Nano-assembly based on immune checkpoint inhibitor and its preparation method and application
A technology of methylation and reaction, applied in the field of medicine, can solve the problems of low treatment response rate, achieve good biocompatibility, strengthen immune response, and relieve tumor immunosuppressive environment effects
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment approach
[0046] The third embodiment of the present invention provides nano-assemblies based on immune checkpoint inhibitors, including the above-mentioned polymers, hyaluronic acid grafted with chlorin (HA-Ce6) and PD-L1 monoclonal antibody ( aPD-L1).
[0047] The fourth embodiment of the present invention provides the preparation method of the above-mentioned nano-assembly. The aqueous solution of the above-mentioned polymer is added to the aqueous solution of grafted chlorin hyaluronic acid, and after mixing uniformly, the polymer and the grafted dihydrogen are obtained. The nanocomposite HC / PM of hydroporphin hyaluronic acid, the solution of PD-L1 monoclonal antibody is added dropwise to the stirred HC / PM solution to obtain a nanoassembly, which is aPD-L1@HC / PM nano Complex.
[0048] In one or more examples of this embodiment, ultrasound is used for uniform mixing.
[0049] In one or more examples of this embodiment, the mass ratio of the polymer to the grafted chlorin hyaluronic...
Embodiment 1
[0055] Example 1: PLL-1-mt molecular synthesis.
[0056] 1) Boc protection of 1-methyl-D-tryptophan amino group (Boc-1-mt): use an analytical balance to accurately weigh 1-methyl-D-tryptophan, NaHCO 3 and di(tert-butyl)dicarbonate were dissolved in a mixed solution of water and tetrahydrofuran at a volume ratio of 1:1, and the mixture was stirred at 0°C for 10 minutes, and then kept at room temperature for 24 hours. After the reaction, the THF was spin-dried, and the aqueous layer was acidified to pH 1.0 with 1M HCl, and then extracted with ethyl acetate. Evaporation of ethyl acetate gave a milky white solid which was the target product Boc-1-mt.
[0057] (2) Synthesis of Boc-1-mt active intermediate ester (Boc-1-mt-NHS ester1): a certain amount of N-hydroxysuccinimide and 1-ethyl-(3-dimethylaminopropyl) carbon Diimine hydrochloride was dissolved in anhydrous DMF, and Boc-1-mt was added. The reaction mixture was stirred at room temperature for 4 hours and used without furth...
Embodiment 2
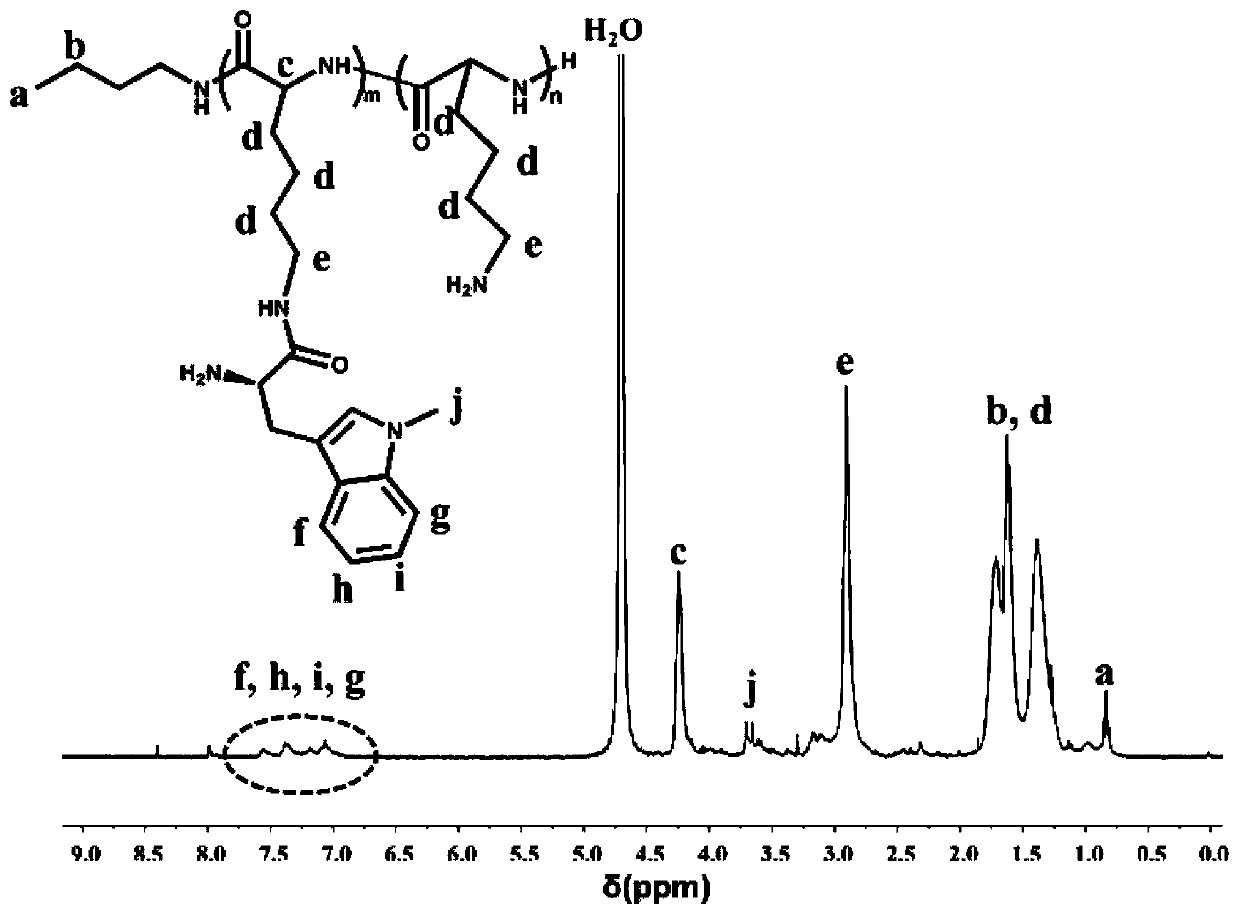
[0060] Embodiment 2: proton nuclear magnetic resonance spectrum ( 1 H-NMR) to identify the molecular chemical structure of PLL-1-mt.
[0061] Weigh about 5 mg of PLL-1-mt prodrug and heavy water (D 2 O) dissolved and placed in a nuclear magnetic tube, using 400MHz proton nuclear magnetic resonance spectrum to measure its hydrogen nuclear magnetic resonance spectrum, using tetramethylsilane as an internal standard, and recording the chemical shift value (ppm) of the compound. The result is as figure 1 As shown, the NMR results can confirm that the characteristic peaks of PLL and 1-mt appear simultaneously in the newly synthesized molecules. pass 1 H-NMR spectrum can confirm the successful synthesis of PLL-1-mt molecule. Through the nuclear magnetic analysis of the PLL, it is obtained that m+n is 44, and through the ultraviolet spectrum analysis, it is obtained that m is 4 and n is 40.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More