Antibody vaccine conjugates and uses therefor
A technology of conjugates and antibodies, applied in the field of antibody vaccine conjugates and their applications, can solve the problems of no effective
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0228] Example 1: Production of βhCG-B11
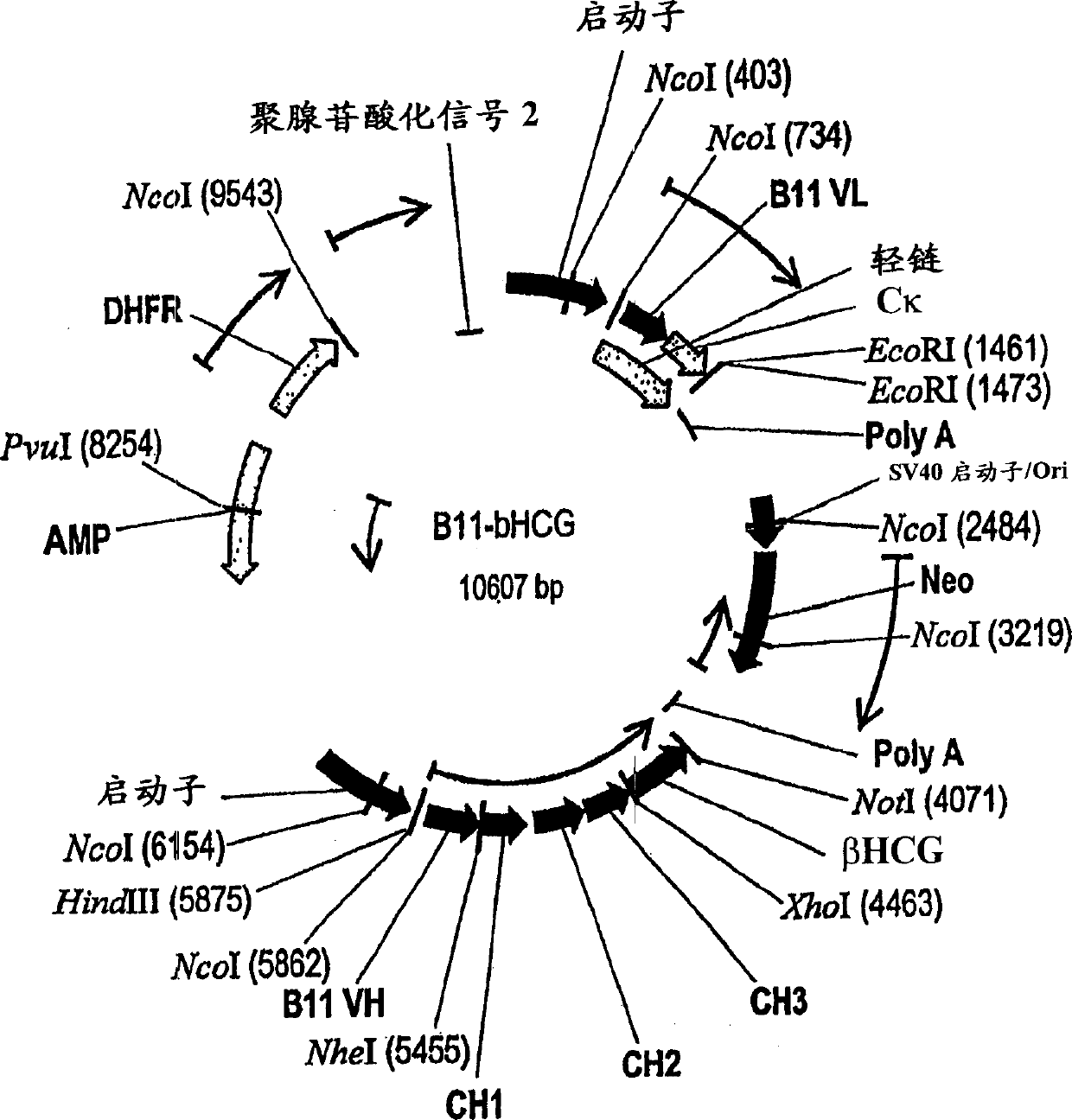
[0229] Design of vaccine conjugates : This construct was generated by linking the βhCG antigen to B11, a fully human antibody that binds to the human macrophage mannose receptor on dendritic cells. Through the gene fusion method, the antigen and the heavy chain of the antibody are bonded through covalent bonding, as shown in FIG. 3 .
[0230] Recombinant Expression of βhCG-B11 Vaccine Conjugate : As shown in Figure 2, a plasmid containing the neomycin gene and the dihydrofolate reductase gene containing the antibody B11 in the heavy chain CH 3 βhCG coding sequence (SEQ ID NO: 9 and 10) fused to the region. The resulting plasmid constructs were transfected into CHO cells using standardized protocols (Qiagen Inc, Valencia, CA). Transfected cells were selected in medium containing the antibiotic G418. Expression is further amplified by growing cells in increasing concentrations of methotrexate. After expansion, cells are cloned b...
Embodiment 2
[0231] Example 2: Production of B11scfv-βhCG
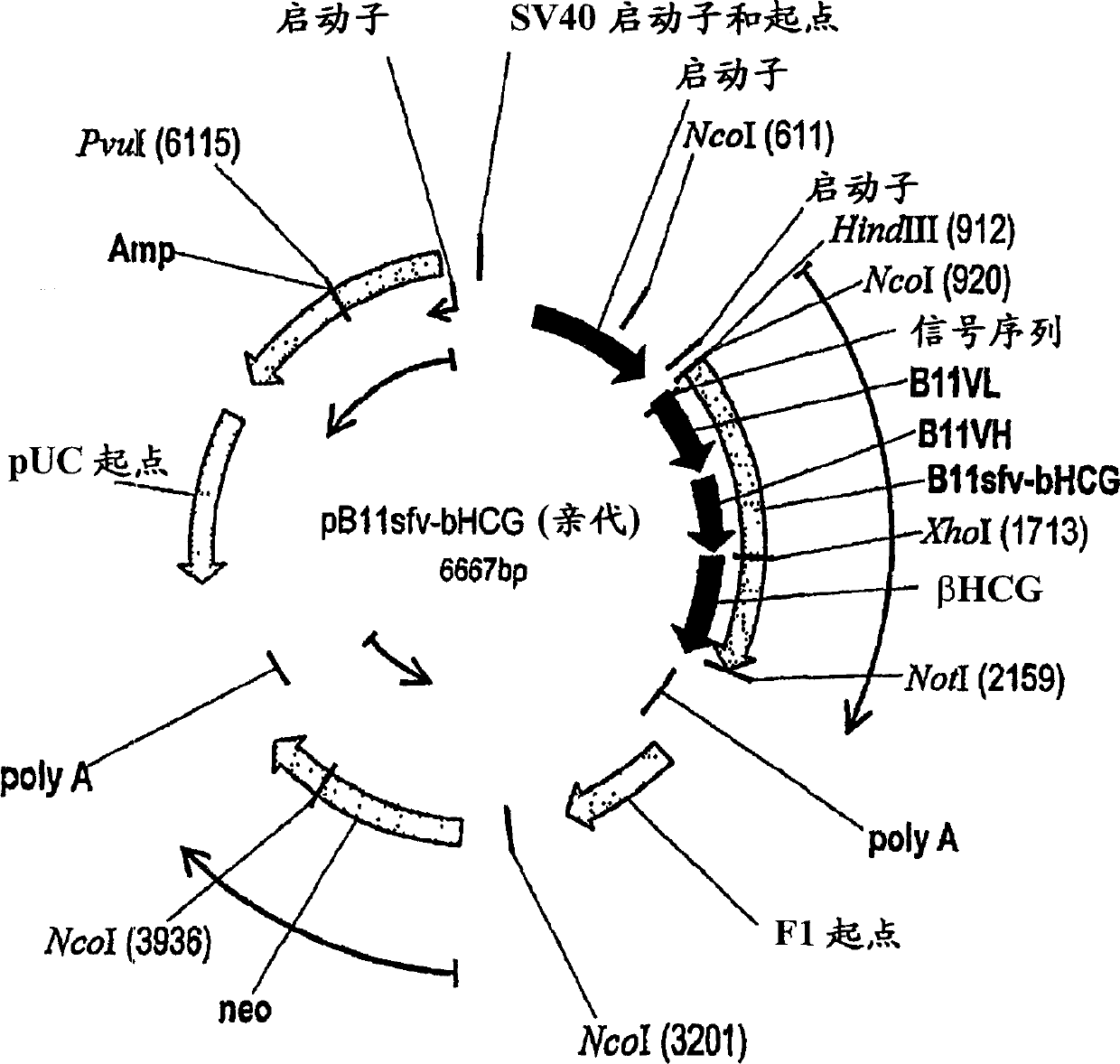
[0232] Design of vaccine conjugates : A second construct was created by linking the βhCG antigen to a B11 single-chain fusion (ScFv) that binds to the human macrophage mannose receptor on dendritic cells and contains a fully human B11 antibody V L and V H fragments of single-chain antibodies. Through the method of gene fusion, the antigen is bonded to the carboxyl terminus of B11ScFv through covalent bonding, as shown in FIG. 1 (referred to as B11sfv-βhCG construct).
[0233] Recombinant Expression of B11sfv-βhCG Vaccine Conjugate : As shown in Figure 1, a plasmid containing the B11sfv-βhCG construct (SEQ ID NO: 11 and 12) was generated. The resulting plasmid constructs were transfected into mammalian cells using standardized protocols (Qiagen Inc, Valencia, CA). Transfected cells were selected in medium containing the antibiotic G418. ELISA was performed to confirm the expression of the B11sfv-βhCG construct.
Embodiment 3
[0234]Example 3: Functional Characteristics of Vaccine Conjugates
[0235] Recognition of antibody-targeted vaccines to their cognate receptors on the surface of APCs is the first step in this drug delivery platform. Flow cytometry studies were used to confirm specific binding of the βhCG-B11 and B11sfv-βhCG constructs to cultured human DC expressing MR ( FIG. 4 ).
[0236] In situ staining of MR on macrophages in human skin DCs and various human tissue sections was examined using an anti-MR antibody as a probe. Cryosections of human tissue were stained with anti-MR human antibody B11. DCs present in the epidermal layer of the skin were clearly labeled with B11 antibody (data not shown). Binding to DC was noted in the epidermal layer of the skin. Furthermore, immunohistochemistry experiments with dendritic cells stained with anti-MR B11 HuMAb in all tissues tested and showed no unexpected cross-reactivity (results not shown). These studies have been repeated with βhCG-B11 ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More