Preparation process of four ingredients in herba cistanche and application of four ingredients
A technology of Cistanche deserticola and Cistanche deserticola water extract, which is applied in the preparation of sugar derivatives, sugar derivatives, sugar derivatives and other directions, can solve the problems of unclear active ingredients, lack of microscopic analysis of medicinal effect substances, etc., so as to reduce costs, pollution, The effect of strong repeatability and stable process quality
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0088] Example 1 Cistanche Extraction Process Investigation
[0089] Soaking time, extraction method, extraction solvent, extraction time, extraction times, solid-liquid ratio and extraction particle size are optimized and investigated by single factor investigation method, and the medicinal material of Cistanche deserticola is cut into less than 1cm 3The small pieces were soaked in water for 1 hour, decocted and extracted, and the number of extractions was 2 times; the investigation items of the orthogonal design experiment were the extraction solvent, extraction time and solid-liquid ratio, and then the response surface design was used to analyze the extraction solvent, extraction time and solid-liquid ratio. The final investigation result is: water is used as the extraction solvent, the extraction time is 1h, and the ratio of solid to liquid is 1:11.85. The investigation is based on the yield of the extract, and the content determination method is based on the chromatograph...
Embodiment 2
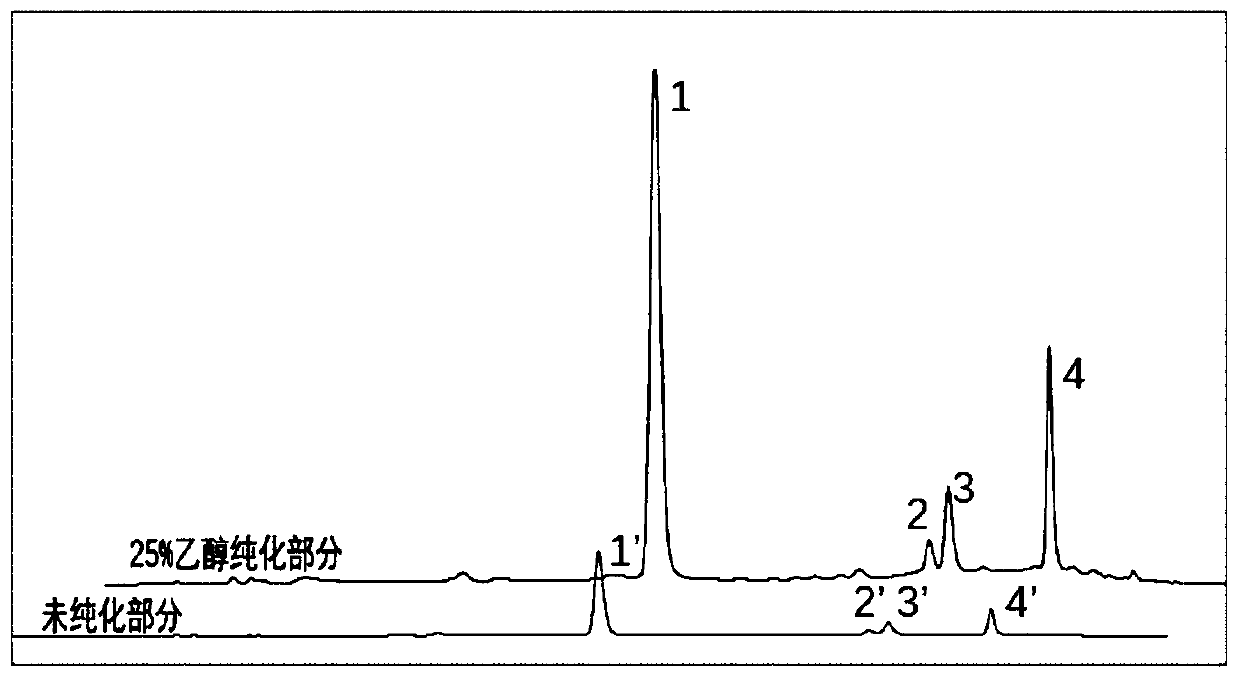
[0104] Macroporous resin purification process investigation of embodiment 2 cistanche extract
[0105] The alcohol precipitation method is used to remove the polysaccharides in the water extract of Cistanche deserticola, and the alcohol precipitation process is optimized from the concentration and density of ethanol. Precipitating the solution, using ethanol with a concentration of 95% to remove the polysaccharides in the Cistanche cistanche water extract obtained in step 1), so that the final concentration of the alcohol precipitating solution is 50-70%, obtaining the supernatant of the alcohol precipitating, and the alcohol precipitating on the The supernatant was allowed to stand at 4°C for 12 hours, and the supernatant was collected by centrifugation. The ethanol was recovered under reduced pressure, and the aqueous solution was lyophilized to obtain the solid matter of the alcohol precipitation supernatant.
