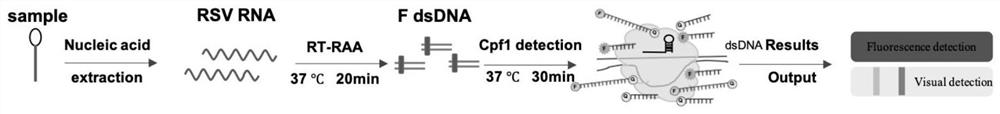
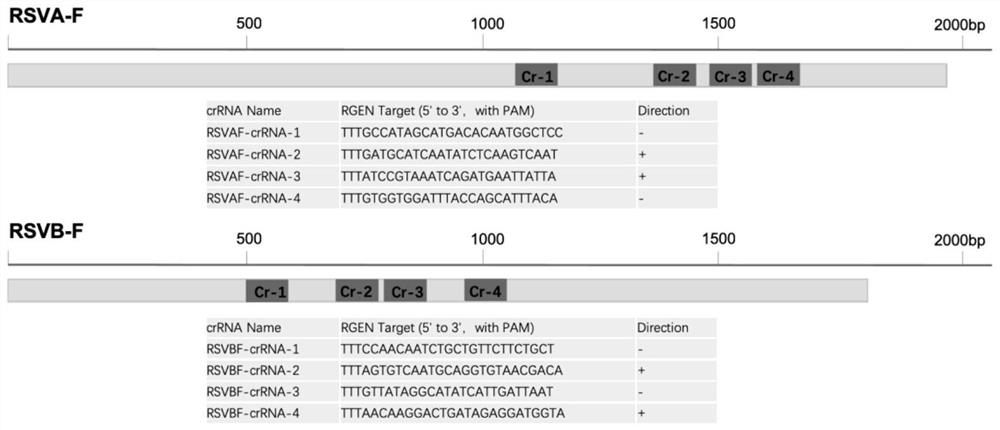
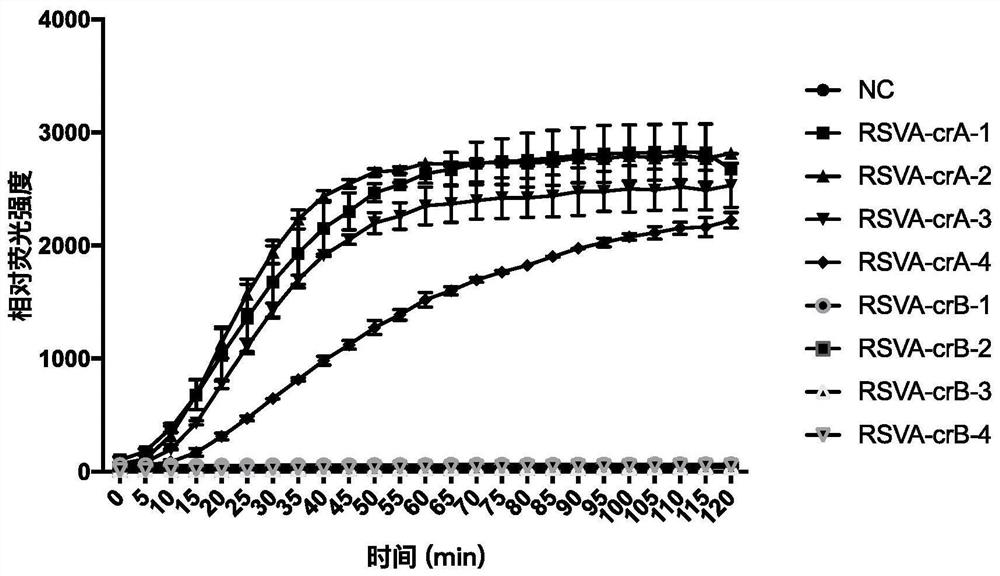
Respiratory syncytial virus nucleic acid rapid detection kit based on CRISPR/Cas12a and detection method
A technology of syncytial virus and detection method, which is applied in the genetic detection of respiratory syncytial virus and the biological field, can solve the problems of being unsuitable for early diagnosis, complicated technology, difficult to judge the results, etc., and achieve high-specificity, rapid visual detection, and high sensitivity. The effect of visual detection, convenient and quick result interpretation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0041] Example 1: Rapid and sensitive detection of respiratory syncytial virus nucleic acid fragments
[0042] 1.1 Nucleic acid preparation
[0043] In this case, the RSV gene fragment refers to the F gene fragment corresponding to the A and B subtypes of RSV in the NCBI database, and the 620bp SEQ NO.1 of the F part gene of the A subtype and the F part of the B subtype were synthesized by Nanjing GenScript Company The 710bp SEQNO.2 of the gene was constructed into the pUC57 vector, named pUC57-RSVA-F and pUC57-RSVB-F, and the T7 promoter was introduced before the synthetic fragment, and the synthesized DNA fragment was transcribed into RNA by in vitro transcription reagent, named as pUC57-RSVA-F-RNA and pUC57-RSVB-F-RNA.
[0044] Utilize RT-RAA amplification primer RSVA-RT-RAA-F (SEQ NO.3) in the present invention, RSVA-RT-RAA-R (SEQNO.4), RSVB-RT-RAA-F (SEQ NO.5) and RSVB-RT-RAA-R (SEQ NO.6), referring to the RT-RAA isothermal amplification operation steps, amplified to ob...
Embodiment 2
[0063] Embodiment 2: CRISPR / Cas12a detects respiratory syncytial virus nucleic acid sensitivity
[0064] In the case of sensitivity detection, the plasmid DNA is transcribed in vitro into RNA of the RSVA / B subtype, converted to copy number according to the molecular weight, and then serially diluted 10 times to obtain 2*e7, 2*e6, 2*e5 per microliter , 2*e4, 2*e3, 2*e2, 2*e1 and 2*e0 copy number (copy / μL). 1 μL of gradient dilution samples were subjected to RT-RAA amplification reaction: 25 μL 2*Buffer, 2 μL RT-RAA-F, 2 μL RT-RAA-R and 2.5 μL magnesium acetate, mixed well, reacted at 37°C for 20 minutes, obtained samples for The next step is nucleic acid testing.
[0065] According to the results obtained in Example 1, RSVAF-crRNA-2 and RSVBF-crRNA-3 have higher sensitivity to the detection of respiratory syncytial virus genes of A and B subtypes, so these two specific crRNAs are used for subsequent detection. This test uses a 20 μL system as shown in Table 3, but is not limi...
Embodiment 3
[0071] Example 3: Rapid detection of respiratory syncytial virus nucleic acid on clinical sample nucleic acid
[0072] In this example, rapid detection of nucleic acid in clinical samples is performed. All samples and operations are completed in the laboratory. In this case, the sample is nucleic acid obtained from throat swab for CRISPR / Cas12a detection. In this example, a viral DNA / RNA extraction kit (magnetic bead method) purchased from Novizyme was used to obtain pretreated nucleic acid. The steps are as follows: wash the oral swab with PBS, add 20 μL proteinase K to the cleaning solution, add magnetic beads to incubate, let it stand at room temperature for 5 minutes, wash the magnetic beads twice with a magnetic stand, add 50 μL eluent to elute and proceed to the next step detection.
[0073] Using RT-RAA amplification primers SEQ NO.3 and SEQ NO.6 in the present invention, referring to the RT-RAA isothermal amplification operation steps, take 2 μl of each sample to be t...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap