Stable formulations of insulinoptropic peptides
A technology of insulin-stimulating peptides and preservatives, applied in the field of pharmaceutical preparations
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
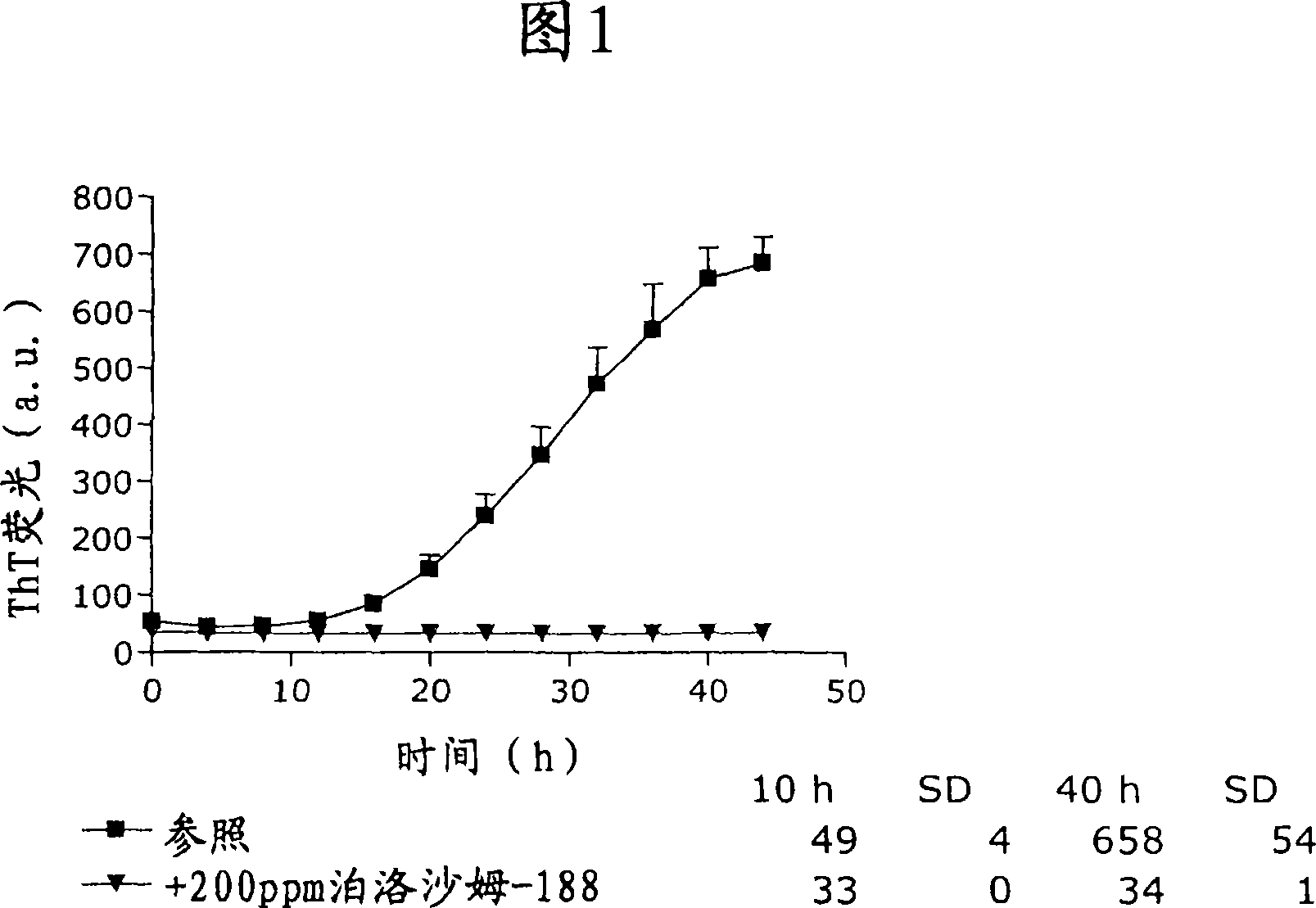
[0162] The ThT fibril formation assay of the pharmaceutical composition of the acylated GLP-1 analog liraglutide is shown in Figure 1 (the experiment was performed according to the method described in "General Methods"). ThT fluorescence emission increased after approximately 10 hours, indicating the onset of fibril formation. The signal increased steadily and reached a plateau before the assay was terminated. However, in the presence of 200 ppm poloxamer 188, the ThT fluorescence signal remained at background levels. This indicates that no fibril formation occurs and thus the pharmaceutical composition is physically stable under these conditions. The pharmaceutical composition used in Example 1 (Fig. 1) had no added buffer.
Embodiment 2
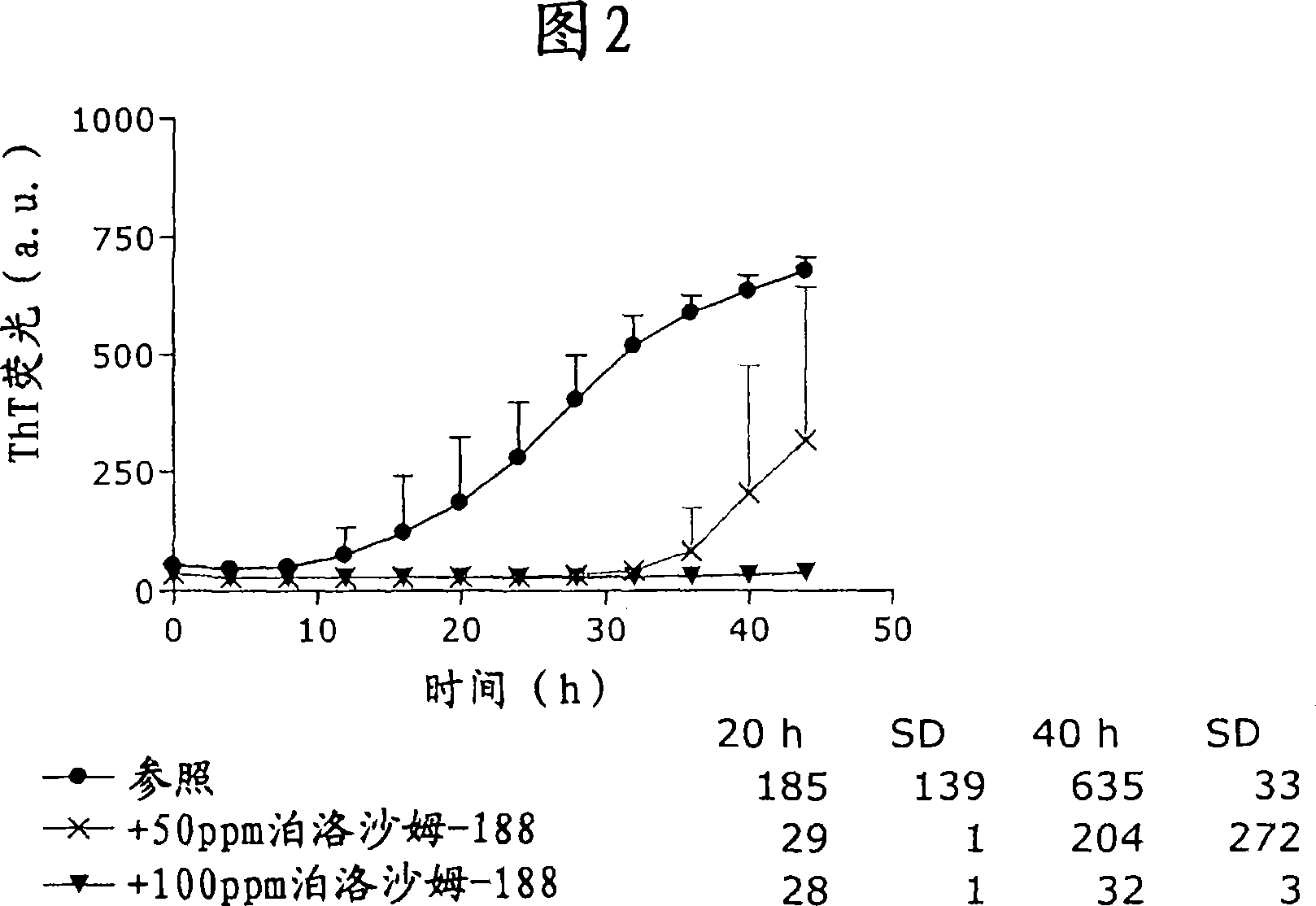
[0164] The effect of poloxamer 188 in liraglutide pharmaceutical compositions comprising sodium phosphate as buffer is shown in Figure 2 (experiments were performed as described in "General Methods"). Here the presence of 50 ppm poloxamer 188 prolongs the lag time before the onset of fibril formation, whereas 100 ppm poloxamer 188 completely inhibits fibril formation during the assay.
