Early detection and diagnosis kit and device for Parkinson's disease
A diagnostic kit and technology for Parkinson's disease, applied in the field of biomedicine, can solve the problems of unable to meet the needs of rapid diagnosis, time-consuming and labor-intensive data analysis, unbearable for patients, etc. less traumatic effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0039] Screening experiment: In this embodiment, the specific technical methods used are as follows:
[0040] 1. Subjects
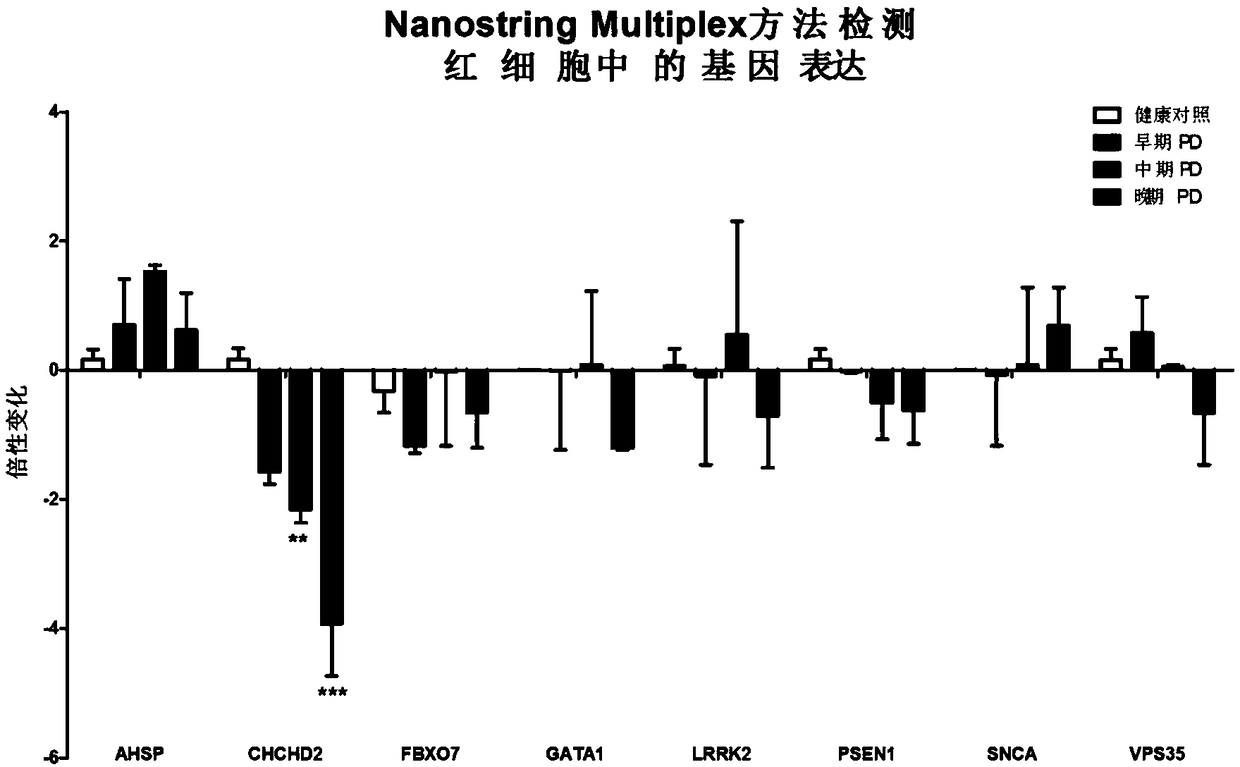
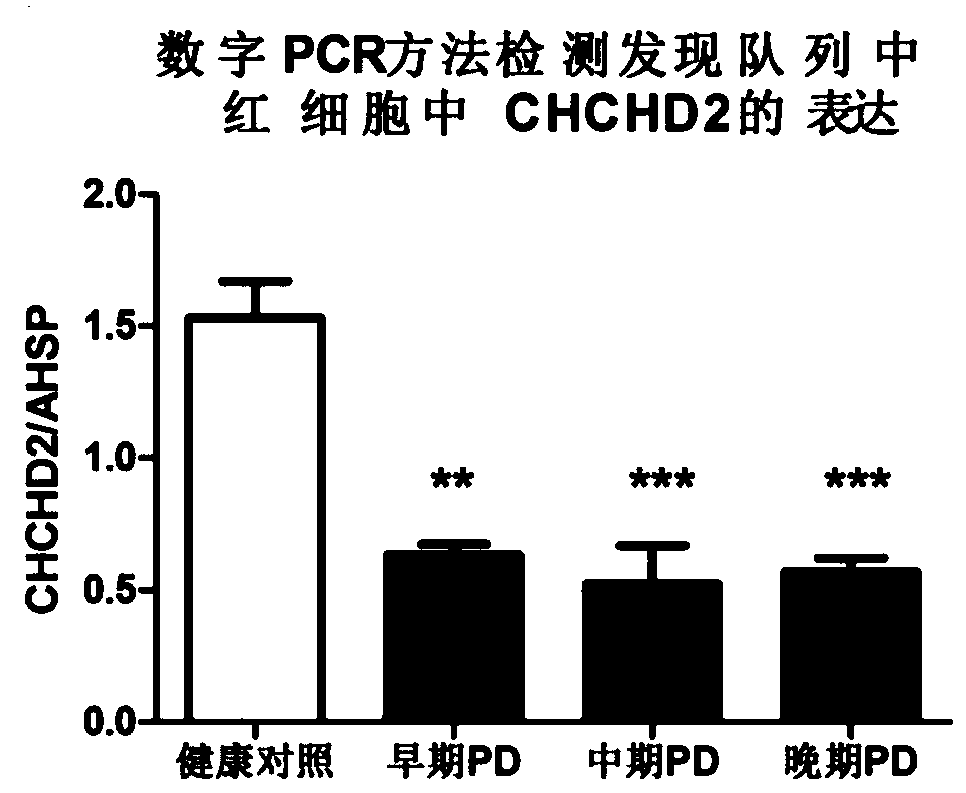
[0041] This study was approved by the ethical review boards of all participating institutions. Both the discovery cohort (total of 48: 12 healthy controls, 36 PD patients) and the validation cohort (total of 340: 135 healthy controls, 205 PD patients) were from Beijing Tiantan Hospital. All participants provided informed consent and underwent medical history taking, neurological examination, laboratory tests, and psychological evaluation by a neurologist. All control subjects were community volunteers with mental status examination scores >27, paragraph recall scores >6, no history of neurological disease, and no history or evidence of cognitive or functional decline. All PD patients met the clinical diagnostic criteria of the UKPD social brain bank. PD patients were further classified to approximate disease stage based on the UPDRS Part III state-moto...
Embodiment 2
[0064] In this embodiment, the components of the kit are as follows in Table 1:
[0065] Table 1
[0066] SuperScript TM III RT Reverse Transcriptase / Platinum TM Taq Enzyme Mix
2X mix buffer
Contains 0.2mM dNTPs, 3mM MgS0 4
CHCHD2 probe
200nM
CHCHD2 upstream and downstream primer pair
500nM each
AHSP probe
200nM
AHSP upstream and downstream primer pair
500nM each
droplet stabilizer
1x
h 2 o
[0067] Note: CHCHD2 / AHSP probes and primers are the same as those of digital PCR in Example 1.
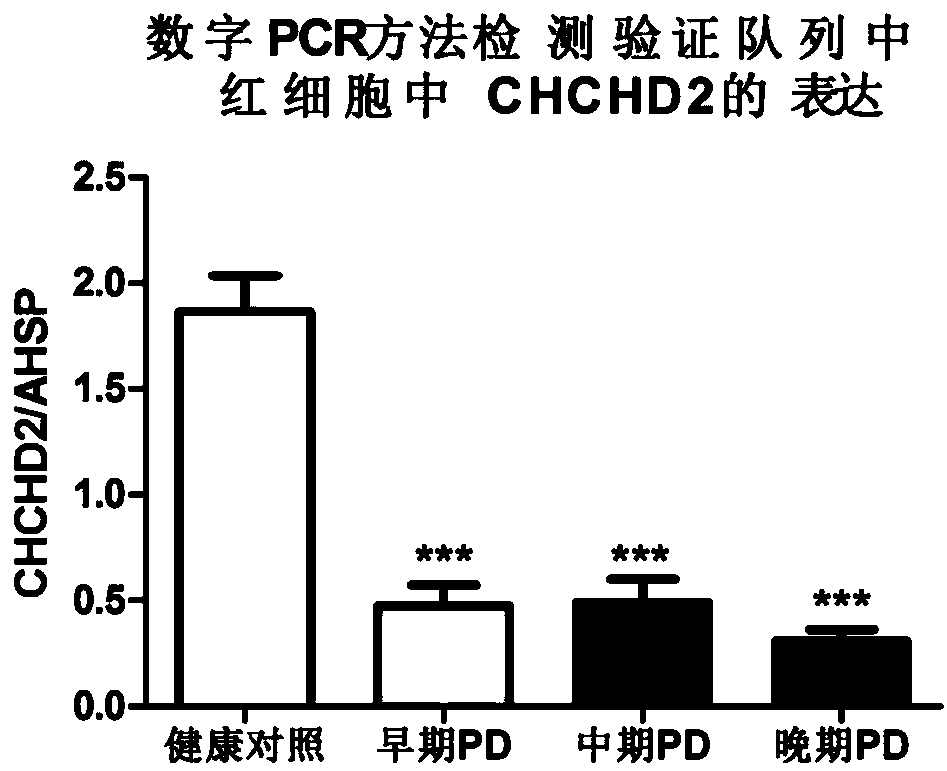
[0068] Subsequently, 135 healthy controls and 205 PD patients were tested using the above kit components by digital PCR method, and the results were consistent with our results in the discovery cohort ( Figure 2A ). The ROC curve showed that the sensitivity and specificity of CHCHD2 in detecting PD exceeded 80%, suggesting that it has a very good diagnostic value. Compared with healthy con...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap