Reactive acid red dye for nylon and preparation method thereof
A acid red, reactive technology, applied in dyeing, azo dyes, organic dyes, etc., can solve the problems of dye falling off, poor fastness, fading and discoloration, etc., and achieve good fastness performance, good wet fastness, The effect of strong binding ability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
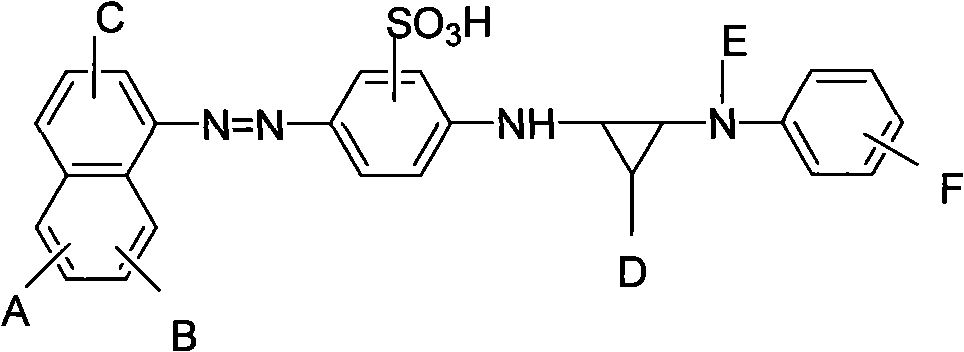
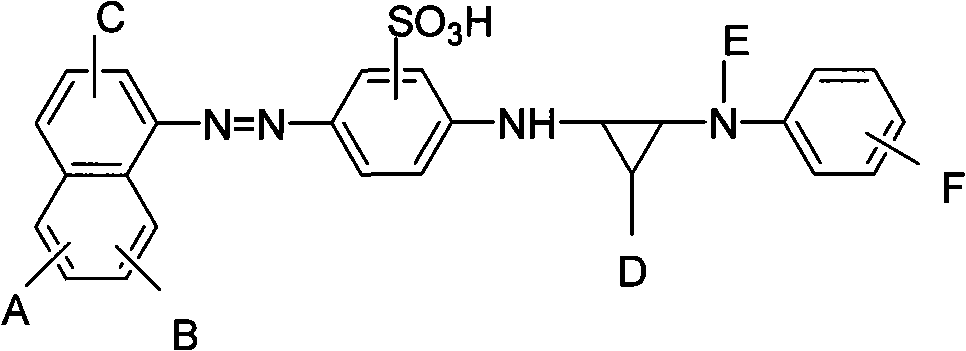
[0067] Synthetic reactive acid red dye, its structure is as follows:
[0068]
[0069] in:
[0070] A=SO 3 H; B=OH; C=NH 2 ; D=Cl; E=H;
[0071] F=SO 2 CH 2 CH 2 OSO 3 H, called a; or F=SO 2 CH=CH 2 , called b;
[0072] The acid red dye is a mixture when F is a and b; a:b=1:0.8-0.1.
[0073] Preparation
[0074] (1) condensation
[0075] Add water and an appropriate amount of ice into the reaction kettle, add cyanuric chloride, stir and beat at 0-5°C, then add m-phenylenediamine sulfonic acid, and react at 0-3°C for 1-3 hours; cyanuric chloride, The ratio of the amount of m-phenylenediamine sulfonic acid is 1:1 (mol / mol)
[0076] (2), diazotization
[0077] Use dilute alkali solution to adjust pH=6.5~7.0, immediately add hydrochloric acid (m-phenylenediamine sulfonic acid: hydrochloric acid: sodium nitrite=1:2.5:1.01 (mol / mol), add ice, slowly add sodium nitrite solution, in 8~10℃, pH=1~2, under the condition of excessive sodium nitrite, carry out diazotizati...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More