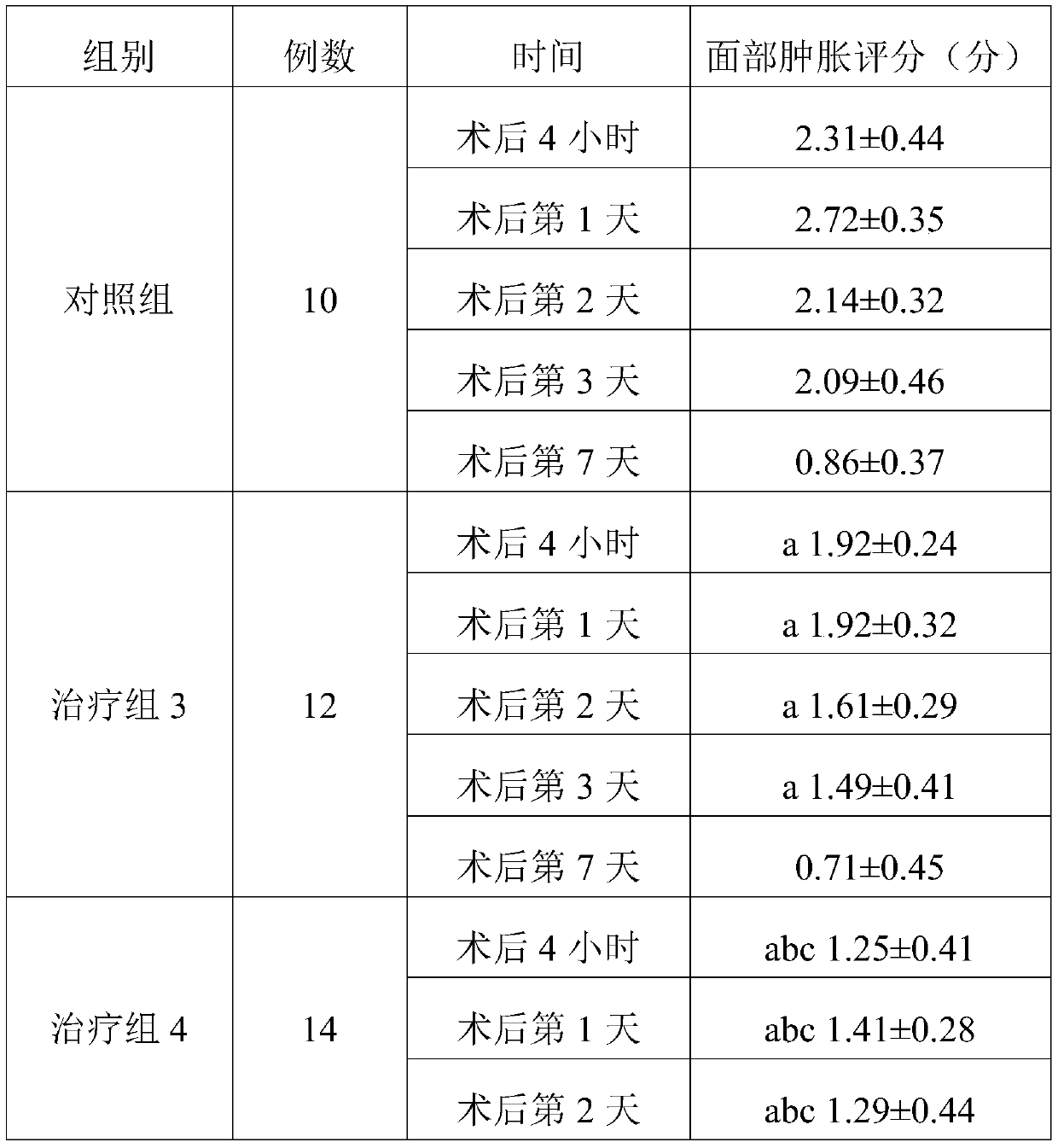
experiment example 2
[0134] Experimental composition 3: non-steroidal anti-inflammatory drug injection (flurbiprofen axetil injection): glucocorticoid injection (dexamethasone): 5-HT3 receptor antagonist (tropisetron): normal saline =25mg: 5mg: 5mg: 100ml.
[0135] Experimental composition 4: non-steroidal anti-inflammatory drug injection (flurbiprofen axetil injection): glucocorticoid injection (dexamethasone): 5-HT3 receptor antagonist (tropisetron): normal saline =25mg: 20mg: 15mg: 100ml.
[0136] In addition, 26 female patients with facial plastic surgery were selected, aged 23 to 47 years, without systemic diseases, bleeding and coagulation abnormalities. Randomly divided into two groups: treatment group 3 and treatment group 4. There were 12 cases in the treatment group 3, 14 cases in the treatment group 4, and the experimental data in the pre-clinical experiment case 1 were used in the control group. The treatment group 3 was 23-47 years old, and the treatment group 4 was 24-43 years old...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap