Anti-adhesion and anti-blue light injury care solution for eyes and preparation method thereof
A nursing solution and anti-adhesion technology, which can be applied to medical preparations with non-active ingredients, medical preparations containing active ingredients, and pharmaceutical formulas, etc. It can solve the problems that have not yet been seen, and achieve long storage time and biocompatibility. The effect of good sex and high safety
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Example Embodiment
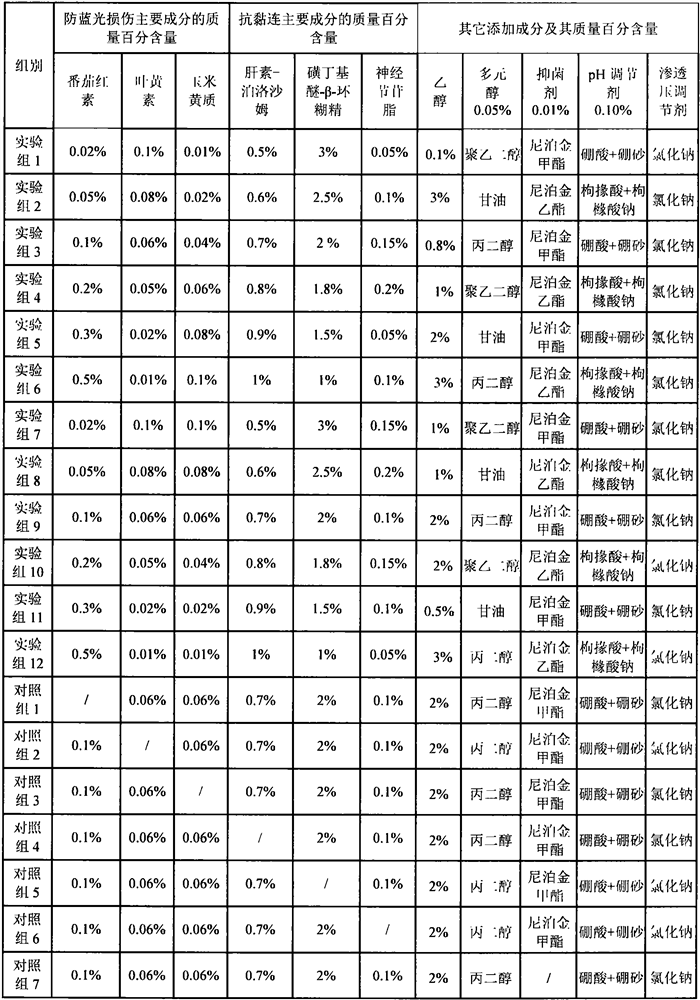
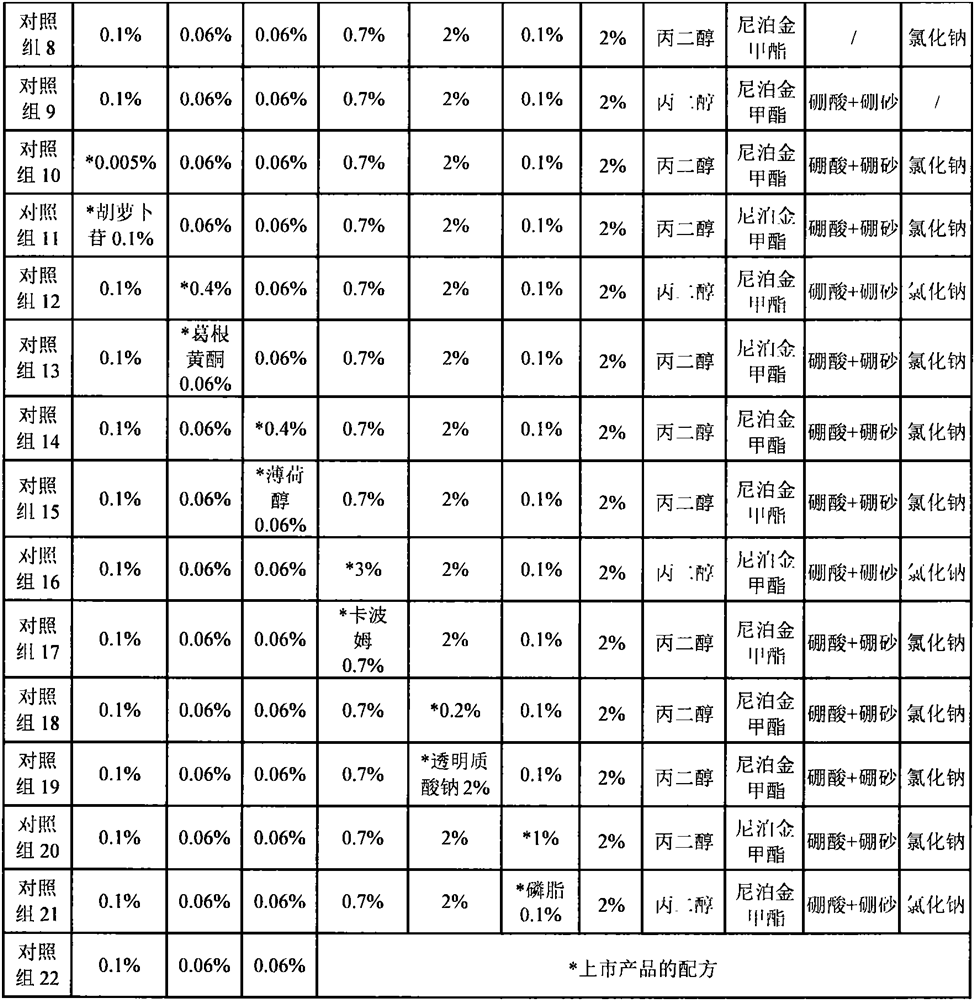
[0026]Example 1 Preparation of samples of each group of eye care solution for anti-adhesion and blue light damage
[0027]According to the component ratios in Table 1, prepare eye care solutions for anti-adhesion and blue light damage. The samples of the experimental group were prepared in a sterile environment as follows:
[0028]Step 1) The heparin-poloxamer copolymer is dissolved in 2 times the mass of ethanol, and lycopene, lutein, zeaxanthin and ganglioside are added and mixed to form a uniform solution;
[0029]Step 2) Add sulfobutyl ether-β-cyclodextrin to water to dissolve, add polyol and mix to form a uniform solution;
[0030]Step 3) Add the solution obtained in step 1) several times in small amounts to the solution obtained in step 2), mix well, add bacteriostatic agent, pH adjuster, osmotic pressure adjuster, and mix well to obtain the anti-adhesion of the eye And anti-blue light damage care solution.
[0031]Control sample: Prepared according to the method of the experimental group.
[0...
Example Embodiment
[0037]Example 2 Stability investigation
[0038]In this example, the eye anti-adhesion and blue light damage prevention solution prepared in Example 1 was used to investigate the stability. The samples of the experimental group and the control group were placed at 20°C for three months in the dark, and the following indicators were observed The change.
