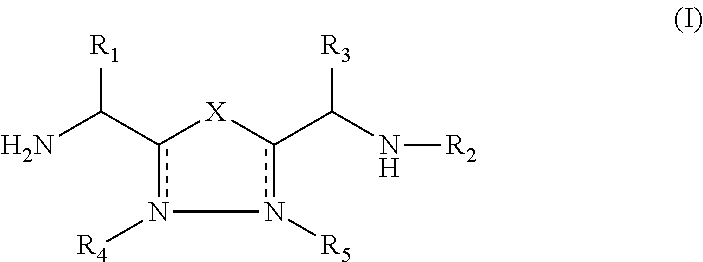
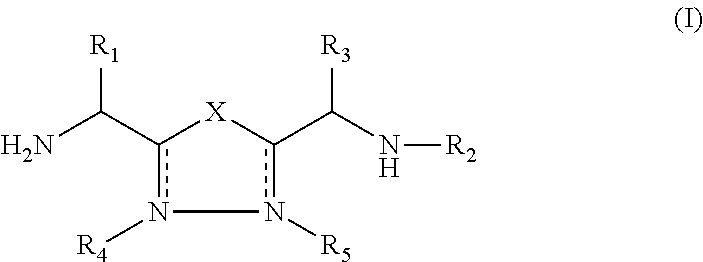
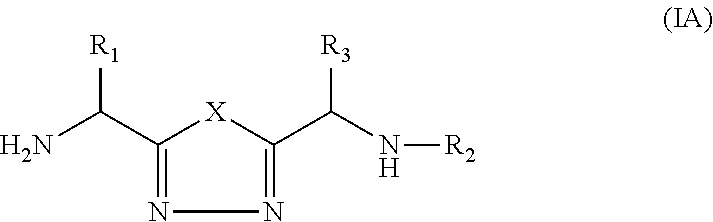
1,3,4-Oxadiazole and 1,3,4-Thiadiazole Derivatives as Immunomodulators
a technology of oxadiazole and thiadiazole, which is applied in the direction of heterocyclic compound active ingredients, biocide, drug compositions, etc., can solve the problems of immune tolerance breakdown and pathogenic autoimmunity, and achieve the effect of suppressing and/or inhibiting the programmed cell death 1 (pd1) signaling pathway
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Synthesis of Compound 1
Step 1a:
[0122]
[0123]Potassium carbonate (7.9 g, 57.39 mmol) and Methyl iodide (1.3 mL, 21.04 mmol) were added to a solution of compound 1a (5.0 g, 19.13 mmol) in DMF (35 mL) and stirred at room temperature for 2 h. The completeness of the reaction was confirmed by TLC analysis. The reaction mixture was partitioned between water and ethyl acetate. Organic layer was washed with water, brine, dried over Na2SO4 and evaporated under reduced pressure to get 5.0 g of compound 1b (Yield: 96.1%). LCMS: 176.1 (M-Boc)+.
Step 1b:
[0124]
[0125]Hydrazine hydrate (7.2 mL) was added to a solution of compound 1b (5.0 g, 18.16 mmol) in methanol (30 mL) and stirred at room temperature for 2 h. The completeness of the reaction was confirmed by TLC analysis. The reaction mixture was evaporated under reduced pressure, the residue obtained was partitioned between water and ethyl acetate. Organic layer was washed with water, brine, dried over Na2SO4 and evaporated under reduced pressure...
example 2
Synthesis of Compound 2
Step 2a:
[0138]
[0139]NMM (1.8 mL, 18.15 mmol) was slowly added to a stirred solution of 1c (2.0 g, 7.26 mmol), 2d (4.3 g, 7.26 mmol), HOBt (1.17 g, 8.7 mmol) and EDC.HCl (1.66 g, 8.7 mmol) in DMF (15 mL) at 0. The reaction mixture was stirred at room temperature for 12 h. The completeness of the reaction was confirmed by TLC analysis. The reaction was quenched with ice, the solid precipitated was filtered and dried under vacuum to afford 3.7 g of pure product 2e (Yield: 59.6%). LCMS: 854.4 (M+H)+.
Step 2b:
[0140]
[0141]To a stirred solution of 2e (3.7 g, 4.33 mmol) dissolved in dry THF (25.0 mL) and DMF (10.0 mL), triphenylphosphine (2.28 g, 8.66 mmol) and iodine (2.2 g, 8.66 mmol) were added at 0. After the iodine was completely dissolved, Et3N (2.5 mL, 17.32 mmol) was added at same temperature. The reaction mixture was stirred at room temperature for 4 h. The completeness of the reaction was confirmed by TLC analysis. The reaction was quenched with ice water and...
example 3
Synthesis of Compound 3
Step 3a:
[0148]
[0149]Lawesson's reagent (2.85 g, 7.03 mmol) was added to a solution of compound 2e (4 g, 4.68 mmol) in THF (40 mL) and stirred at 75° C. for 4 h. The completeness of the reaction was confirmed by TLC analysis. The reaction mixture was evaporated under reduced pressure and the obtained residue was partitioned between ice water and ethyl acetate. The organic layer was washed with NaHCO3 solution followed brine solution. The organic layer was dried over Na2SO4, filtered and evaporated under reduced pressure to get residue which was further purified by silica gel column chromatography (eluent: 0-5% ethyl acetate in hexane) to afford 2.7 g of compound 3a (Yield: 67.66%). LCMS: 852.3 (M+H)+,
Step 3b:
[0150]
[0151]Fmoc group on compound 3a was deprotected by adding diethylamine (3.8 mL) to the solution of compound 3a (1 g, 1.17 mmol) in CH2Cl2 (3.8 mL). The reaction mixture was stirred at room temperature for 30 min. The resulting solution was concentrate...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Immunogenicity | aaaaa | aaaaa |
| Antiproliferative | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More