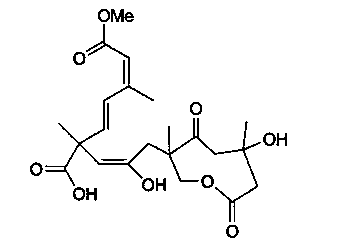
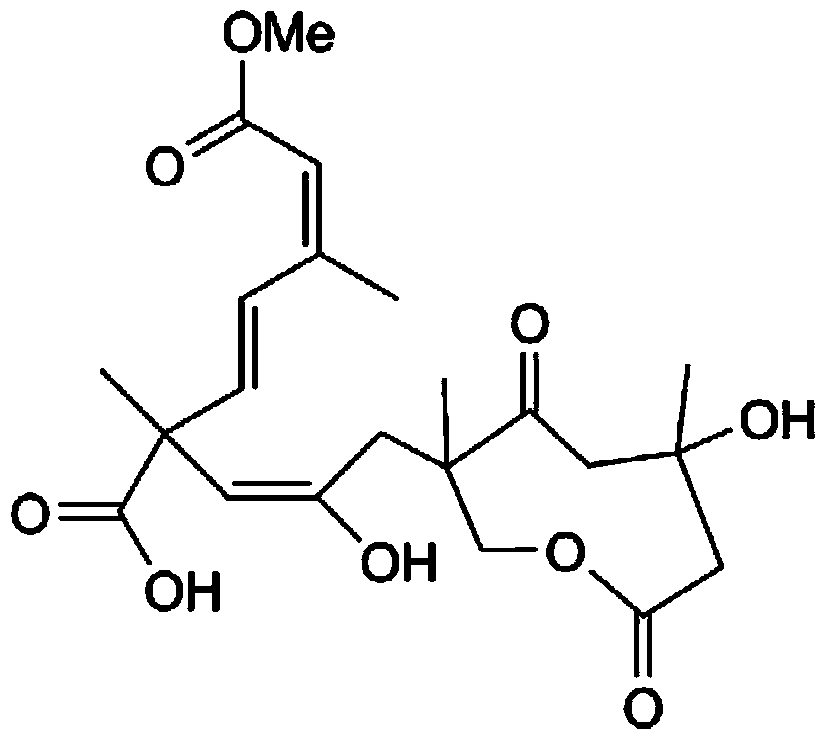
Application of Lycojaponicumin B in preparation of medicines for resisting human body fungi
An antifungal and drug technology, applied in antifungal agents, medical preparations containing active ingredients, pharmaceutical formulations, etc., can solve the problems of digestive tract irritation, strong accumulation of toxicity, etc. active effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0013] Example 1: Preparation of Sarcaboside B tablets, the compound involved in the present invention:
[0014] Take 20 grams of the compound Sarcaboside B, add 180 grams of conventional excipients for preparing tablets, mix them, and prepare 1,000 tablets with a conventional tablet press.
Embodiment 2
[0015] Example 2: Preparation of Sarcaboside B capsules of the compound involved in the present invention:
[0016] Take 20 grams of the compound Sarcaboside B, add 180 grams of conventional auxiliary materials for preparing capsules, such as starch, and mix them, then encapsulate them to make 1000 tablets.
[0017] The following pharmacodynamic experiments will further illustrate its drug activity.
experiment example
[0018] Experimental example: Sarcaboside B anti-human fungal activity
[0019] The anti-human fungal activity test is to use the concentration dilution method, and each test is repeated three times. The test pathogens are Trichophyton rubrum, Microsporum laniformis and Trichophyton spp. The concentration of the bacterial solution is 10 5 Pieces / mL. The initial concentration of Sarcaboside B is 50.0μg / mL (5% dimethyl sulfoxide DMSO), and the gradient is diluted to 0.098μg / mL. The same volume of bacteria liquid and test sample are mixed and cultured in a 96-well plate. Human fungal culture temperature Respectively, observe at 28°C for 24 hours of incubation. If no colony formation is found, it is the lowest anti-human fungal concentration of the sample, that is, the MIC value. The positive control of this experiment is ketoconazole, and the results of Sarcaboside B against human fungi are shown in Table 1.
[0020] Table 1 Sarcaboside B anti-human fungus MIC value (μg / mL)
[0021]
...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More