[0039](1) Appearance changes: Observe the uniformity of the system with naked eyes, whether there is discoloration, delamination or turbidity, etc.
[0040](2) Changes in the content of lycopene, lutein and zeaxanthin: HPLC method was used to determine the content of lycopene, lutein and zeaxanthin in the system, and to evaluate whether they had changed.
[0041](3) Changes in the number of microorganisms: Refer to the pharmacopoeia method to check the number of microorganisms in the system and evaluate whether they have changed.
[0042](4) Changes in pH and osmotic pressure: Refer to the pharmacopoeia method to measure the pH and osmotic pressur...
Example Embodiment
[0046]Example 3 Evaluation of drug release behavior
[0047]Take 1 mL of each sample of the experimental group and the control group prepared in Example 1, place them in a 3500Da dialysis bag, put the test tube in a beaker containing 1000 mL of distilled water, place it in a 37°C water bath, put a magnetic stirrer, and adjust Rotation speed is 50r / min, sampling at intervals and supplementary sampling with the same volume of distilled water to ensure the sink condition. The sample solution was filtered through a 0.45μm microporous filter membrane, diluted by an appropriate multiple, and then the content of lycopene, lutein and zeaxanthin was determined by liquid chromatograph, and the drug release behavior of the three was counted. According to whether the release curve is stable and the duration of the grading from 1 to 10, "10" represents the best (the release curve is stable and has a long duration), and "1" represents the worst (the release curve fluctuates greatly and lasts for a l...
PUM
 Login to view more
Login to view more Abstract
Description
Claims
Application Information
 Login to view more
Login to view more - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap