Vector system
a vector system and system technology, applied in the field of neurodegenerative disorder, can solve the problems of limited efficacy and reproducibility of procedures, no satisfactory cure for parkinson's disease, and limited functional recovery
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Cloning of the Human Tyrosine Hydroxylase 1 Type 2 cDNA
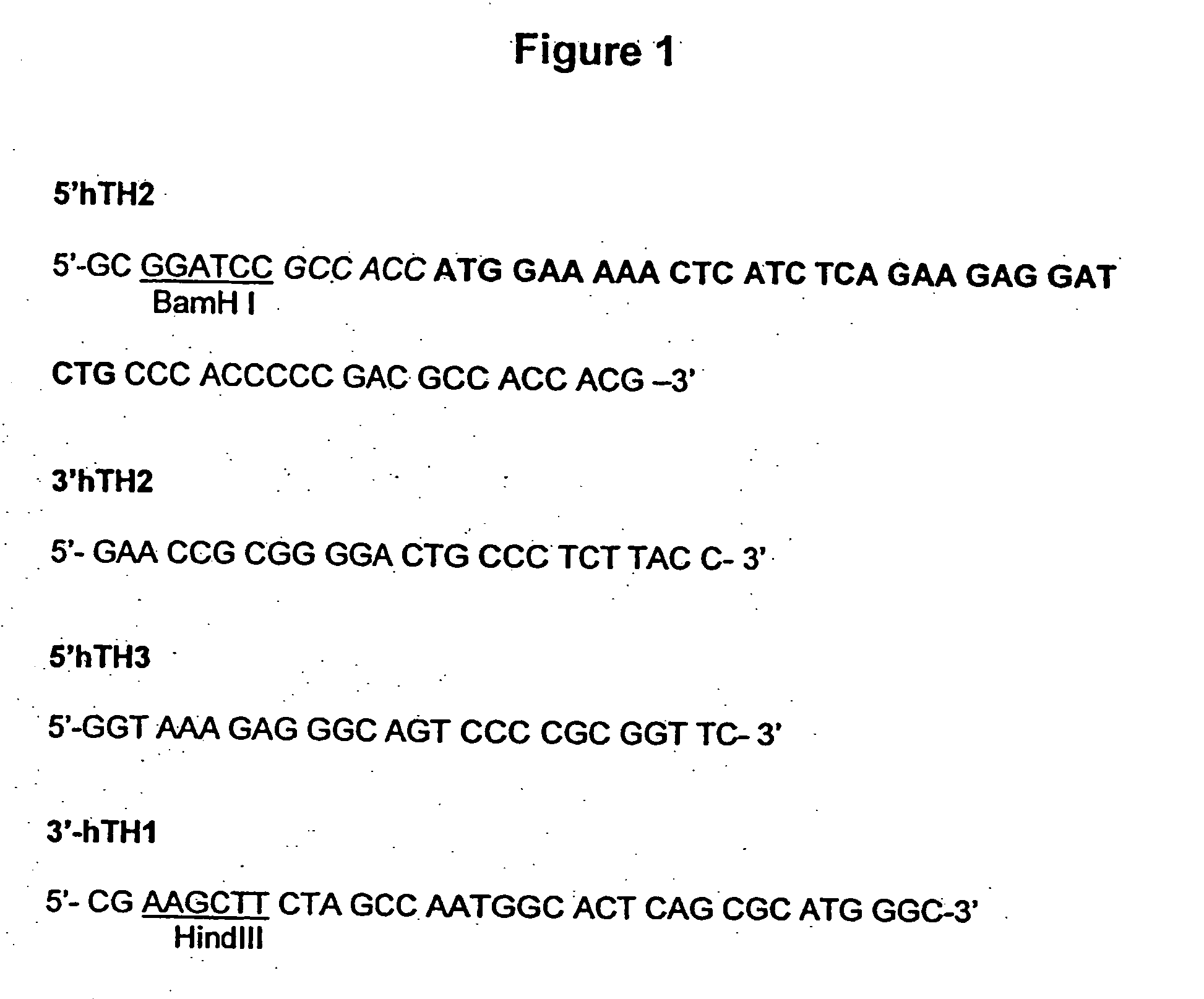
[0238] The human Tyrosine Hydroxylase 1 Type 2 cDNA (Accession number X05290) is amplified by RT-PCR from human Substantia nigra poly A+ mRNA (Clontech) and epitope tagged with the c-myc epitope using the primers described in FIG. 1. A 169 bp fragment corresponding to the 5′ end of the gene is amplified using 5′hTH2 and 3′hTH2 primers (FIG. 1) whilst the 1418 bp 3′ end fragment of the tyrosine hydroxylase cDNA is obtained using primers 5′hTH3 and 3′hTH1 (FIG. 1).
[0239] Titan One Tube RT-PCR kit (Boehringer) was used to perform the RT-PCR reaction. Typically the reaction is composed of two solutions.
[0240] Solution A
[0241] Contains 0.2 kg of human substantia nigra poly A+ RNA, 32 μM each dNTPs, 10 mM DTT, 1 μl RNAse Inhibitor (RNAsin, Promega), ˜100 ng each primer and water, up to 25 μl.
[0242] Solution B
[0243] Contains 10 μl of 5×RT-PCR Buffer, 1 μl Enzyme mix, and water up to 25 μl.
[0244] Solutions A and B are mixed and t...
example 2
Cloning of the Human Aromatic Amino Acid Dopa Decarboxylase 1 cDNA
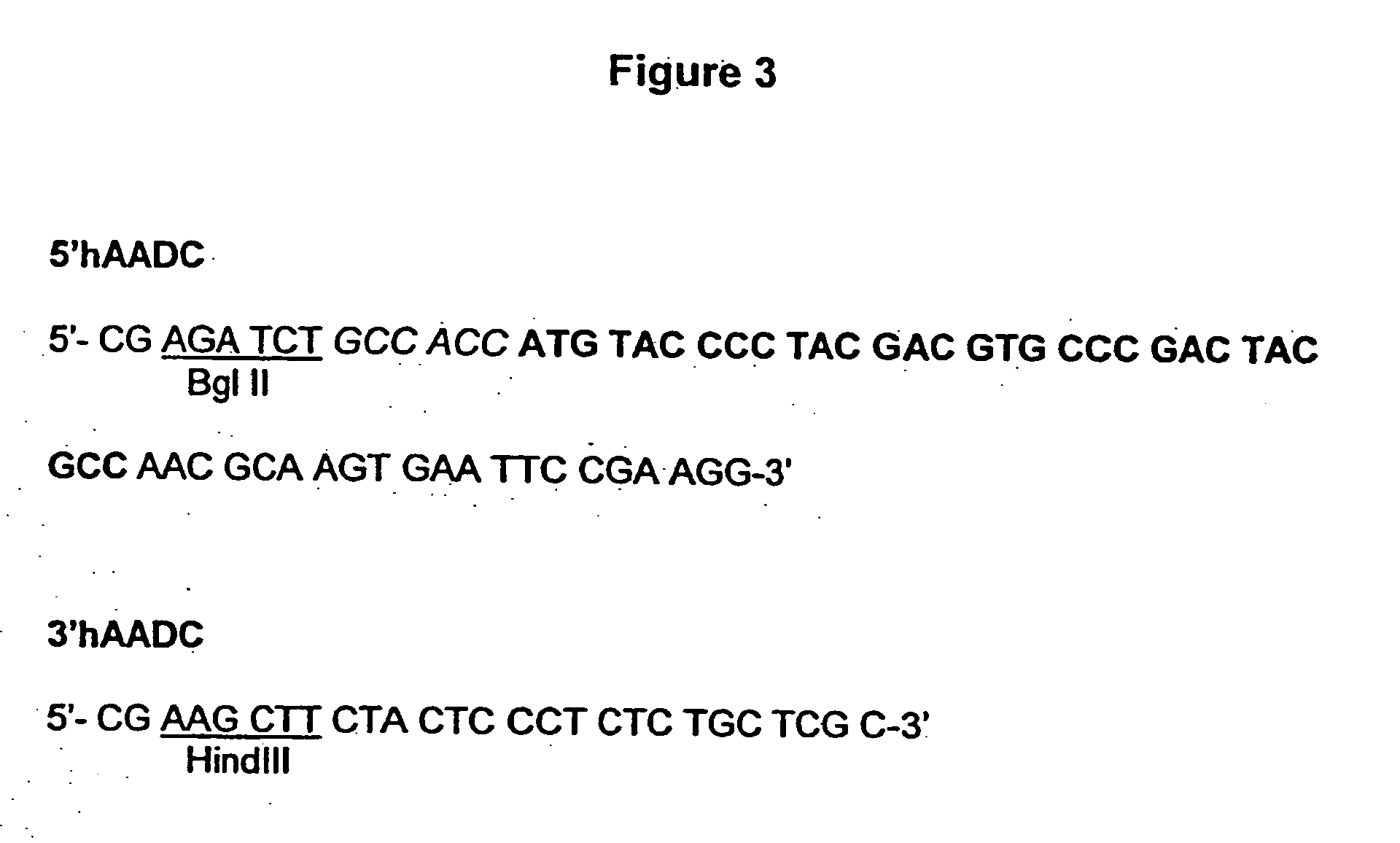
[0249] The human Aromatic Amino Acid Dopa Decarboxylase (AADC) cDNA (Accession number M76180 M30772) is amplified from a human liver cDNA expression library (Clontech) and epitope tagged with the HA epitope using the primers 5′hAADC and 3′hAADC, described in FIG. 3. The PCR reaction was performed using a KlenTaq kit (Clontech) following the instructions of the manufacturer. The reaction contains 4 μl human liver cDNA and 1 μM of each primer, in a final volume of 50 μl. The PCR conditions are as follows: A first step, 30 sec at 94° C.; a second step, 5 cycles of 30 sec at 94° C., 30 sec at 58° C. and 2 min at 68° C. and a third step, 30 cycles of 30 sec at 94° C., 30 sec at 55° C. and 2 min at 68° C.
[0250] The PCR amplifies the two expected bands, 1.485 kb and 1.36 kb, corresponding to the two transcripts of the Aromatic Amino Acid Dopa Decarboxylase (AADC). The 1.485 kb band is purified and cloned into pGEM-Teasy ve...
example 3
Cloning of the Human GTP-Cyclohydrolase 1 cDNA
[0251] The human GTP-cyclohydrolase I (GTP-CH1) cDNA (Accession number U19523) is amplified from Poly A+ mRNA from human Substantia nigra and epitope tagged with the FLAG epitope using the primers 5′hGTP and 3′hGTP (FIG. 5). Titan One Tube RT-PCR kit (Boehringer) was used to perform the RT-PCR reaction. Typically the reaction is composed of two solutions, as described above in Example 1. Solutions A and B are mixed and the RT-PCR conditions set as follows: 50° C., 30 min, to allow the RT reaction to take place, followed by 30 sec at 94° C., and 35 cycles of 30 sec at 94° C., 30 sec at 60° C. and 1 min at 68° C.
