Compositions and methods for therapeutic use
a technology of cancer and compositions, applied in the direction of peptide/protein ingredients, p53 protein, peptide sources, etc., can solve the problems of significant morbidity and mortality of bladder cancer, and none of the methods tried to achieve enhanced delivery of a tumor suppressor gene to the bladder for the treatment of bladder cancer
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
experimental examples
Example 1
Ethanol Improves Gene Transfer in the Bladder
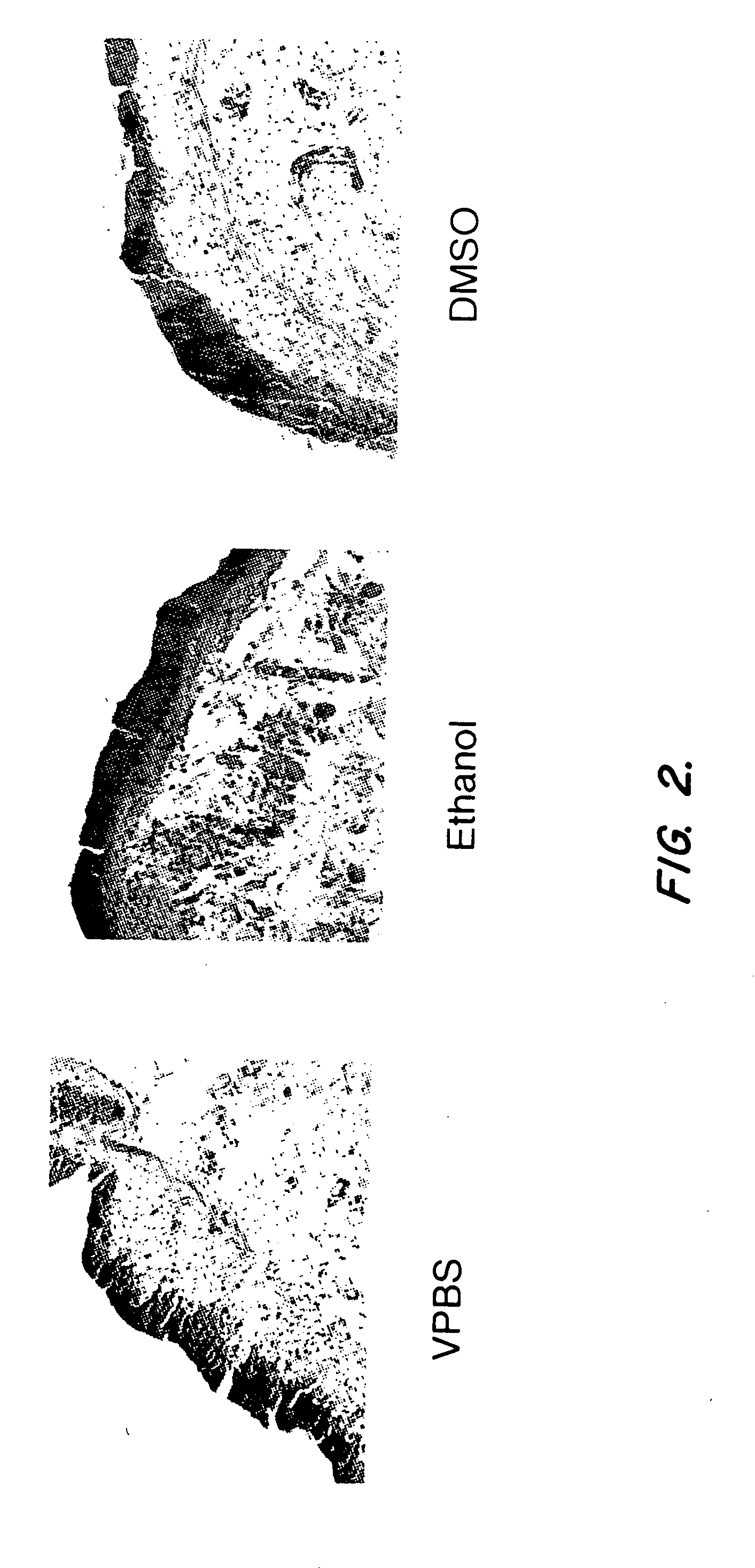
[0039]Initial experiments have shown that several factors including virus concentration, time of administration, and volume of dosing can influence gene transfer to the bladder epithelium after intravesical administration to rats. Because increased penetration of dyes can be achieved by intravesical administration of different solvents, modification of the adenovirus formulation was also investigated as an alternative strategy to increase adenovirus transgene expression in the bladder (Monson et al. Urology 145:842-845 (1991)). The instant experiments focused on the use of ethanol to increase adenovirus transgene expression in the bladder.
[0040]Nine female buffalo rats (Harlan Sprague Dawley) were anesthetized with isoflurane and received a single intravesical administration of a human recombinant adenovirus encoding the lacZ gene (rAd-βgal). The human recombinant adenoviral vector comprising the lacZ gene (rAd-βgal) is described...
example 2
Dose-Dependent Transgene Expression in the Rat Bladder
[0042]In another experiment, 18 female Sprague-Dawley rats were anaesthetized with isoflurane and received a single 0.5 ml intravesical bolus of rAd-βgal at concentrations of 2×107, 2×108, 2×109, 2×1010, and 2×1011, PN / mL in a 22.5% (v / v) ethanol formulation. After a 45 minute incubation, the bladders were flushed with PBS, and animals were permitted to recover from anesthesia. Two days later, animals were sacrificed, and bladders were harvested, fixed, and whole organs were stained with Xgal solution to evaluate adenovirus transgene expression. β-galactosidase expression in the luminal bladder epithelium correlated with the concentration of the administered recombinant adenovirus (FIG. 3). No striking differences were observed among animals receiving 2×1010 or 2×1011 PN / mL, suggesting a saturation of transgene expression in this model: Analysis of the volume voided after instillation indicated only a minimal reduction in the inf...
example 3
ACNRB Gene Transfer in the Mouse Bladder
[0043]A pilot study was conducted to specifically evaluate expression of the RB transgene using a RT-PCR assay. The recombinant adenovirus used in this study was based on serotype 5 human adenovirus from which the viral early region 1 encoding E1a, E1b, and pIX proteins have been deleted. This adenovirus is limited to propagation in 293 cells which produce the Ad5 El gene products required for replication. Transfer plasmids encoding either full length or truncated Rb were generated from pACN (Wills et al. Cancer Gene Therapy 2:191-197 (1995)) and were, in turn, used to construct the recombinant adenoviruses. Either a full-length RB cDNA (1-928 amino acids), subcloned as a 2.8 Kb Xba I-Bam HI fragment from the plasmids pETRbc (Huang et al. Nature 350:160-162 (1991) or a truncated fragment (amino acids 381-928) subcloned as a 1.7 KB Xba 1-Bam HI cDNA fragment, was placed downstream of the CMV promoter / enhancer and the Ad 2 tripartite leader cDNA...
PUM
| Property | Measurement | Unit |
|---|---|---|
| time | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More