Method for controlling impurities in pipericide
A technology of piperbocilide and a control method, which is applied in the field of impurity control in breast cancer drug piperbacillide, can solve problems such as poor impurities and low product purity, and achieve the effects of excellent quality, high purity and simple control method.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0073] In the present embodiment, the purity of each intermediate is controlled as follows:
[0074] The purity of intermediate I is as follows: the compound I content is 0.4%, the compound II content is 1.5%, the largest single unknown impurity content is 1.2%, and the total impurity content is 3.4%.
[0075] The purity of intermediate II is as follows: the content of intermediate I is 0.1%, the largest single unknown impurity content is 1.0%, and the total impurity content is 2.3%.
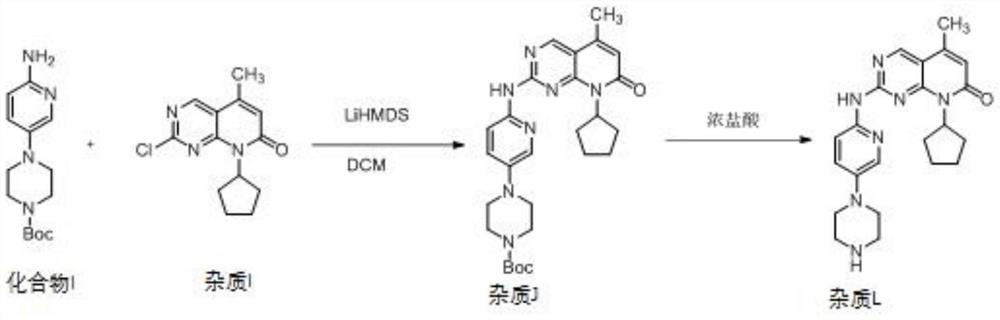
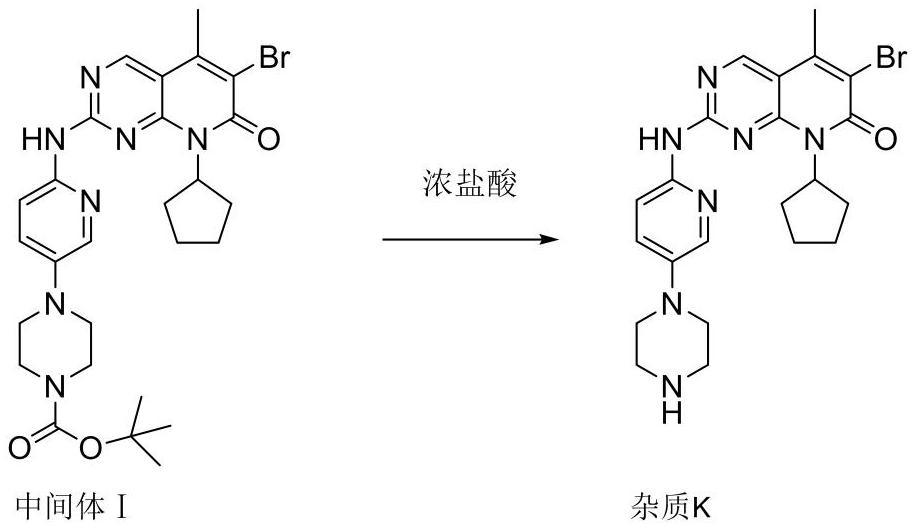
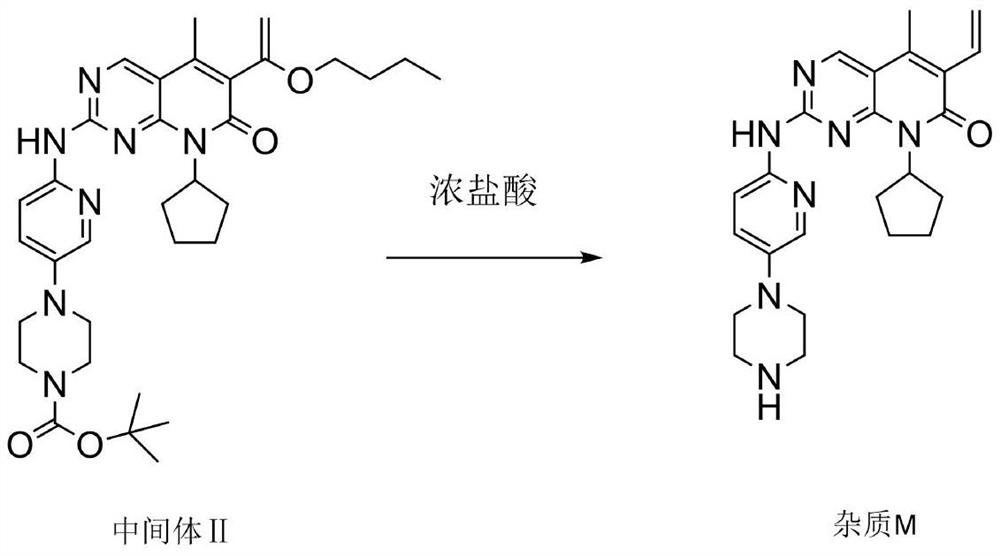
[0076] The purity of palbociclib salt is as follows: the content of impurity L contained is 0.1%, the content of impurity K shall not exceed 0.1%, the content of impurity M is 0.2%, the content of impurity N is 0.2%, the content of the largest single unknown impurity is 0.1%, and the content of total impurities is 0.8%. .
[0077] The content of the generated final product palbociclib and the content of impurities contained therein were detected, and the results are shown in Table 1.
Embodiment 2
[0079] In the present embodiment, the purity of each intermediate is controlled as follows:
[0080] The purity of intermediate I is as follows: the compound I content is 0.3%, the compound II content is 1.1%, the largest single unknown impurity content is 0.9%, and the total impurity content is 2.7%.
[0081] The purity of intermediate II is as follows: the content of intermediate I contained is 0.1%, the largest single unknown impurity content is 0.9%, and the total impurity content is 1.8%.
[0082] The purity of palbociclib salt is as follows: the content of impurity L is 0.1%, the content of impurity K is 0.1%, the content of impurity M is 0.1%, the content of impurity N is 0.2%, the content of the largest single unknown impurity is 0.1%, and the content of total impurities is 0.7%.
[0083] The content of the generated final product palbociclib and the content of impurities contained therein were detected, and the results are shown in Table 1.
Embodiment 3
[0085] In the present embodiment, the purity of each intermediate is controlled as follows:
[0086] The purity of intermediate I is as follows: the compound I content is 0.3%, the compound II content is 1.4%, the largest single unknown impurity content is 1.1%, and the total impurity content is 3.5%.
[0087] The purity of intermediate II is as follows: the content of intermediate I is 0.2%, the largest single unknown impurity content is 0.9%, and the total impurity content is 3.0%.
[0088] The purity of the palbociclib salt is as follows: the impurity L content is 0.1%, and the impurity K content is 0.1%. The impurity M content is 0.1%, the impurity N content is 0.2%, the largest single unknown impurity content is 0.2%, and the total impurity content is 0.9%.
[0089] The content of the generated final product palbociclib and the content of impurities contained therein were detected, and the results are shown in Table 1.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More