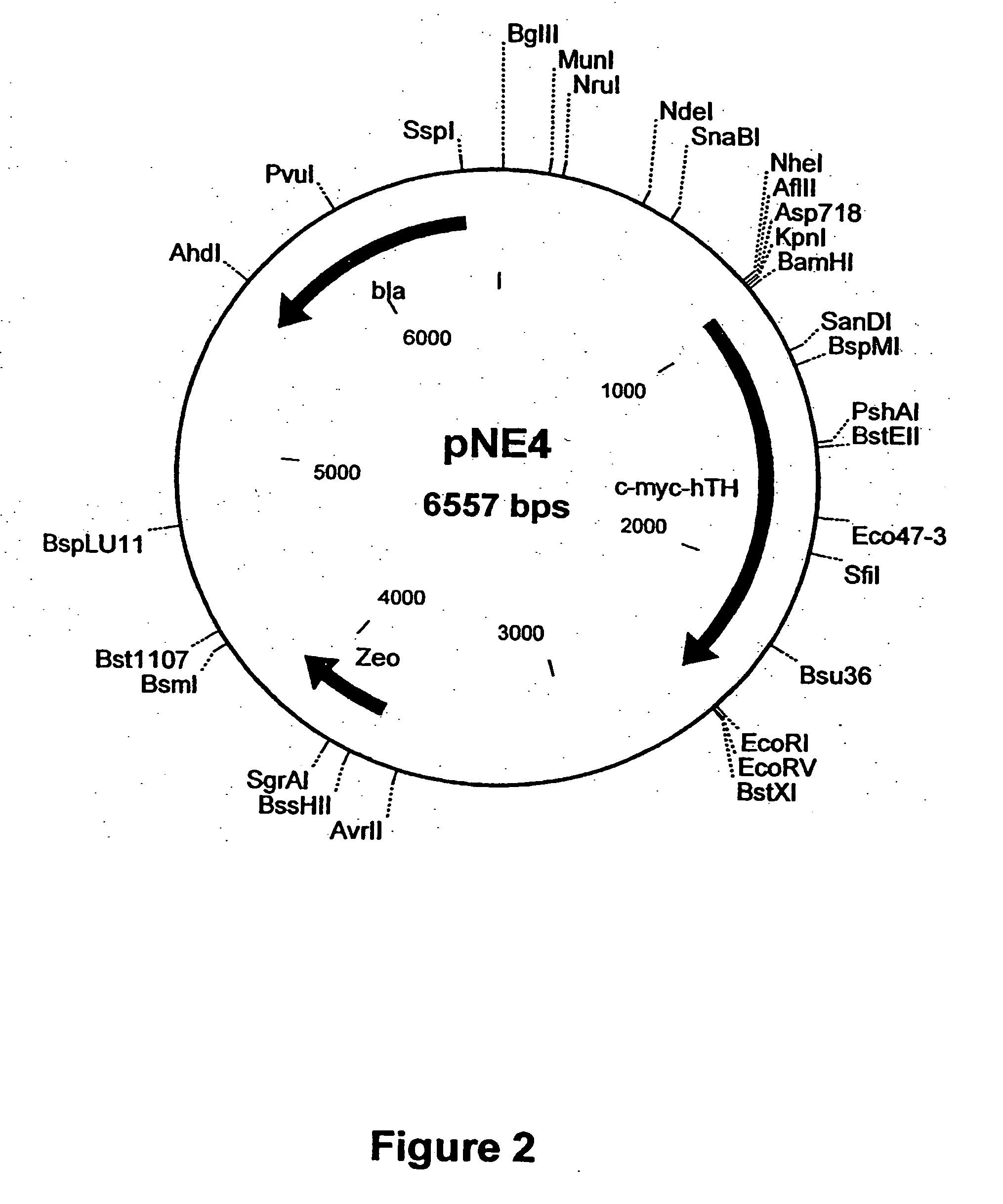
[0252] The RT-PCR product (˜0.75 kb) is purified and cloned into pGEM-Teasy vector (Promega) to generate plasmid pNE5. The GTP-CH cDNA is excised from pNE5 as a ˜0.75 kb BgIII-NotI and ligated to pcDNA3.1 / Hygro digested with BamHI and NotI enzymes to generate pNE6 (FIG. 6).
PUM
| Property | Measurement | Unit |
|---|---|---|
| volume | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
| apparent molecular weight | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More