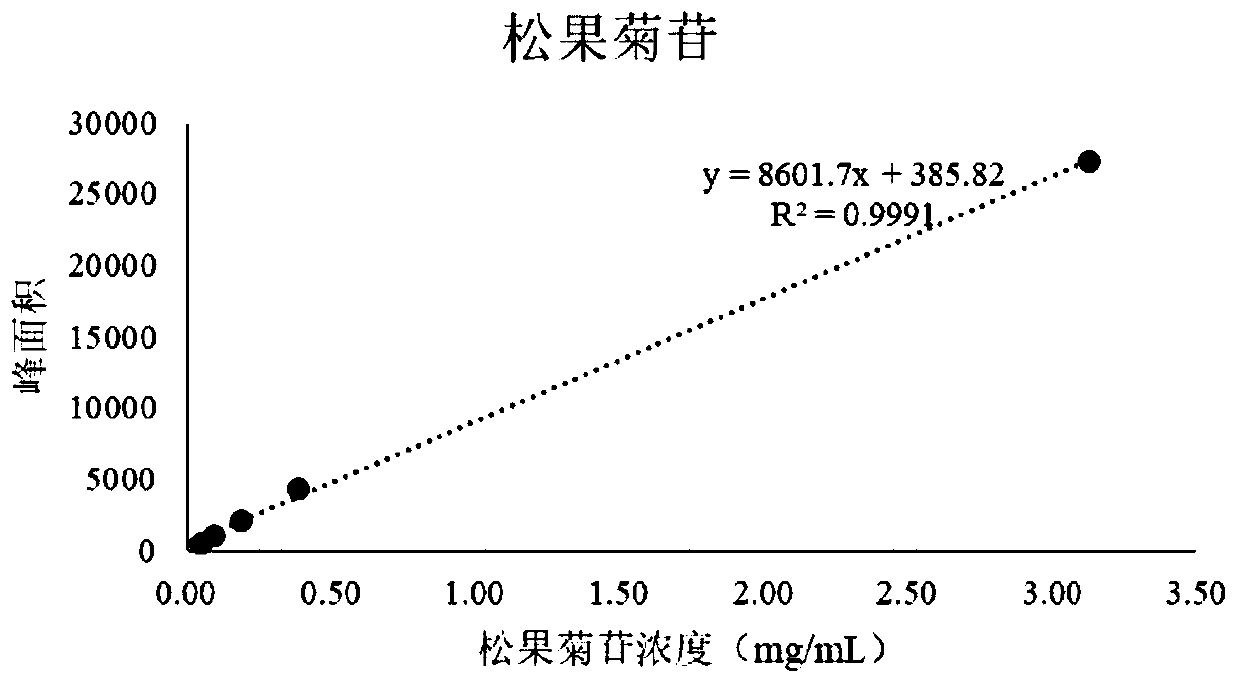
[0106] The optimization index is the transfer rate of echinacos...
Embodiment 3
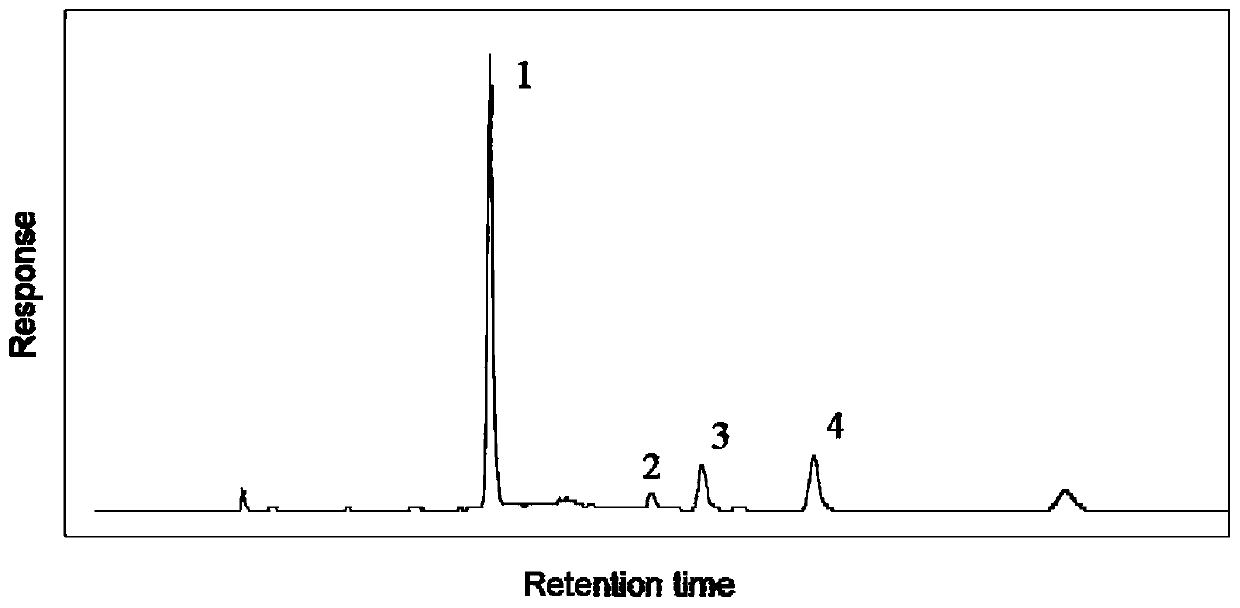
[0115] Example 3 Preparation of four high-purity chemical components using the HPLC-DAD / MS component automatic collection system
[0116] Take Cistanche medicinal material (particle size is less than 1cm 2 ) 250g, placed in a 10L round bottom flask, added 11.85 times the amount of water (2750mL), soaked for 1h, extracted twice, heated and decocted for 1h each time, filtered, and combined the filtrates. Rotary evaporation, concentrate to 357mL (0.7g crude drug / mL), add 773mL 95% ethanol, stand at 4°C for 12h, centrifuge to take the supernatant, rotary evaporation, recover under reduced pressure to remove ethanol, freeze-dry the water to obtain the supernatant of alcohol precipitation Liquid solids 130g.
[0117] Get the alcohol-precipitated supernatant solid after alcohol precipitation, dissolve in water, make the homogeneous clear solution that echinacoside concentration is 2.8mg / mL, take the processed 32g HPD-300 and 32g NKA-9 (wet weight ) Wet packing, put into a glass col...
PUM
| Property | Measurement | Unit |
|---|---|---|
| wavelength | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap