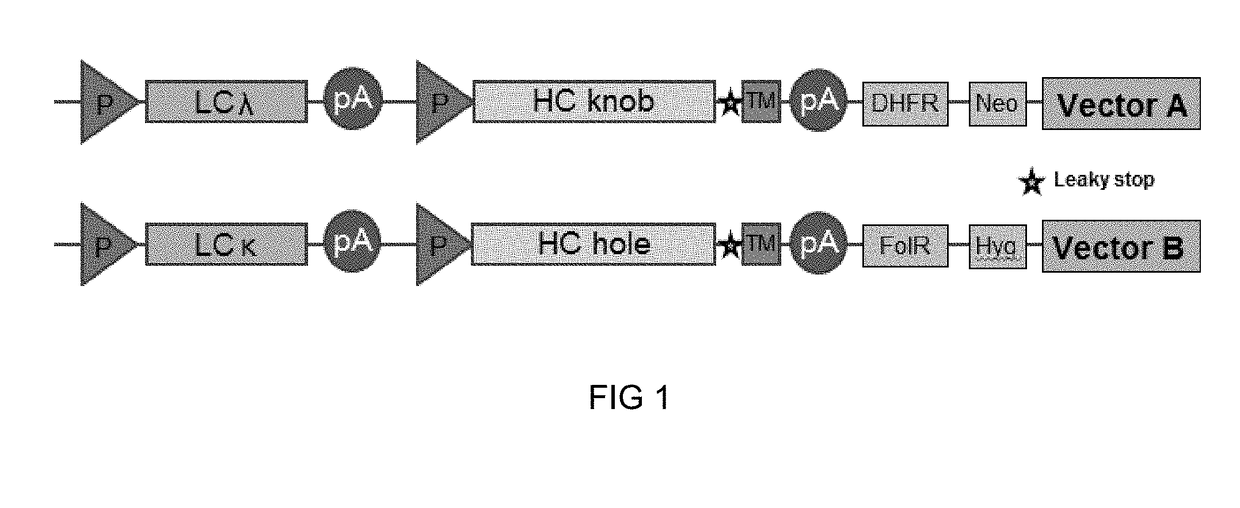
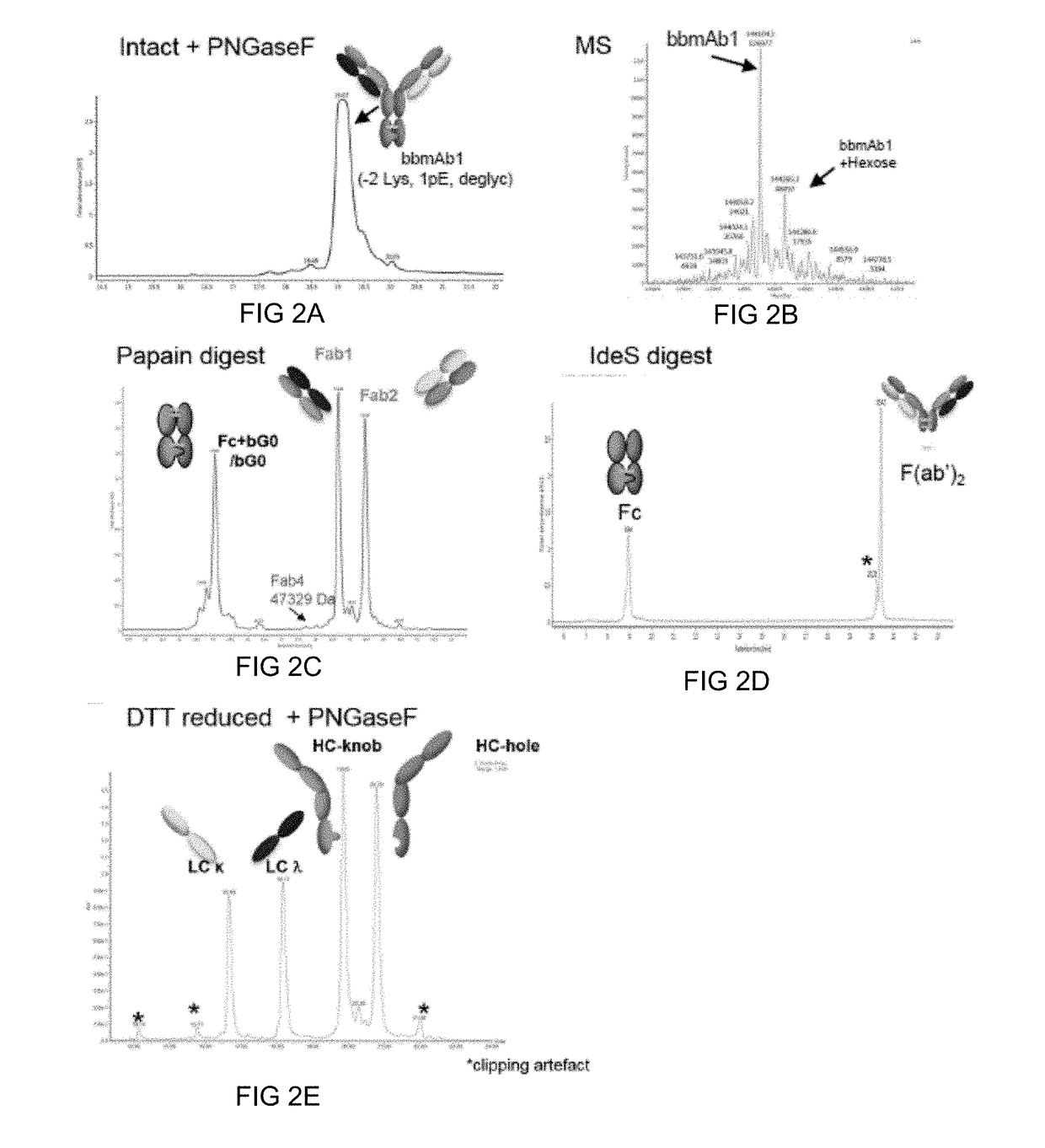
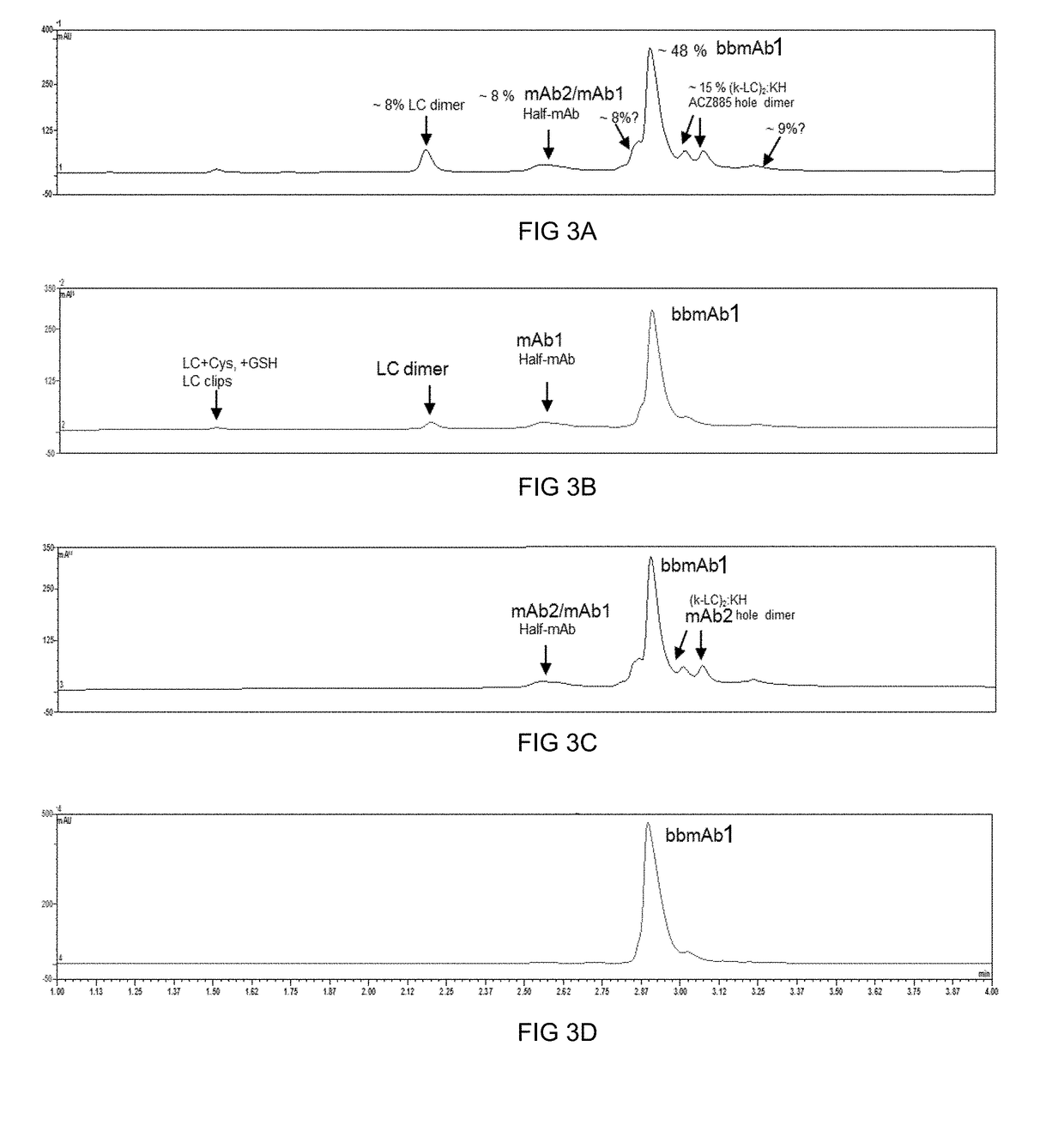
Method of manufacturing bispecific antibodies, bispecific antibodies and therapeutic use of such antibodies
a technology of bispecific antibodies and antibodies, applied in the field of bivalent bispecific monoclonal antibodies, can solve the problems of difficult depletion of related impurities, and achieve the effects of reducing the number of antibodies
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 3
7. Effects of Combined IL-1β and IL-1β Stimulation and Blockade in PBMC
[0323]Inflammasome activation-dependent cleavage of the effector cytokines IL-1β and IL-1β leads to the induction of secondary pro-inflammatory mediators and promotes immune cell recruitment / activation not only systemically but also at the site of inflammation. In two different mouse models for lethal systemic inflammation (a) LPS injection model and (b) FCAS mice (activating missense mutations in NLRP3), the simultaneous absence / inhibition of both IL-1β and IL-1β was more protective from lethality compared to the single IL-1β or single IL-1β absence / inhibition, demonstrating additive or synergistic mechanisms for immune activation (Brydges 2013, van den Berghe 2014). bbmAb1 is a human / marmoset IL-1β / IL-1β reactive bi-specific mAb with no rodent cross-reactivity and thus cannot be tested in mouse models. Therefore, we used LPS / IL-12 to mimic inflammasome-dependent pathway activation in vitro for the stimulation ...
example 5
9. Pharmaceutical Compositions
[0371]Provided herein are pharmaceutical compositions comprising the bbmAb antibodies, such as bbmAb1, formulated together with a pharmaceutically acceptable carrier. The compositions can additionally contain one or more other therapeutic agents that are suitable for treating a medical condition. Pharmaceutically acceptable carriers enhance or stabilize the composition, or can be used to facilitate preparation of the composition. Pharmaceutically acceptable carriers include solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents, and the like that are physiologically compatible.
[0372]A pharmaceutical composition described herein can be administered by a variety of methods known in the art. The route and / or mode of administration vary depending upon the desired results. It is preferred that administration be intravitreal, intravenous, intramuscular, intraperitoneal, or subcutaneous, or administe...
PUM
| Property | Measurement | Unit |
|---|---|---|
| pH | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
| pH | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More