Embodiment 3
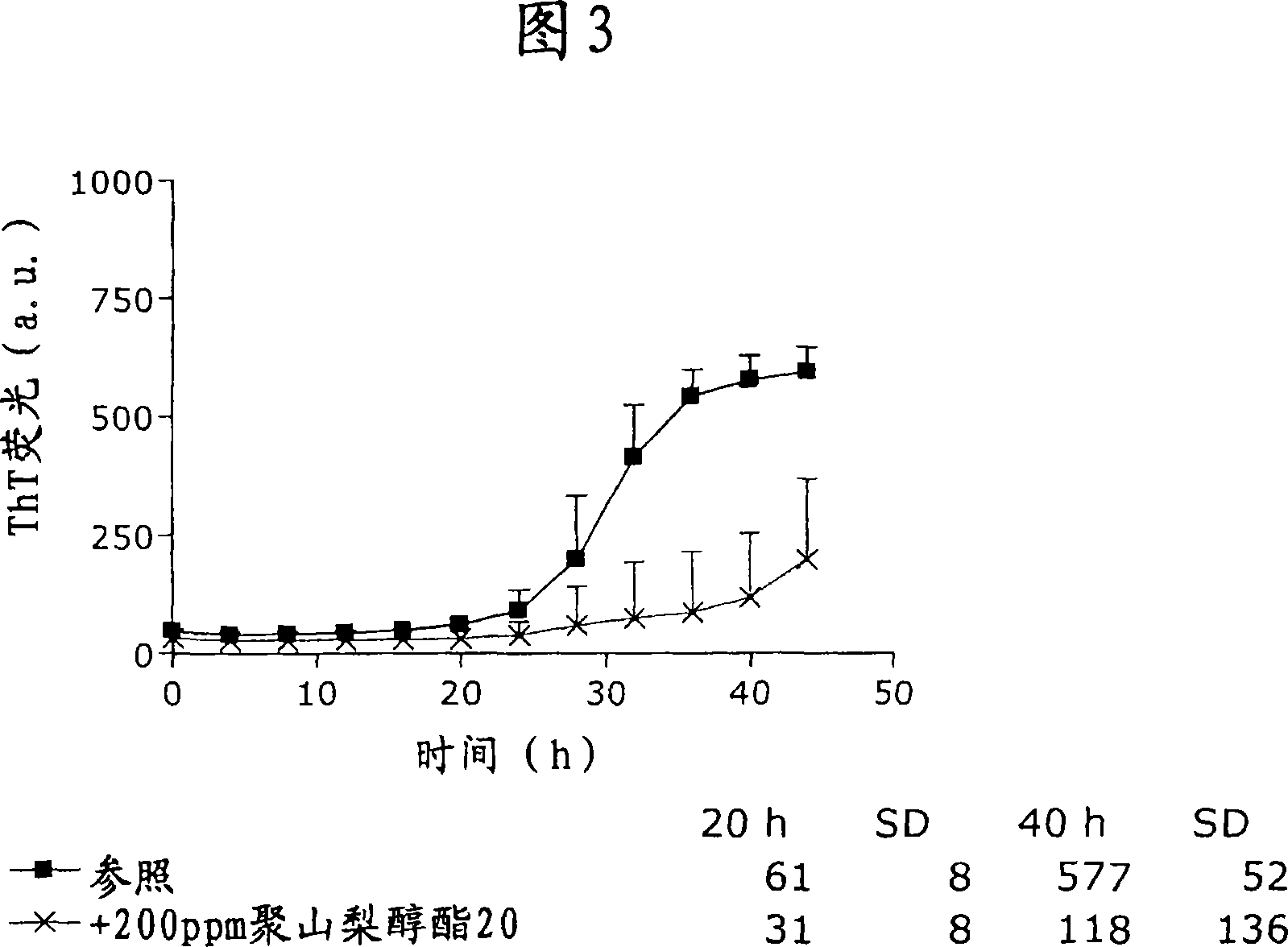
[0166] Polysorbate 20 also stabilized formulations of liraglutide. One such example is shown in Figure 3 (experiments were performed as described in "General Methods"). The presence of 200 ppm polysorbate 20 attenuated fibril formation, which was observed as a slower growth rate of the ThT fluorescent signal. Thus, significantly less ThT fluorescence signal was observed in the polysorbate 20 samples than in the reference after 40 hours of incubation.
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More