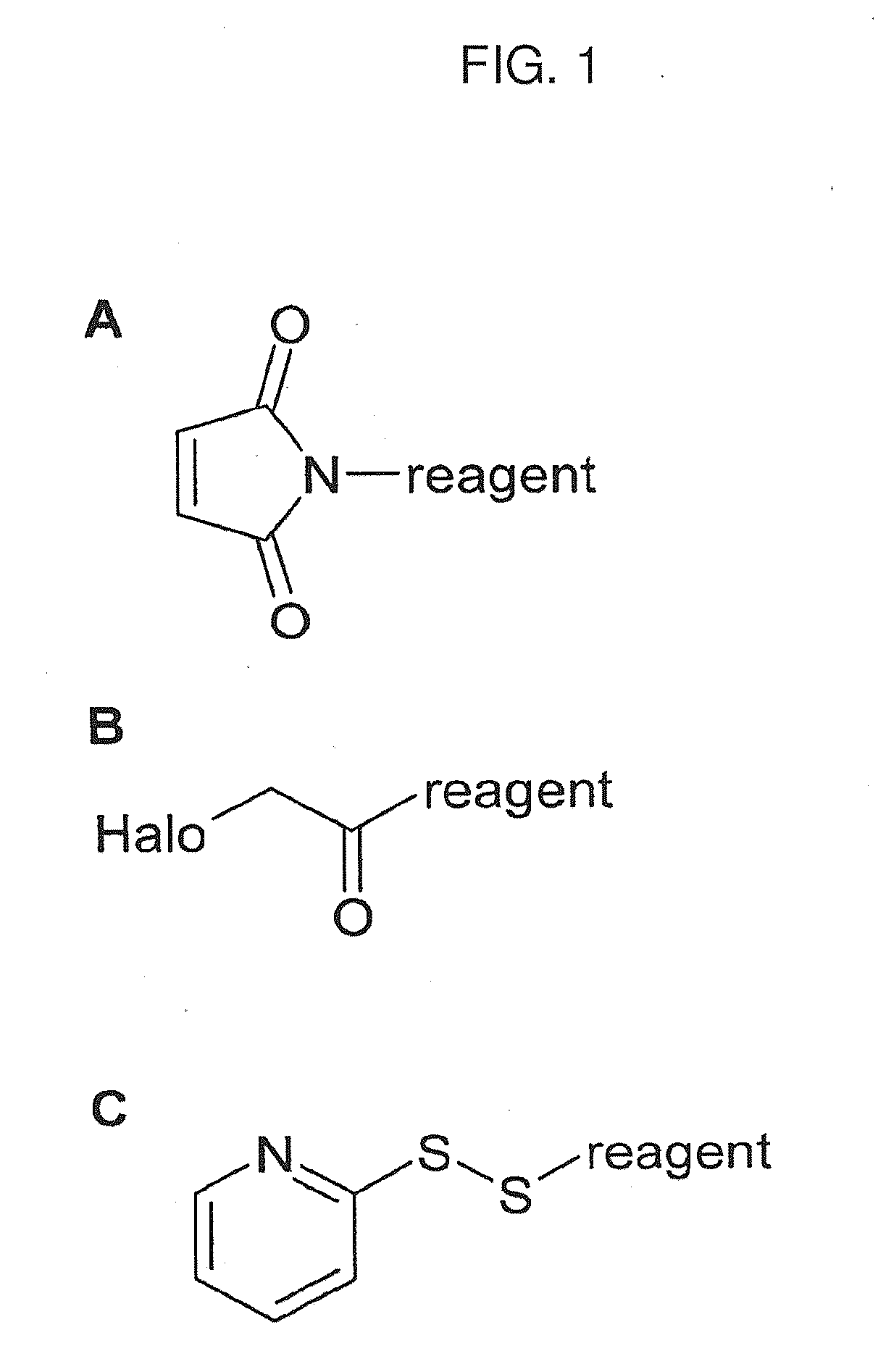
Mass labels
a technology of mass labels and labels, applied in the field of mass labels, can solve the problems of increasing the complexity of the mass spectrum, not always possible available synthetic standards, etc., and achieve the effect of limiting the multiplexing rate of a single isobaric mass tag set, rapid and inexpensiv
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Synthesis of dimethylpiperidine-βalanine-dithiopyridine: A Cysteine-Reactive Mass Label
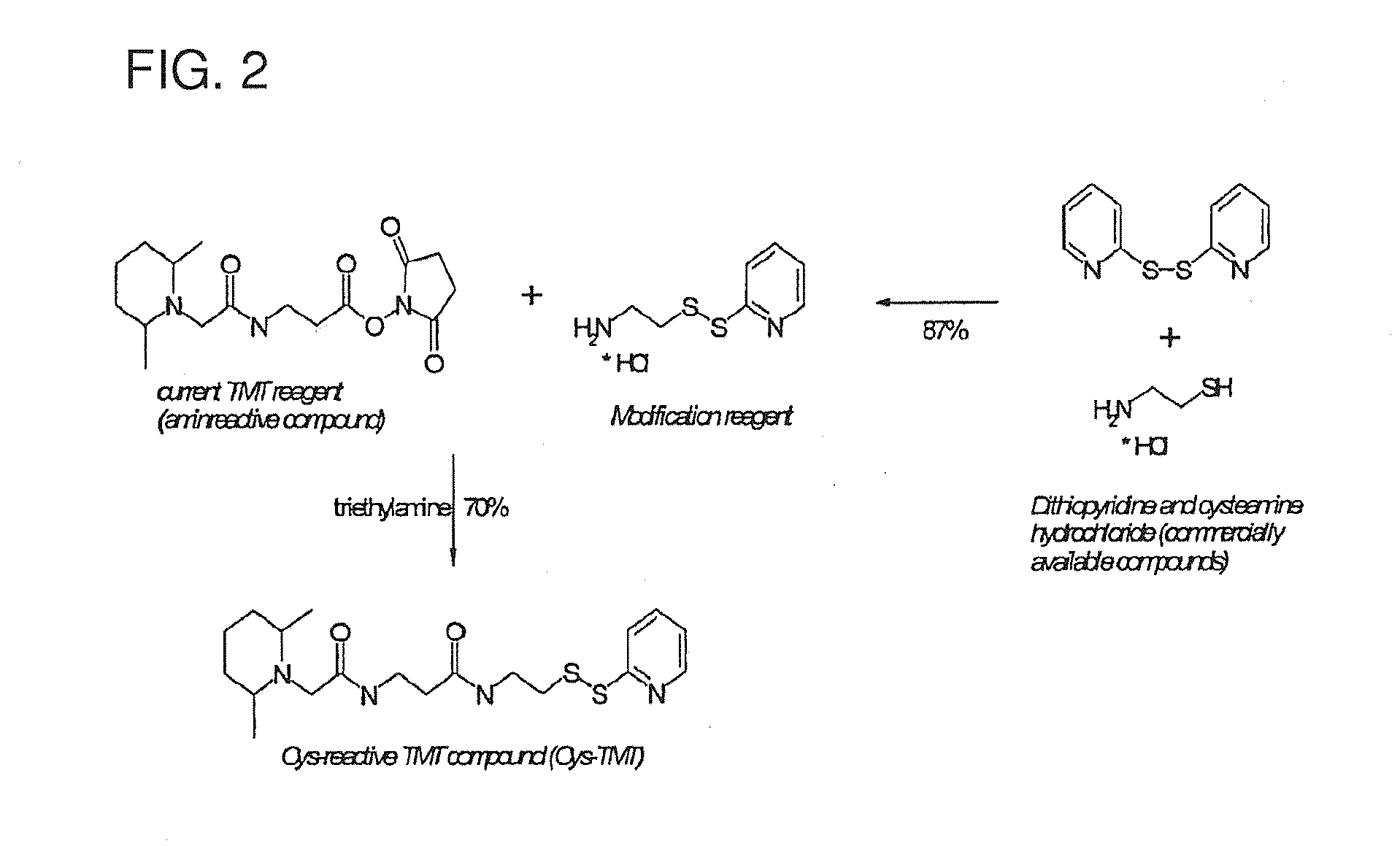
[0310]It is a convenient feature of the DMPip-βALA-OSu structure that alternate reactivities can be readily created by reaction with the succinimide ester reactive group. Thus, using the known TMT (Tandem Mass Tag or mass label) structure previously disclosed in WO2007 / 012849 as a starting point and commonly available building blocks, a reaction scheme was designed which requires only one more reaction step to convert the amino-reactive compound into the respective cysteine-reactive compound. We first synthesised the dithiopyridine modification reagent from commercially available compounds in a single reaction with good yield. This reagent was then allowed to react with DMPip-βALA-OSu to generate the DMPip-βALA-DTP reagent (see FIG. 2). Analysis by HPLC, MS and MS / MS revealed the correct identity and a purity >90%.
example 2
Labelling of Synthetic Peptides Using DMPip-βALA-DTP
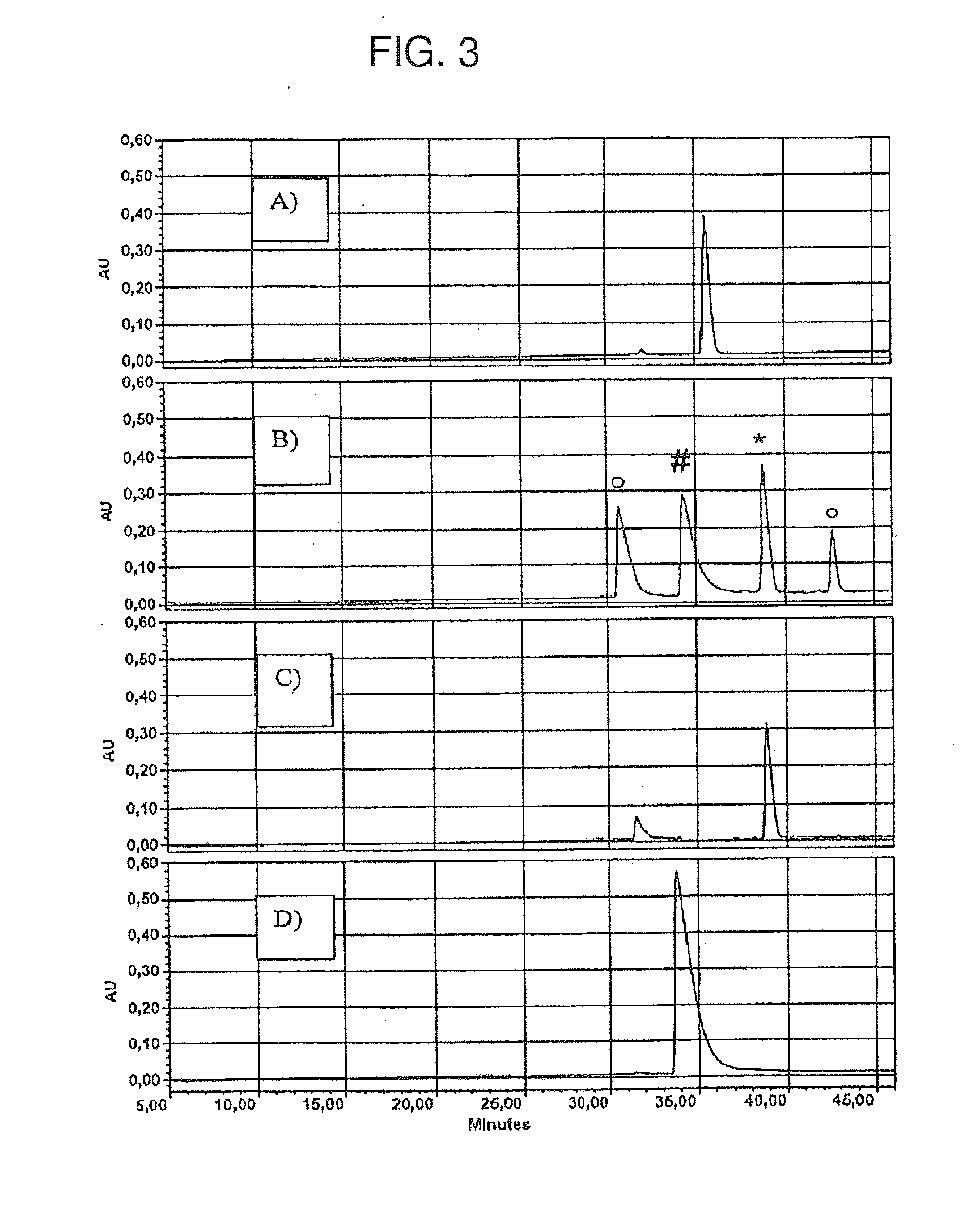
[0311]The quality controlled DMPip-βALA-DTP reagent was then applied to develop a labelling protocol. An individual cysteine-containing peptide (H-Vat-Ala-Thr-Val-Cys-Leu-Pro-Arg-OH) was chosen to allow for an easy monitoring of the reaction progress by HPLC with UV-readout. A protocol was developed that yields an essentially complete labelling. 50 μg BSA were dissolved in 100 μL TEAB (100 mM, pH 8.5) including 0.1% SDS. After reduction with 1 mM of tris[2-carboxyethyl]phosphine * HCl for 1 h at 55° C., the Cys residue was labelled with 5 mM DMPip-βALA-DTP (provided as 200 mM stock solution in methanol). Purification of the reaction mixture using RP and SCX cartridges obtained modified peptide in a highly purified form. FIG. 3 shows HPLC monitoring of the individual species in the labelling reaction.
example 3
Synthesis of Mass Labels for Steroid Analysis
[0312]The analysis of hormones is typically performed by radioimmunoassay and colorimetric or chemiluminescent enzyme immunoassay. However, current immunoassays are disadvantageous since they are limited to quantitative detection of only one steroid hormone per assay, may lack specificity, and as a result of such cross-reactivity can show up to 15 fold variance in the quantitative results of the same sample when using kits from different manufacturers.
[0313]An alternate approach for measurement of steroids is gas chromatography-mass spectrometry (GC-MS). GC-MS is both sensitive and specific, but requires tedious and time-consuming sample preparation. Liquid chromatography-MS (LC-MS) and liquid chromatography-tandem MS are equally specific and offer simpler approaches to sample preparation often without such complex sample derivatization steps. Recently, a number of LC-MS-based methods using different ion sources have been reported for the...
PUM
| Property | Measurement | Unit |
|---|---|---|
| mass spectrometry | aaaaa | aaaaa |
| mass | aaaaa | aaaaa |
| reactive mass | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More